Fig. 15.1
Thyroglossal cysts typically present as midline neck masses at the level of the hyoid bone. (Reprinted from Seminars in Pediatric Surgery, Vol.15, Foley DS, Fallat ME, Thyroglossal duct and other congenital midline cervical anomalies, pp. 70–5, Copyright (2006) with permission from Elsevier.)
TGDC frequently become infected, usually causing a painful erythematous swelling or mass found in the midline or occasionally just left of the midline of the neck [3, 8, 9]. The mass can be associated with regional lymphadenopathy. While most patients lack fever, chills, or signs of systemic disease, abscesses frequently occur [10, 11]. TGDC have a tendency for getting recurrently infected and abscesses will commonly drain to the anterior neck, either spontaneously or via a previous site of surgical drainage. Patients with TGDC will complain of various symptoms including dysphagia, cough, airway obstruction, or a foul taste in the mouth if the TGDC spontaneously drains at the level of the foramen cecum [2, 3]. Complications of TGDC include deep neck infections, which can include extension of the lesion posterior to the hyoid bone with involvement of the preepiglottic space [12]. In rare cases this can result in airway compromise and death [10]. Extension of the infection into the mediastinum, lungs, and visceral vascular space has also been reported [12, 13].
Diagnosis
While a complete history and physical examination can suffice in the workup of a TGDC, imaging is frequently required to confirm the diagnosis. When an infected TGDC is suspected, a contrasted computerized tomography (CT) or magnetic resonance imaging (MRI) of the neck is recommended to define the anatomy and evaluate the extent of the infection.
A comprehensive workup for a TGDC may also include a neck ultrasound and a TSH level. If a midline solid mass or elevated TSH levels are found, a scintiscan can be helpful in the diagnosis of a median ectopic thyroid. Some have advocated that a scintiscan should be performed on any patient with a TGDC, but the radiation exposure and the low yield of positive results mean that a more targeted approach is clinically sound.
Microbiology
TGDC infections occur as a result of accumulation of secretions from the thyroglossal duct epithelial cells. This accumulation leads to dilation of the duct and development of the cystic structure. Low oxygen tension within the cyst and lack of drainage of the cyst secretions leads to bacterial overgrowth. As the cyst is usually connected to the mouth floor, organisms that are frequently identified in infected TGDC are primarily oropharyngeal flora, including streptococcal species and oral anaerobes such as Prevotella, Porphyromonas spp, Bacteriodes spp and Peptostreptococcus spp [3, 10, 14–16]. Skin flora including Staphylococcus aureus and Streptococcus pyogenes are also frequently identified [3, 10, 15–17]. Additional organisms that have been identified in the literature include Haemophilus influenzae [18], Streptococcus pneumoniae and Neisseria meningitidis [19]. Group B streptococcus was reported in an infant with an infected TGDC [20]. A very unusual TGDC infection with coccidioidomycosis has also been reported [21].
Therapeutic Approaches
Empirical antimicrobial therapy should include coverage for oropharyngeal flora. Options include clindamycin or the combination of ampicillin or amoxicillin plus a betalactamase inhibitor (i.e., ampicillin/sulbactam for IV therapy or amoxicillin/clavulanate for PO therapy). Rarely, patients will have severe or life-threatening infections and then most experts recommend ensuring adequate empirical coverage for methicillin-resistant S. aureus such as with vancomycin as part of the regimen while culture results are pending.
The route of antibiotic administration depends on the severity of the infection. Intravenous therapy is recommended for toxic-appearing children, children who are unable to tolerate oral therapy, or children who are at risk of respiratory compromise due to the location or severity of the infection. Oral therapy can be considered for children with only minor infections or in children who are clinically improved after initial intravenous therapy.
Definitive antimicrobial therapy should be directed by culture and susceptibility results. Therefore needle aspiration of an acutely infected cyst early in the course of treatment is optimal. Purulent material should be sent for gram stain and aerobic culture, anaerobic culture, fungal culture, mycobacteria culture, and histopathology review to optimize the opportunity to identify the causative organism(s) responsible for the infection as well as to differentiate a TGDC from other congenital neck lesions [10]. Findings on histopathology review that are consistent with (but not specific for) the diagnosis of TGDC include colloid, macrophages, lymphocytes, neutrophils, and ciliated columnar cells [22].
Given that TGDC are a source of chronic inflammation, they are at risk for infection, and they may become malignant, elective surgical excision is the treatment of choice. Complete excision of the entire thyroglossal duct, including the central portion of the hyoid bone and a core of tissue from the muscle at the base of tongue, is paramount to a successful surgery and significantly reduces the risk of postoperative infection and recurrence. A core excision at the base of tongue is recommended since it has been demonstrated that the duct splits into ductules at that level for many patients with a TGDC. Incomplete resections can lead to recurrences in as many as 70 % of cases [2, 6].
The management of infected TGDC differs from standard neck abscesses. Given the need to subsequently operate on any diagnosed TGDC, a more conservative approach including antimicrobial therapy and needle aspiration is recommended. Most patients respond adequately to this approach. Children with progressive induration or airway compromise, or who are critically ill, may require emergent incision and drainage, but this should be avoided if possible to prevent seeding of ductal cells. Other children with fluctuant abscesses not amenable to antimicrobial therapy alone should undergo complete surgical excision when clinically feasible. Complete surgical excision is generally preferred over incision and drainage, as incision and drainage procedures have been associated with a higher risk of recurrence and can obscure landmarks and damage tissue planes therefore making it more difficult to excise the congenital lesion in the future [2, 15].
Children who are treated with antimicrobial therapy alone should undergo surgical removal of the TGDC and the entire length of the tract after the inflammation has completely resolved (usually approximately 6 weeks after completing antimicrobial therapy). Timing of this surgery can occasionally be challenging given that recurrent inflammation and infection of TGDC is common. Sistrunk procedures done on previously incised and drained TGDC should remove all of the operated skin and subcutaneous tissue via an elliptical excision [2]. Figure 15.2 presents an infected thyroglossal duct cyst specimen in situ with a walled-off abscess inferiorly.
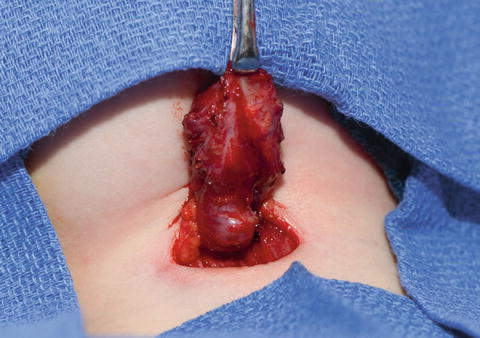

Fig. 15.2
Infected thyroglossal duct cyst in situ with a large walled-off abscess inferiorly in the neck
Controversy exists regarding the management of median ectopic thyroids. Treatment options include medical treatment with suppressive doses of exogenous thyroid hormone or full resection with postoperative thyroid replacement [2]. Given the need to administer thyroid hormone in both cases and the small but present risk of malignancy in exogenous tissue over a patient’s lifetime, many clinicians choose the surgical path for their patients.
Branchial Cleft Cysts, Sinuses and Fistulae
Branchial arch anomalies rank as the second most common congenital cervical anomaly found in children, at 30 % of all congenital head and neck lesions [23]. They comprise cysts, sinuses and fistulae from the first, second, third and fourth branchial arch. Second branchial arch anomalies represent 95 % of all branchial cleft malformations, followed by first, third and fourth branchial arch anomalies [1, 23].
General Branchial Embryology
Branchial arches which will form the head and neck structures start developing at 4 weeks of embryonal development as an external arch covered with ectoderm, an internal pouch covered with foregut endoderm and a midlayer of mesodermal tissue comprising a dominant artery, a nerve, a cartilage rod and muscle [2, 23] (Fig. 15.3). Four out of the six arches are well-defined and form into the structures of the head and neck. Branchial anomalies are remnants of these arches and clefts that fail to regress or obliterate normally [23]. Because the second, third and fourth branchial arches all exit at the cervical sinus of His, a failure of these structures to regress usually ends up in a similar external opening in the neck. However, each type of branchial arch anomaly has a distinct internal course and precise knowledge of its anatomy is crucial during surgical resection to avoid iatrogenic injury to important neck structures and to avoid recurrences [23].
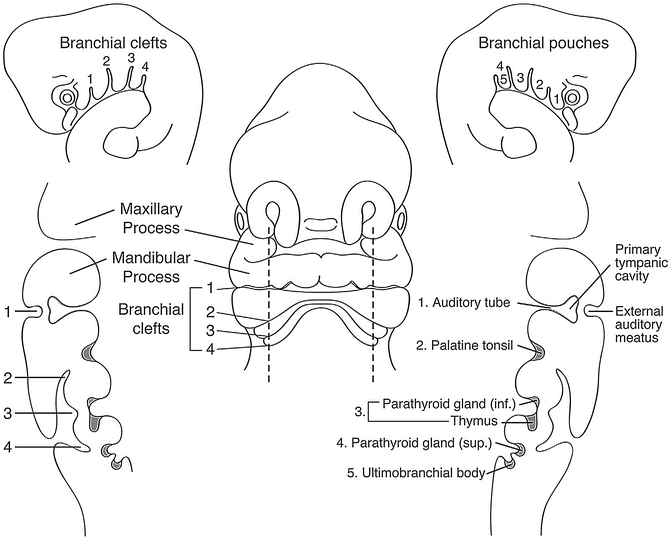

Fig. 15.3
Diagram of the branchial embryology of a 5-week embryo showing how important head and neck structures are derived from branchial clefts and pouches. (Reprinted from Seminars in Pediatric Surgery, Vol. 15, Waldhausen JH, Branchial cleft and arch anomalies in children, pp. 64–9, Copyright (2006) with permission from Elsevier.)
Branchial cysts are usually lined with ciliated and columnar respiratory epithelium, while tracts contain squamous epithelium. In addition, cysts can contain lymphoid tissue and cholesterol crystals within the mucous fluid. Squamous cell carcinoma can be found in branchial cleft lesions, especially in adulthood, although their incidence is extremely rare.
General Clinical Presentation
Infection is very common within branchial anomalies. About 20 % of them have been infected at least once by the time they are excised [24]. Patients with branchial cleft cysts can present with a history of a nontender mass in the neck that appears to fluctuate in size with upper respiratory tract infections [12]. Infected branchial cysts can progress to form abscesses or rupture spontaneously to form draining sinus tracts. Occasionally, internal spontaneous drainage occurs into the pharynx or external auditory meatus. Infants and children with infected branchial cysts can present with respiratory compromise or difficulty swallowing if the cysts enlarge enough to compromise the upper airway or digestive tract [10, 23, 25]. Additional complications include deep neck infections such as parapharyngeal abscesses and mediastinitis [26].
General Microbiology
The microbiology of infected branchial cleft cysts is similar to the microbiology of infected TGDC lesions. Oropharyngeal flora (including streptococcal species and anaerobes such as Eikenella corrodens, Bacteroides species, and Actinomyces odontolyticus), skin pathogens (such as S. aureus and S. pyogenes), and occasionally middle ear/upper respiratory pathogens (such as H. influenzae and S. pneumoniae) or gram negative enterics (such as Klebsiella, Escherichia coli, Pseudomonas aeruginosa, Citrobacter, and Proteus) are identified [27, 28]. More unusual organisms such as fungal, mycobacterial, and even parasitic organisms have been reported [28].
General Therapeutic Considerations
Empirical therapy should include the combination of ampicillin or amoxicillin plus a betalactamase inhibitor or clindamycin. For severe or life-threatening infections, broad-spectrum intravenous therapy is recommended, including empirical coverage for methicillin-resistant S. aureus such as with vancomycin plus ampicillin/sulbactam or piperacillin/tazobactam.
Definitive antimicrobial therapy should be directed by culture and susceptibility results. Therefore needle aspiration early in the course of treatment is optimal, and material obtained should be sent for gram stain and aerobic culture, anaerobic culture, fungal culture, mycobacteria culture, and histopathology review.
Surgical excision is recommended as the definitive treatment for all types of branchial anomalies, although specific considerations are detailed below. In order to prevent infections and scarring, early excision might be best, although waiting until the child is 2 or 3 years old might be optimal to minimize surgical and anesthetic risks. All acute infections should be primarily treated with antibiotics and needle aspiration, avoiding incision and drainage as much as possible to prevent field scarring [23]. Further surgical approaches are described below for individual branchial cleft types.
First Branchial Arch Anomalies
Embryology
The first branchial arch is also called the mandibular arch and forms the mandible, a portion of the maxillary process of the upper jaw and portions of the inner ear. The cleft and pouch form the external auditory canal, the Eustachian tube, the middle ear cavity and the mastoid air cells [23]. First branchial anomalies have a close relationship to the facial nerve and they can course deep, between branches or superficial to it. They are usually deep to the superficial lobe of the parotid gland (Fig. 15.4).
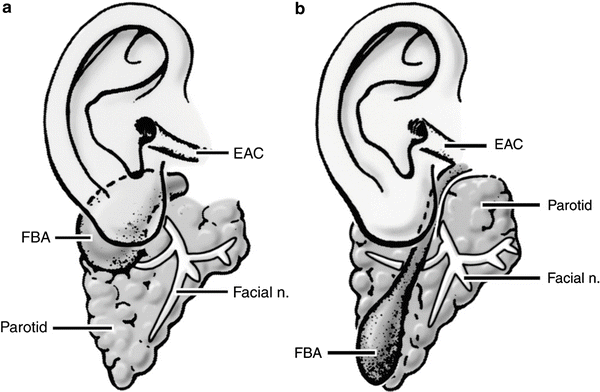

Fig. 15.4
Type 1 (a) and 2 (b) first branchial cleft anomalies (FBA) according to the Work classification. In type 1 anomalies, the opening of the cyst does not connect to the external auditory canal (EAC). In type 2 anomalies, the opening of the cyst connects to the EAC and is located within the deep lobe of the parotid gland (Reprinted from Neuroimaging Clin N Am, Vol 10, Mukherji SK, Fatterpekar G, Castillo M, et al. Imaging of congenital anomalies of the branchial apparatus, pp. 75–93, Copyright (2000), with permission from Elsevier.)
Clinical Presentation
A first branchial sinus or fistula can open at the level of the external auditory canal, the middle ear cleft, the postauricular region or the angle of the mandible [29]. First branchial cleft anomalies can be classified according to a few existing systems, the most commonly used being the Arnot and Work classification [30, 31] (Table 15.1).
Table 15.1
Classification of first branchial cleft malformations
Type | Origin | Description |
|---|---|---|
I | Cystic mass of purely ectodermal origin | Duplication of the membranous external auditory canal which courses lateral to the facial nerve |
II | Cyst, sinus and/or fistula of ectodermal and mesodermal origin | Lesion may be found in a preauricular, postauricular, infraauricular or angle of the mandible location and it courses medial to the facial nerve |
Many first arch anomalies are confused with preauricular sinuses and are therefore inadequately treated, with recurring infections preceding surgical excision. Symptoms include the presence of a chronically draining pit at the angle of the mandible, which turns into purulent drainage and submandibular adenitis if the tract becomes infected. Inflamed tracts within the parotid gland can present as a rapidly expanding mass, while tracts connecting to the external auditory canal or tympanic membrane can present with mucous or purulent otorrhea [23]. Complications from infections of a first branchial cleft cyst include damage to the parotid gland and/or facial nerve.
Diagnosis
First branchial cleft cysts are diagnosed clinically, but a contrasted CT or MRI can be very useful in determining the type of lesion and the depth of parotid extension, if it is suspected.
Therapeutic Approaches
Most first branchial cleft tracts lie superficial to the facial nerve, although some investigators have shown that sinuses more often lie superficial to the facial nerve while fistulae are more likely to pass deep to it. The tract is also more likely to be deep to the facial nerve in patients who are less than 6 months at the age of presentation. The safest approach to first branchial cleft cysts and tracts is therefore a conservative superficial parotidectomy with the use of a facial nerve monitor [29]. Involved skin and cartilage from the external auditory canal should also be excised. If the tract extends into the middle ear, this should be excised as well. Incomplete excisions are unfortunately common given the complex course of first branchial anomalies [23].
Second Branchial Arch Anomalies
Embryology
The second branchial arch is also called the hyoid arch and it forms portions of the hyoid bone. The pouch becomes the palatine tonsil and supratonsillar fossa [23]. As such, a fistulous second branchial arch anomaly can connect the tonsillar fossa to the anterior neck.
Clinical Presentation
A diagnosis of branchial cleft cyst is probable when a unilocular, nontranslucent cystic swelling is found in the deep anterior portion of the sternocleidomastoid muscle [12]. A fine needle aspirate containing mature squamous cell and cholesterol crystals is diagnostic [12]. Second cleft anomalies also usually present as a draining cyst of fistula at the lower anterior border of the sternocleidomastoid muscle, which can increase in size with upper respiratory tract infections. Contrary to many other congenital lesions of the head and neck, cysts are most commonly found in adults between the third and fifth decade of life. Fistulae are more easily diagnosed in infancy and childhood, as they are associated with chronic drainage. Infected tracts and cysts can lead to respiratory distress, torticollis and dysphagia [23].
Diagnosis
In order to differentiate second branchial cleft anomalies from masses such as macrocystic lymphatic malformations, lymphadenopathy, plunging ranula and others, an array of diagnostic imaging methods may be necessary, such as ultrasound, CT and MRI. The mass will usually appear as round and sharply demarcated with a thin to imperceptible wall and on ultrasound, a posterior acoustic enhancement is demonstrated in 70 % of cases. A “notch sign”, which is an extension of the cyst between the internal and external carotid artery, just above the carotid bifurcation, is pathognomonic on CT scan [32].
Therapeutic Approaches
Second branchial arch anomalies can be excised via a transverse cervical approach, with the addition of a step-ladder approach to visualize the proximal portion of the tract as it enters the superior pharynx.
Third and Fourth Branchial Arch Anomalies
Embryology
The third branchial pouch divides into the inferior parathyroid gland from its dorsal component and the thymus from its ventral component [33]. The fourth branchial pouch forms the superior parathyroid gland by combining the dorsal portion of its caudal pharyngeal complex to the rudimentary fifth pouch. It is thought that it contributes to thyroid development and to the thymus gland as well [34]. The pharynx is formed from a combination of the third and fourth branchial cleft pouches.
Some controversy exists regarding third and fourth branchial pouch anomalies [33]. Some authors define third and fourth branchial anomalies based on the type of tissue that is contained within a cyst, while others define them based on the location of the internal opening of the sinus [23]. For both third and fourth branchial anomalies, the internal opening is at the pyriform sinus; the former opens at the base and the latter, at the apex. They both are more common in the left side of the neck, possibly due to the differential development of the arch vasculature. The course of each anomaly is theoretically predictable, with third arch anomalies passing deep to the internal carotid artery and glossopharyngeal nerve, entering the thyroid membrane above the internal branch of the superior laryngeal nerve and entering the pharynx at the pyriform sinus. Fourth arch anomalies should pass deep to the internal carotid artery, loop around the vessel associated with each side (subclavian artery on the right and aortic arch on the left), ascend to the level of the hypoglossal nerve and descend along the anterior border of the sternocleidomastoid muscle to enter the pharynx at the level of the pyriform apex beneath the superior laryngeal nerve.
Clinical Presentation
Third and fourth branchial cleft cysts generally present in a similar fashion as second branchial cleft cysts, although recurrent neck infections are an even more common presentation, usually after upper respiratory tract infections [33]. In addition, neonates can have airway compromise from large third and fourth branchial cysts [34]. Third branchial anomalies can rarely cause hypoglossal nerve palsy if they become infected. Fourth branchial anomalies are intimately associated with the thyroid gland and infections can manifest with suppurative thyroiditis [23]. In fact, as the thyroid is largely resistant to infection (given its extensive blood supply and lymphatics, strong capsule that protects it from surrounding structures, and high iodine content), any patient with acute or recurrent infectious thyroiditis should be evaluated for an underlying lower brachial cleft abnormality [35, 36] (Fig. 15.5). Skin openings for fourth branchial anomalies are thought to only result from infection and fistulous formations or inadequate surgery.
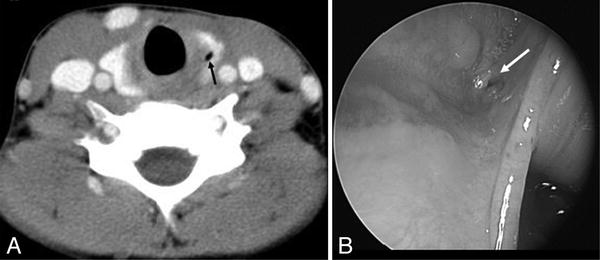

Fig. 15.5
(a) Contrast-enhanced axial CT scan at the level of the thyroid gland which is showing a fourth branchial sinus tract remnant within the left lobe of the thyroid (black arrow). (b) The opening of the sinus tract at the apex of the pyriform sinus can be seen on this pharyngoscopy image (white arrow) (From Thomas B, Shroff M, Forte V, et al. Revisiting Imaging Features and the Embryologic Basis of Third and Fourth Branchial Anomalies. AJNR Am J Neuroradiol 2010; 31(4):755–60. Republished with permission.)
Diagnosis
Aside from a thorough history, physical examination and neck imaging, upper airway endoscopy and contrast studies can sometimes help in the diagnosis of third and fourth branchial cleft anomalies and rule out thyroiditis or suppurative lymphadenitis. These studies also help in sorting out the differential diagnosis, which includes infected neck nodes, hemangiomas, thyroid cysts, ectopic thyroid gland and cervical thymic cysts [37]. Neck imaging can be done by ultrasonography or contrasted CT or MRI, although CT seems to be best at demonstrating fistulae [23]. Upper airway endoscopy is used to identify the opening into the pyriform sinus and many authors suggest that all suspected cases must undergo direct laryngoscopic examination. Barium esophagram, if performed at least 6 weeks away from an acute infection, can demonstrate a sinus tract in the vast majority of cases [37] (Fig. 15.6).
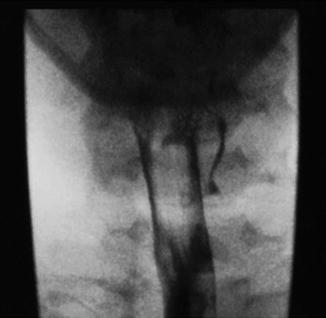

Fig. 15.6
Barium swallow demonstrating a left pyriform sinus tract (Reprinted from the International Journal of Pediatric Otolaryngology, Vol. 68, Pereira KD, Losh GD, Oliver D, et al., Management of Anomalies of the Third and Fourth Branchial Pouches, pp. 43–50, Copyright (2004), with permission from Elsevier.)
Therapeutic Approaches
Infected lesions should be managed by intravenous antibiotics with avoidance of incision and drainage as much as possible, as this can alter anatomic planes and complicate subsequent surgical excision [37]. Transverse cervical excision is usually the best and most definitive approach to treating non-infected third and fourth branchial arch anomalies. Chemo-cauterization of the internal opening has also been used, although there are no long-term results of the technique [23, 37]. An ipsilateral hemithyroidectomy must also be performed at the time of removal of fourth arch anomalies in order to remove the entire tract, which should be followed in its entirety, sometimes going through the thyroid cartilage itself via an oblique thyrotomy above the cricothyroid joint [34]. In all cases, closure of the pyriform sinus is key to preventing recurrence [34]. The rate of recurrence after surgical excision is around 15 %.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


