Chapter 74 Infantile esotropias
“First, we must, I believe, firmly embed strabismus in the matrix where it belongs – an anomaly of neurodevelopment.” John T Flynn1
Infantile esotropia (IET) is a constant non-accommodative esotropia (ET) with onset before 6 months of age in a neurologically normal child (Fig. 74.1). The angle is > 30 prism diopters with mild or no amblyopia and mild hyperopia. “Congenital esotropia” is often used synonymously but the condition is rarely present at birth. These children may have potential for good binocularity if they are treated early, suggesting the underlying defect is not congenital. It is often accompanied by dissociated vertical deviation (DVD) 50–90%,2,3 inferior oblique muscle overaction (IOOA) 70%,4 latent nystagmus 40%, and optokinetic asymmetry. IET and the secondary associations are often grouped under “infantile strabismus complex;” some components may exist in the absence of strabismus.
History
The term infantile esotropia was introduced by Costenbader to include age of onset less than 1 year.5 He had previously defined “congenital” ET as having an onset less than 6 months.6 The term congenital ET reflects Claud Worth’s theory7 in which ET is caused by a congenital defect of fusion – “The essential cause of squint is a defect of the fusion faculty.” Worth’s theory implies that the “congenital defect” is irreparable and that surgery can be carried out at leisure, largely for cosmetic reasons. The opposing view, that the infant visual system is normal initially until ET occurs as a result of extrinsic factors, was proposed by Chavasse.8 Chavasse thought that “most infants with congenital squint are capable of developing fusion if the deviation is fully corrected before the age of two years.” This view was accepted by Costenbader,9 Parks, and his fellows and became widely accepted after the work of Ing.10 It is not so widely accepted in Europe where surgery is often carried out much later.11 The evidence supporting Chavasse’s view that corrective surgery should be carried out early in infancy appeared in the 1960s and, during the 1980s, there was an explosion of knowledge of vision development in infancy12–14 and a refinement of surgical techniques. Surgical case series have enabled consolidation of clinical knowledge concerning IET.15–17 There has been progress in documenting the effects of early surgery on binocular outcomes and motor alignment.
Epidemiology
The prevalence of any ET in a population of 38 000 children aged 1–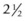 years was found to be 0.9%.18 The prevalence of IET in one area of the US was relatively constant at 0.25% of the population.19 The rate of strabismus surgery in childhood has declined substantially in the UK in recent decades.20 The decrease may be due to the routine prescribing of the full cycloplegic refraction for strabismus. Despite the decrease in strabismus surgery, Carney et al. stated that the rate of surgery in children under 1 year was unchanged.21
years was found to be 0.9%.18 The prevalence of IET in one area of the US was relatively constant at 0.25% of the population.19 The rate of strabismus surgery in childhood has declined substantially in the UK in recent decades.20 The decrease may be due to the routine prescribing of the full cycloplegic refraction for strabismus. Despite the decrease in strabismus surgery, Carney et al. stated that the rate of surgery in children under 1 year was unchanged.21
IET runs in families so there is a hereditary component and primary monofixation syndrome occurs much more commonly in the parents and families of children with IET than in the general population.22 Maumenee et al. concluded that the inheritance pattern was most likely Mendelian codominant.23
The natural history of the development of ocular alignment
In a study of normal neonates, Sondhi et al.24 found that 55% had a constant deviation (66% of whom an exodeviation) and 30% were orthotropic. No neonatal esodeviations were seen after 2 months of age and 97.2% of their infants were orthotropic at age 6 months. In another study25 the same authors concluded that the time of onset of IET was between 2 and 4 months of age. Horwood described “neonatal misalignments” (NMs) as fleeting, large angle, mainly convergent, ocular deviations26 occurring commonly in the first two months of life (73.2% of infants in the first month of life). The NMs declined to almost zero by 4 months of age except in those children who went on to develop IET where the frequency of misalignment increased.27 There was no relationship between frequent NMs in the first 2 months of life and the subsequent development of IET. Horwood’s report of a high prevalence of convergent misalignments in (older) normal infants is in direct contrast to those of Archer et al.’s25 high prevalence of divergent deviations (especially in the immediate newborn period).The Sondhi and Archer24,25 studies used a method which relied on the infant fixating on the examiner’s face. This may have had an artifact induced by the large positive angle kappa seen in early infancy and the particular fixation pattern of neonates when looking at a face.28 Friedrich and de Decker29 also found a high (50%) incidence of exotropia (XT) in neonates, but only one of 1024 newborn infants developed an ET that was still present at 3 months of age. Children who develop IET begin to be different from normal infants at approximately 4 months of age. Because transient misalignment of the eyes is common parental reports the exact age of onset of strabismus must be viewed with caution.
Birch et al. reported that none of 66 infants with misalignment ≥ 40 pd of ET and an onset between 2 and 4 months of age had spontaneous regression suggesting that they were suitable candidates for early surgery.30 The Congenital Esotropia Observation Study (CEOS)31 reported that, before 12 weeks of age, the majority of esotropic infants had an intermittent or variable deviation, whereas after 12 weeks of age the deviations were more constant. They concluded that normal infants > 10 weeks old with a constant ET of ≥ 40 PD and ≤ 3.00 diopters of hyperopia had very little prospect of spontaneous regression (2%) and could be offered early surgery (Box 74.1).32,33 However, if the deviation was < 40 PD, intermittent or variable, it usually resolved.
Box 74.1
Clinical profile of infants who will benefit most from early surgery, i.e. unlikely to spontaneously resolve
1. Presence or persistence of ET between 10 weeks and 6 months of age
2. Constant ET 40 pd at near (1/3 m) on two examinations 2–4 weeks apart
4. Absence of any of the following conditions:
Adapted from Spontaneous resolution of early-onset esotropia: experience of the Congenital Esotropia Observational Study. Am J Ophthalmol 2002; 133: 109–18 by Wong AMF. Can J Ophthalmol 2008; 43: 643–51.
Clinical patterns
Dissociated vertical deviation (alternating sursumduction, occlusion hyperphoria)
In DVD, the eye elevates and extorts and is not associated with a corresponding downwards movement of the other eye when fixation is resumed (Fig. 74.2). DVD does not follow Hering’s Law. It can occur spontaneously or only when the fellow eye is occluded. DVD can vary in size and frequency of occurrence. Rarely, the patient adopts a head tilt to the same side. For management purposes, DVD can be quantified with base down prism. The prism is placed in front of the eye with DVD until no more downward movement takes place on alternate cover testing.
Inferior oblique muscle overaction
Inferior oblique muscle overaction (IOOA) is visible in adduction only and associated with fundus excyclotorsion (Fig. 74.3) and a V pattern. DVD and IOOA are usually bilateral, but can be asymmetric. If the eye elevates in adduction and there is a corresponding hypodeviation in the opposite eye, the deviation is due to IOOA. If there is no corresponding hypodeviation, then DVD is present. If the elevation of the adducted eye exceeds the hypodeviation in the fellow eye, combined DVD and IOOA are present.
Dissociated horizontal deviation
Dissociated horizontal deviation (DHD) is much less frequent than DVD. Often asymmetric, it can occur simultaneously with DVD. DHD may merely be DVD with a large horizontal component. It can cause confusion with consecutive XT and the two may coexist. The two can be distinguished by a difference in horizontal measurements when measuring with the prism in front of each eye. The treatment is a supramaximal lateral rectus recession of the affected eye.34
Latent nystagmus
Latent nystagmus (LN) is common in IET and occurs on monocular occlusion. The fast phase is toward the uncovered eye. The nystagmus is lessened in adduction and worsened in abduction. LN can lower the acuity in the uncovered eye. This can be obviated by fogging the eye not being tested with a +3.00 lens rather than occluding it. Manifest latent nystagmus is “latent” nystagmus which is present in binocular viewing (see Chapter 88).
Differential diagnosis
The differential diagnosis and clinical forms of IET can be considered as either truly congenital or early in onset (within the first 6 months; Box 74.2). In most cases a thorough history and ophthalmic examination helps to differentiate the subtypes (Fig. 74.4).
Box 74.2
Differential diagnosis of infantile esotropia
| True congenital onset ET | Infantile onset ET |
CP, cerebral palsy; PVL, periventricular leukomalacia; DS, Down’s syndrome.
Pseudoesotropia is common. It can be confirmed by performing the Brückner, Hirschberg, and cover tests to determine that the eyes are orthotropic (Fig. 74.5). The Brückner test35 can have false positives in children younger 8 months of age.36 Examination of photographs with the parents can confirm the diagnosis.
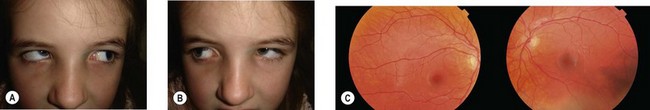
In type 1 Duane’s syndrome, the diagnosis is confirmed by limitation of abduction and globe retraction in adduction. Bilateral type 1 Duane’s syndrome can be difficult to distinguish from IET, particularly if the child is cross-fixating as the globe retraction in adduction can be quite subtle in infants. Performing a doll’s eye maneuver to demonstrate full abduction will exclude Duane’s syndrome and congenital sixth nerve palsy (a benign condition which usually resolves by 6 weeks of age).37 Other techniques to elicit abduction include rotating the child or using monocular occlusion. A sixth and seventh nerve palsy may be due to Möbius syndrome, which may be associated with orofacial and limb abnormalities (Fig. 74.6).
A full ophthalmic examination including cycloplegic refraction will exclude any anterior or posterior segment cause for a sensory ET (see Chapter 72).
Ciancia syndrome
Ciancia described a subset of esotropes38 that others refer to as “cross-fixation congenital esotropia.”17,39
Ciancia syndrome has the following features:
Nystagmus blockage syndrome
Nystagmus blockage syndrome (NBS) was first described by Adelstein and Cuppers.42 von Noorden reported a prevalence of between 4.8% and 10.3% of congenital esotropes.43 The syndrome is characterized by:
1. Nystagmus with early onset large, variable angle ET.
3. Head turn in direction of fixing eye.
4. Absent nystagmus with the fixing eye in adduction.
5. Increasing magnitude of manifest nystagmus with abduction.
NBS and Ciancia syndrome should be considered in any esotropic child with abducting nystagmus. However, the underlying pathophysiology is different. In Ciancia syndrome, the ET and nystagmus are coincidental,15 whereas in NBS there is congenital idiopathic nystagmus with the null point in adduction (causing ET). Accommodation/convergence innervation may play a role and pupillary constriction in adduction may assist in making the diagnosis of NBS.44 With alternating ET and cross-fixation, there is no nystagmus in primary gaze or abduction. The fixating eye is in the primary position and the non-fixating eye adducted (unlike in NBS where both eyes are in adduction). There is often no amblyopia (unlike in NBS).43 Treatment in all these syndromes is often the same (large bilateral medial rectus recessions (BMRc)) with good motor outcomes45 in most cases.
The prevalence of strabismus in children with developmental delay is reported to be between 27% and 100%.46,47 Haugen et al found that strabismus in Down’s syndrome occurred in 42% and was predominantly early-onset ET (84%) associated with hyperopia.48 The rate of strabismus in patients with cerebral palsy is 44% on average (15–62%).49
Surgery for ET in developmentally delayed children has variable postoperative outcomes. Some groups report high rates of overcorrections and unpredictable outcomes.50 Muen et al. reported suboptimal surgical results in seven patients who were unexpectedly diagnosed with periventricular leukomalacia after presenting with strabismus.51 While some authors decrease the surgical dosage, Yahalom suggested that reduction of standard surgical dosages for ET in Down’s syndrome is unnecessary.50 We follow this practice.
Does stability of preoperative alignment affect outcomes?
Ing showed that the majority of patients observed prior to surgery for IET showed an increase of at least 10 pd with 50% showing an increase of at least 20 pd.52 When surgery was based on the measurements taken 1 day before surgery, there was no difference in alignment between the group with an increase in angle and those with stable measurements. In a study of infants with IET grouped into stable (<10 pd difference in measurements between visits) or unstable (≥10 pd difference in measurements), Birch et al. demonstrated that there was no difference in postoperative alignment, reoperation rates, rates of prescription of hyperopic or bifocal spectacle or stereoacuity.53 In a further study, the motor outcomes (alignment) were the same regardless of the stability of measurements before surgery.54 There is an improvement in measurement stability after 6 months of age.55
Measurement uncertainty
Schutte et al. identified sources of human error in strabismus surgery.56 Half of reoperations were due to inaccuracy in measurement of the angle of strabismus, variability in surgical strategy, or imprecise surgery.
In the Early vs. Late Infantile Strabismus Surgery study (ELISS) pilot study57 three masked examiners found high intraobserver agreement with the Hirschberg test, but in 10% of cases it exceeded 10o (20 pd). In another study, experienced strabismologists reviewed photographs of strabismic patients and estimated the angle of squint using the Krimsky and Hirschberg tests with an expert examiner prism cover test (PCT) as the “gold standard.”58 The majority of examiners overestimated at least one patient by 10 pd with the Krimsky test. Differences of 5 pd were difficult to distinguish. With the Hirschberg test, each patient was underestimated by 10 pd by at least one examiner, with more inaccuracy for larger angles. The authors concluded that the Krimsky test is more accurate than Hirschberg, but both are less accurate than a cover test. Thompson and Guyton reported that it is impossible to accurately measure extremely large angles of strabismus with prisms.59 A Pediatric Eye Disease Investigators Group (PEDIG) study60 looked at interobserver reliability of the prism and alternate cover tests. They concluded that, in childhood ET of more than 20 pd, differences of 12 pd or more are likely to indicate real change, but smaller differences could be due to measurement error. We should be sceptical about the degree of accuracy of our measurements.
The PCT is the “gold standard” for strabismus measurement but can be limited by the child’s cooperation. The Krimsky test is easy to perform with the prism split between the two eyes for angles of squint greater ≥40 pd. When the child is uncooperative, we use a combination of the Krimsky and Brückner tests.61 We use loose plastic prisms for the PCT. Plastic right-angled prisms should be held in the frontal plane position59 and plastic isosceles prisms should be held close to the position of minimum deviation.62 This means that for both types of prism, each prism is always held with the base in the sagittal plane. We acknowledge that the prismatic effect of two split prisms is not the arithmetic sum of the two individual prisms,59 but for most angles of ET the difference is within the measurement error. The measurement of the distance deviation is problematic in small infants, but it is always possible to measure the near angle. We usually base our surgical plan on the near deviation.
Non-surgical management
Refraction is crucial to the management of IET, especially in the postoperative period. Hyperopia occurs in 15% of children with IET.63,64 At presentation, we treat infants with ≥2.5 D hyperopia with full spectacle prescription63,65 to determine whether the ET is partially or fully accommodative. This resolves the ET completely in almost 50% of cases, therefore preventing unnecessary surgery.63,64 However, 20% of infants initially controlled with glasses will decompensate and require surgery.66 Spectacle correction can reduce a large angle strabismus such that only two muscles might need to be operated on rather than three. Correction of hyperopia is essential to maintain postoperative stability.
Amblyopia can be inferred by the child’s fixation pattern. Fixation should be maintained through a blink in both eyes if the vision is equal.67,68 Cross-fixation occurs when an infant fixes across their nose at the object of interest (Fig. 74.7). As an object of interest is moved from one side to the other, fixation switches at the midline if the vision is equal. If fixation switches past the midline, amblyopia is present in the eye towards which the toy is being moved.69
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree