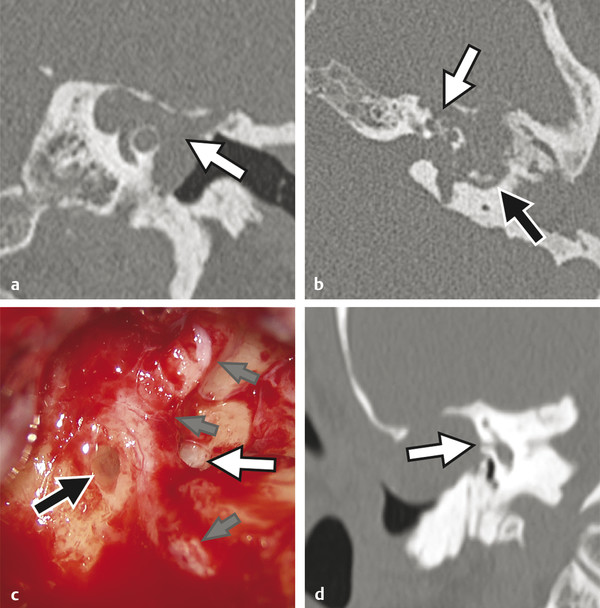
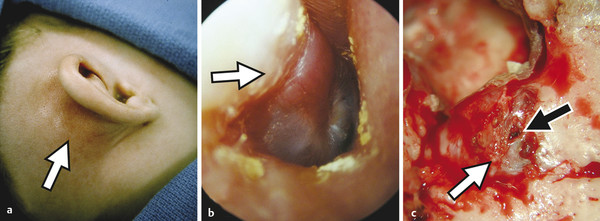
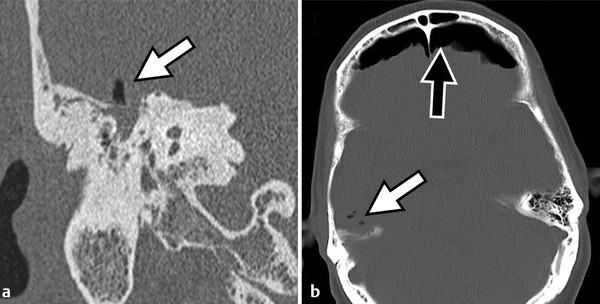
Prior to the commercial availability of antibiotics in the 1930s and 1940s, otologic surgeons were preoccupied with the treatment of life-threatening complications associated with acute and chronic ear disease. Mastoiditis with subperiosteal abscess was a common presentation of acute otitis media (AOM); approximately one in 20 patients with recurrent infection developed intracranial complications, and as many as 2.5% of hospital mortalities were attributed to advanced otitis.1,2,3 Radical mastoid exenteration with mallet and chisel was commonly performed in order to halt advancing infection and marsupialize aural cholesteatoma. The discovery of antibiotics revolutionized the treatment of otitis media; disease-associated morbidity and mortality plummeted and, likewise, the need for surgical intervention significantly decreased. The debut of antibiotics, combined with adoption of the pneumatic dental drill and illuminated magnification, permitted early pioneering surgeons the unprecedented opportunity to focus on hearing restoration rather than disease eradication. The modern era of otologic surgery welcomes a new set of challenges with regard to the management of complicated otitis media. Even at a tertiary referral center, the otolaryngologist may practice many years before encountering a patient with petrous apicitis, otogenic meningitis, or cerebral abscess. The low prevalence of advanced complications combined with the effectiveness of modern medical therapy may contribute to complacency, under-recognition of telling symptoms, delay in diagnosis, and unfamiliarity with treatment. Patients with recurrent acute or chronic otomastoiditis have often received dozens of treatments with ototopical and enteral antibiotics, selecting for greater pathogen virulence and increased antibiotic resistance. Finally, the mounting number of patients with compromised immunity from conditions such as advanced age, diabetes, chronic glucocorticoid use, and immunosuppressive drugs may lead to opportunistic infection and masked symptoms. Whereas chronic ear disease is the primary focus of this book, the current chapter concentrates on intratemporal and intracranial complications of otitis media, which may result from acute or chronic infection. Following a general overview, a focused discussion of individual complications is presented, concentrating on disease presentation, diagnostic evaluation, treatment, and outcomes. Complications of otitis media involve spread of disease beyond the confines of the pneumatized tympanomastoid space to adjacent structures. The development of one complication implies aggressive disease and warrants vigilance to rule out other concurrent or subsequent complications.4,5 For example, the incidence of labyrinthine fistula is much higher in patients presenting with facial paralysis, and the finding of cerebritis may indicate antecedent epidural abscess or lateral sinus thrombosis.4,5,6,7 Patterns of complication and pathogen involvement are often associated with the chronicity of disease.8,9 AOM most commonly involves Streptococcus pneumonia, Haemophilus influenza, and Moraxella catarrhalis and is more likely to cause facial paralysis, labyrinthitis, and meningitis. Chronic otitis media (COM), on the other hand, is more commonly associated with labyrinthine fistula, encephalocele, lateral sinus thrombosis, subdural empyema, and cerebral abscess and more often involves combinations of Pseudomonas aeruginosa, Staphylococcus aureus, Proteus mirabilis, Escherichia coli, and Klebsiella aeruginosa. There are three primary mechanisms by which infection may spread, including hematogenous seeding, retrograde thrombophlebitis, and direct extension through preformed pathways resulting from congenital or acquired defects.2,4 Cadaveric studies have demonstrated that the facial nerve may lack circumferential bony encasement in more than 50% of temporal bones, most commonly within the tympanic segment.10 Dural dehiscence of the tegmen, tympani and mastoideum can naturally result from congenital growth arrest and herniating aberrant arachnoid granulations.11,12 Inner ear malformations may be associated with the development of perilymphatic fistulae involving the oval and round windows.13,14 Anomalous temporal bone development can result in a dilated petromastoid (subarcuate) canal, persistent tympanomeningeal (Hyrtl’s) fissure, or patent Fallopian canal, all of which are associated with an increased risk of cerebrospinal fluid (CSF) fistula and meningitis.15 Similarly, acquired defects resulting from trauma or surgery, as well as bone erosion from acute coalescence mastoiditis or chronic ear disease, facilitate direct unimpeded spread of infection to important adjacent structures.4 Cholesteatoma is the most common cause of bone resorption from COM and therefore deserves special consideration (▶ Fig. 17.1). The pathophysiology of osseous erosion from cholesteatoma remains poorly defined but is theorized to result from several mechanisms.16 Trapped squamous epithelium and keratin debris continue to expand in a rigidly confined space, exerting increasing pressure on surrounding bone. Cell cycle dysregulation results in accelerated cellular proliferation and hyperkeratosis. Additionally, the secretion of nitric oxide and chemotactic cytokines, such as tumor necrosis factor (TNF)-α, interleukin (IL)-1, and IL-6, results in inflammatory cell recruitment and osteoclast activation. Fig. 17.1 Advanced aural cholesteatoma. (a) Coronal CT demonstrating extensive cochlear fistula (white arrow), invasion of the Fallopian canal, and tegmen erosion. (b) Axial CT revealing erosion of the carotid canal (white arrow) and inner ear (black arrow). (c) Intraoperative view of a right canal wall-down surgery demonstrating a large horizontal semicircular canal fistula (black arrow) and an extensively dehiscent facial nerve (small gray arrows); the oval window is labeled with a white arrow. (d) Coronal CT revealing a large fistula involving the lateral semicircular canal (white arrow). Varying patterns of erosion are observed with different subtypes of disease. Classically, congenital cholesteatoma develops within the anterior-superior middle ear cleft, resulting in anterior tegmen tympani dural dehiscence, ossicular erosion, and involvement of peritubal air cells. Primary acquired cholesteatoma most commonly involves an atticoantral disease process, with resultant retraction of the pars flaccida that characteristically evolves into epitympanic cholesteatoma with associated scutal destruction and early erosion of the incus; however, severe posterior pars tensa retraction in the setting of chronic tubotympanic otitis media and severe tympanic membrane atelectasis is also a common pathway for acquired cholesteatoma that, in contrast to the classic pars flaccida version, originates within the mesotympanum. With progression of either version of acquired cholesteatoma, loss of the stapes superstructure, dehiscence of the tympanic segment of the facial nerve, and lateral semicircular canal fistula may occur. Finally, secondary acquired cholesteatoma, which is derived from squamous epithelial ingrowth into the middle ear from a pars tensa perforation, generally involves the mesotympanum, protympanum, and hypotympanum. All patients suspected of having complicated otitis media should undergo a complete head-and-neck examination, including otomicroscopy and tuning fork testing. Close attention should be given to cranial nerve function, and a fundoscopic examination should be performed in patients with symptoms suggestive of elevated intracranial pressure. Audiometric evaluation should be obtained to evaluate for sensorineural hearing loss, which may occur with otogenic labyrinthitis, labyrinthine fistula, or meningitis. Contrast-enhanced computed tomography (CT) of the head and fine-cut temporal bone sequences should be considered in patients with an atypical presentation or concerning symptoms. Magnetic resonance imaging (MRI) and magnetic resonance venography may be particularly helpful in early detection of cerebritis, brain abscess, and lateral sinus thrombosis. In the absence of contraindications, lumbar puncture should be pursued in subjects with evidence of intracranial spread in order to evaluate opening pressure, cellular differential, protein level, gram stain, and bacterial culture. Early consultation with neurosurgery and infectious disease specialists should be considered. The treatment paradigm for advanced otogenic complications has evolved considerably over the last century. Contemporary management has been significantly influenced by a combination of improved medical therapy, evolving surgical techniques, and advanced clinical imaging. Complications of AOM without coalescent infection are generally remedied by ventilation tube placement, aural toilet, and intravenous antibiotic therapy. Conversely, COM largely remains a surgical disease. Classically, canal wall-down surgery is performed in order to exteriorize cholesteatoma, permit unrestricted drainage, and offer easy in-office surveillance.17 However, within the last several decades, the authors have favored canal wall-up surgery combined with aggressive medical therapy when feasible. The management of otogenic intracranial infection remains highly controversial. Because the morbidity of intracranial surgery may be high in the setting of acute infection, the authors favor initial aggressive medical management of intracranial disease, combined with early surgical treatment of the ear once the patient is deemed neurologically stable. With this approach, craniotomy is often reserved for severe or recalcitrant disease. Regardless of treatment strategy, a multidisciplinary approach, including the otologist, neurosurgeon, neurologist, infectious disease subspecialist, and radiologist, is critical to optimize patient outcomes. The clinical distinction between otitis media and mastoiditis is somewhat nebulous because tympanic space infections invariably involve the mastoid air cells as a result of communication through the aditus ad antrum. Furthermore, asymptomatic mastoid air cell opacification is a common finding in patients with Eustachian tube dysfunction. To clarify, acute mastoiditis implies accumulation of walled-off purulence within the mastoid cavity or subperiosteum. Collecting granulation tissue results in antral block, inhibiting drainage of the growing suppurative process. Building pressure may cause the breakdown of bony trabeculae, manifesting the radiologic diagnosis of “coalescent mastoiditis.” The presence of such coalescence is often used clinically as a threshold that designates a case of mastoiditis as being a complication of otitis media rather than routine disease extension. Septic thrombosis of the mastoid emissary vein or direct mastoid cortex erosion can result in postauricular swelling, posterior canal wall edema, otalgia, and proptosis of the auricle (▶ Fig. 17.2). Extratemporal spread, involving the various deep spaces of the neck and skull base, may also occur and is described by a myriad of eponyms including Bezold’s abscess involving the sternocleidomastoid, Luc’s abscess including the posterior canal wall, and Citelli’s abscess invading the posterior digastric muscle. In contrast to acute infection, chronic mastoiditis commonly follows an indolent course, presenting with conductive hearing loss, otalgia, and persistent effusion or otorrhea. Fig. 17.2 Acute coalescent mastoiditis. (a) Pediatric patient with acute mastoiditis and subperiosteal abscess (white arrow). (b) Otomicroscopy reveals characteristic posterior canal wall erythema and edema (white arrow). (c) Intraoperative view demonstrating mastoid cortex erosion (white arrow) and emissary vein thrombosis (black arrow). The initial management of acute mastoiditis includes empiric antibiotic therapy, covering Streptococcus pneumonia, Staphylococcus aureus, and Haemophilus influenzae.18,19 Early myringotomy with culture can assist with aural toilet and directed antibiotic therapy, which may hasten symptomatic improvement and time to resolution. Thin-slice CT of the temporal bones with and without contrast may be useful in guiding initial treatment and evaluating for associated complications. Cortical mastoidectomy should be considered for patients with radiographic coalescence, those who do not respond to initial medical therapy, subjects with evidence of complicated or progressing mastoiditis, and immunocompromised patients. The petrous apex is continuous with the posterior petrous temporal bone and middle ear, and consequently apicitis may occur as an extension of tympanomastoid disease. As a result of the unique anatomic position of the petrous apex, several distinct signs and symptoms may arise with infection.20,21 An envagination of the tentorium cerebelli at the trigeminal impression holds the gasserian ganglion, forming Meckel’s cave. Additionally, the petrosphenoidal (Gruber’s) ligament creates an inflexible fibrous band over the petroclival fissure and contributes to Dorello’s canal, housing the sixth cranial nerve and the inferior petrosal venous network. Gradenigo’s triad suggests the diagnosis of petrous apicitis and is described by the constellation of ipsilateral otorrhea, retro-orbital eye pain from trigeminal irritation, and abducens palsy from compression within Dorello’s canal.20,21 Similar to the mastoid, anterior petrous air cell opacification must be carefully evaluated to distinguish simple effusion, infection, and diplopic bone.22,23 Petrous apex development is variable; the majority of ears demonstrate poorly pneumatized bone with persisting marrow whereas up to one-third of petrous apices are found to have varying degrees of pneumatization, which may be asymmetric in approximately 5% of individuals. Magnetic resonance imaging with gadolinium is considered the workhorse for evaluating petrous apex lesions and is complemented by temporal bone CT. Heterogeneous enhancement on T1 post-contrast imaging and hyperintensity on T2-weighted MRI, combined with septal breakdown seen on CT, suggest the diagnosis of petrous apicitis. Empiric intravenous antibiotics should be instituted and early surgical drainage should be strongly entertained. Several series have demonstrated successful treatment using directed intravenous antibiotics with or without mastoidectomy and ventilation tube insertion.24 Depending on hearing status as well as the severity of disease, surgical drainage may be achieved through a variety of transtemporal approaches that either violate the inner ear with sacrifice of hearing and balance function (transcochlear or translabyrinthine approach) or spare the inner ear (infracochlear, retrolabyrinthine, and subarcuate approach). Rarely, if a surgeon desires to spare the inner ear but disease location and/or temporal bone pneumatization preclude one of the transtemporal approaches, a middle fossa craniotomy may be used to access disease; however, this approach does not readily establish a drainage pathway into the middle ear and adds the morbidity of temporal lobe retraction in the setting of infection and surrounding edema. Labyrinthitis may result from direct bacterial contamination (suppurative labyrinthitis) or from the diffusion of sterile inflammatory mediators across the oval window or round window membrane (serous labyrinthitis). Clinically, labyrinthitis manifests with sensorineural hearing loss, tinnitus, and severe vertigo. Physical examination yields spontaneous horizontal-jerk nystagmus with the fast phase directed toward the affected ear (irritative) in the immediate acute phase, but this usually will rapidly reverse direction away from the affected ear (hypofunctional) within 24–48 hours as vestibular neurosensory function declines. In addition to audiovestibular function, the labyrinth is important because it represents a critical anatomical barrier separating the middle ear and intracranial space. The inner ear communicates freely with the subarachnoid space via the cochlear aqueduct; therefore, otogenic labyrinthitis threatens to become ascending meningitis and, conversely, primary meningitis risks development of bilateral labyrinthitis. Thin-cut CT of the temporal bones is helpful for evaluating predisposing conditions, such as Mondini malformations or labyrinthine fistula.13,25 MRI will exhibit the sensitive but nonspecific findings of post-contrast T1 hyperintensity and increased fluid-attenuated inversion recovery (FLAIR) signal intensity within the labyrinth and, in addition, can help exclude other conditions such as vestibular schwannoma and stroke.26 Patients with fever, headache, nuchal rigidity, and mental status changes should undergo lumbar puncture to evaluate for ascending meningitis. The distinction between serous and suppurative labyrinthitis is purely based on patient history and convalescence since perilymph analysis is not performed. Therefore, practically speaking, all patients with suspected labyrinthitis should receive glucocorticoids and antibiotic therapy with favorable blood–brain barrier penetration. Early tympanomastoidectomy should be considered in the setting of acute coalescent disease. Patients with recurrent meningitis and congenital malformations of the inner ear may undergo vestibule obliteration.14 All patients acquiring bilateral severe-to-profound sensorineural hearing loss following suppurative labyrinthitis or primary meningitis should be followed closely with T2-weighted MRI, with consideration for early cochlear implantation at the beginning signs of labyrinthitis ossificans. Inner ear fistula, most commonly involving the lateral semicircular canal, occurs in approximately 10% of aural cholesteatoma and is the second most frequent intratemporal complication of chronic ear disease behind ossicular erosion.27 Labyrinthine fistula poses an imminent risk to inner ear function. Sensorineural hearing loss may result from labyrinthitis or mechanical injury to the membranous labyrinth during tympanomastoidectomy. Depressed bone conduction, a positive fistula test, and complaints of imbalance should raise concern over labyrinthine fistula and prompt temporal bone CT. However, because inner ear fistula may be present despite normal testing, the absence of concerning findings does not exclude the possibility of disease.28 Intraoperative findings, including stapes superstructure erosion and facial nerve dehiscence, should indicate an increased possibility of fistula (▶ Fig. 17.1c).29 Management of labyrinthine fistula is complex and controversial. Traditionally, a canal wall-down procedure is performed, and the overlying cholesteatoma matrix is left undisturbed. This approach remains the preferred strategy when operating on an only-hearing ear with labyrinthine fistula.30 Over the last three decades, several centers have reported good success with same-surgery fistula repair for small dehiscence or staged removal at a planned second-look procedure for a large, complex fistula or in the setting of acute inflammation.27,31 With this strategy, a canal wall-up tympanomastoidectomy is performed, and the fistula is addressed in the final stages of cholesteatoma dissection. After delicately removing the opposing matrix, the fistula may be occluded with bone wax or resurfaced with bone paté or fascia. Facial paralysis in the setting of otitis media is a rare and distressing symptom for both the patient and clinician. The treatment and prognosis are strongly associated with onset, severity, and duration of weakness as well as underlying cause.6,32,33,34 Whereas the mechanisms behind facial weakness with suppurative otitis remain speculative, most theorize that nerve irritation and edema result from surrounding osteitis or direct contact in natural areas of Fallopian canal dehiscence, most commonly in the horizontal segment near the oval window. Imposing granulation tissue and infection can result in tracking neural edema, which in turn can lead to vascular “choke” and ischemia. Facial palsy with AOM commonly presents with an abrupt incomplete paralysis.33 Thin-cut temporal bone CT should be obtained to evaluate for coalescence and associated complications. Initial management includes empiric antibiotic therapy and steroids. Additionally, myringotomy with pressure equalization tube placement will facilitate the culture and the administration of ototopical therapy. Patients who do not respond to initial conservative therapy or those with encapsulated infection may benefit from cortical mastoidectomy. Because the prognosis of facial neuropathy with AOM remains very favorable, electrodiagnostic testing and neural decompression is not advocated. In contrast to AOM, the outcome of facial paralysis in the setting of COM with cholesteatoma is more variable.6,32,34 Seventh nerve paralysis may result from local osteitis, direct neural invasion, and, potentially, from compression. Early treatment with antibiotics, steroids, and tympanomastoidectomy remains critical to a good outcome. Patients who undergo early surgical treatment are significantly more likely to recover normal facial motor function compared to those with long-standing neuropathy.34 Facial nerve decompression of the adjacent proximal and distal segment may reduce ischemia with little added morbidity; however, opening of the epineurium raises the theoretical risk of introducing infection and is not recommended. Classically, a canal wall-down approach is utilized, and adherent granulation tissue and cholesteatoma matrix are left in place. The authors have increasingly employed canal wall-up techniques with upfront or staged removal of disease depending upon the degree of inflammation and ease of dissection. Meningoencephalocele and CSF leak may result from a variety of conditions, including trauma, prior surgery, and erosive processes such as cholesteatoma and tumor.35 Over time, recurrent acute or chronic otomastoiditis may thin overlying tegmen, leading to dural exposure and weakening of the meninges. Repetitive dural pulsation, increased intracranial pressure, and age-related dural atrophy may lead to meningeal herniation and CSF fistulization. Temporal bone encephalocele and CSF leak commonly present with symptoms of chronic ear disease, including conductive hearing loss and otorrhea; rarely does clear unilateral rhinorrhea, meningitis, or clinical pneumocephalus occur at disease presentation (▶ Fig. 17.3).35,36 In patients with persistent clear otorrhea or effusion, a β-2-transferrin assay should be performed to confirm suspicion of an encephalocele, and a thin-cut CT of the entire skull base should be performed to diagnose and characterize tegmen dehiscence. Magnetic Resonance Imaging is a useful adjunct to distinguish encephalocele from other pathologies, including effusion, cholesterol granuloma, and cholesteatoma. Fig. 17.3 Skull base erosion. Elderly patient with a history of COM presenting with acute mental status changes, fever, and otorrhea. (a) Coronal CT of the right temporal bone demonstrating a large defect in the tegmen mastoideum with intracranial air (white arrow). (b) Axial head CT revealing extensive pneumocephalus (black arrow) emanating from the right temporal bone defect (white arrow).
17.2 General Considerations
17.3 Intratemporal Complications
17.3.1 Acute Mastoiditis
17.3.2 Petrous Apicitis
17.3.3 Labyrinthitis
17.3.4 Inner Ear Fistula
17.3.5 Facial Paralysis
17.3.6 Meningoencephalocele and Cerebrospinal Fistula
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


