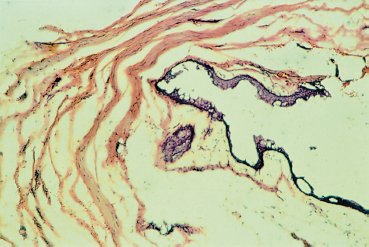
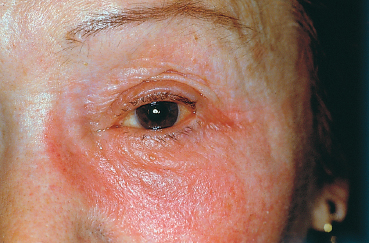
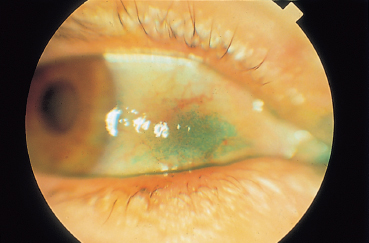
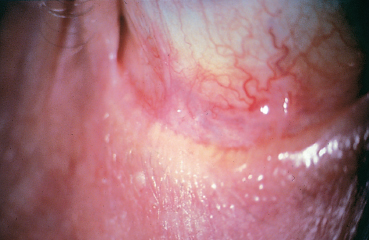
Chapter 8 Iatrogenic Disorders of the Ocular Surface Fig. 8.1 Contact allergy 48 hours after eyedrop instillation Fig. 8.2 Lissamine green staining localized to the nasal bulbar conjunctiva (right eye), in the region of maximal eyedrop contact Fig. 8.3 Lissamine green staining of the inferior cornea, at level of the tear meniscus, suggestive of direct eyedrop toxicity Fig. 8.4 Subconjunctival fibrosis secondary to chronic use of antiglaucoma eyedrops However, on closer scrutiny, or using more refined tools, the ocular surface, and patient comfort too, are often far more severely affected by topical treatments. Besides the simple problem of discomfort, severe abnormalities can develop insidiously, only becoming symptomatic at a very late stage, and sometimes in a totally unexpected manner. For example, how to establish the link between progressive fibrosis of the ocular surface and a commonplace, well-tolerated, topical treatment started fifteen years previously? Another illustration is the role of glaucoma treatments in the increasing failure rate of glaucoma surgery due to fibrosis of the filtration site. These examples uncover new pharmacological problems, especially regarding the identification of the responsible factors (not always the drugs themselves) and the development of alternatives with better long-term safety. This chapter will only deal with iatrogenic problems affecting the ocular surface associated with topical products, leaving to one side complications linked to contact lenses, corneal thesaurismosis due to systemic treatments, and corneal complications of intraocular drug injections. As the cornea and conjunctiva are contiguous, this analysis cannot reasonably be restricted to the conjunctiva. A brief reminder of the defense mechanisms of the ocular surface will be provided to clarify the multiple mechanisms involved in drug toxicity for the cornea and conjunctiva. Clinical aspects, investigatory techniques, and pharmacological specificities will also be considered. Allergic Mechanisms Allergic phenomena used to be divided into two main mechanisms: —Type I hypersensitivity, corresponding to true allergic reactions, induced by IgE and triggered by mast cell degranulation; —Type IV hypersensitivity (contact allergy), governed by specific cellular immunological reactions. In ophthalmology, type I hypersensitivity is classically associated with seasonal and perennial conjunctivitis, and vernal and atopic keratoconjunctivitis. In contrast, type IV hypersensitivity corresponds to contact allergies triggered by drugs and other chemicals. While the latter can affect all patients, the former is usually favored by a genetically determined background, in which high mast cell densities in tissues and abnormally abundant IgE synthesis predisposes the subject to acute allergic reactions, in a phenomenon known as atopy. However, improved knowledge of immunological mechanisms1,2 has recently led to a reexamination of this classification, by revealing that IgE-dependent immune reactions do not exclusively occur in atopic subjects. Indeed, cell types other than mast cells possess surface receptors for IgE. This is especially the case of eosinophils, macrophages, dendritic cells, platelets, and some T lymphocytes, which can therefore play a direct role during IgE-dependent inflammatory reactions. In addition, we now know that the lymphocyte system comprises two types of helper T cell: Th1 lymphocytes and Th2 lymphocytes.1 The first govern delayed-type hypersensitivity reactions involving IgG and IgA secretion, through the action of certain cytokines (especially IFN-α, IL-2, and TNF-α). In contrast, Th2 lymphocytes regulate IgE-dependent reactions by stimulating IgE production, with the involvement of cytokines such as IL-3, -4, and -5. So-called naive lymphocytes, i.e., those that have never been in contact with an antigen, have neither Th1 norTh2 functions. After an initial contact, according to the characteristics of the antigen (size, chemical nature, concentration, mode of administration, single or repeated contact), T lymphocytes enter one of the two Th systems. This means that an IgE-dependent Th2 reaction can occur in nonatopic subjects who have no particular genetic predisposition. It also explains why manifestations identical to those of immediate hypersensitivity reactions can occur after relatively long periods, resembling delayed hypersensitivity reactions. These notions throw a different light on the mechanisms of allergy, as the background, clinical manifestations, and chronology of the inflammatory event are no longer reliable signs of a given pathogenic mechanism. In addition, allergy, and even immune activation, are not the only mechanisms of drug intolerance. In many cases an irritant, purely toxic action is involved, leading to secondary inflammatory reactions that are associated more with cicatrization than with immunization. Immunological Mechanisms Drug-induced intolerance reactions with an immunological component occur, on average, in 3-10% of prescriptions; the immunological mechanism is often very difficult to identify, because it can be either primary or secondary, and is often indistinguishable from a nonspecific inflammatory response. Despite the common use of the word allergy, these reactions rarely involve a type I immediate hypersensitivity mechanism, but rather a delayed type IV hypersensitivity reaction, even if combinations of the different mechanisms can exist.3 Most drugs have low molecular masses (500-1000 Da) and are not directly immunogenic. To become immunogenic, they must form stable conjugates with endogenous proteins, in a process known as haptenization. Sometimes the sensitizing hapten is a metabolite or degradation product of the drug, or an impurity. A history of sensitization in a given subject provides few indications on the mechanism involved. The sensitization may go unnoticed if it involves a cross-reaction, a solvent shared by another drug, or even food. Previous well-tolerated administration of a given drug, even repeatedly, does not protect the subject against a later immunological reaction. In addition, the immune terrain seems to have relatively little importance. The atopic patient is classically more at risk, but this notion is only relative: these subjects may react more violently but not more frequently than “immunologically normal” individuals. Immediate allergic reactions resulting from mast cell de-granulation are relatively rare. They are associated with intense pruritus, urticaria, and chemosis, occurring seconds or minutes after eyedrop instillation. They have mainly been described with topical anesthetics and mydriatics (especially phenylephrine). Immediate hypersensitivity reactions also have a delayed phase, occurring 24-48 hours later, and may become chronic if the responsible eyedrops continue to be used. Nevertheless, poorly known mechanisms of Th2 system activation, with IgE secretion, can also occur late, closely resembling cell-mediated immunological reactions. Delayed-type hypersensitivity reactions, after a process of sensitization that lasts from 10-14 days, occur about 8 hours after the triggering event, with a maximum at 48-72 hours. This stage is associated with marked epithelial and sub-epithelial edema, as well as infiltration by Langerhans cells, CD4 lymphocytes, and sometimes mast cells and basophils. This type of reaction, which is encountered with strongly sensitizing agents, sometimes involves a long period of sensitization (months or even years) to less powerful allergens. This long interval accounts for the frequent diagnostic difficulties. Irritants and Toxic Effects Some drugs are only irritants3,4 through a purely cytotoxic action, their pH, the osmolarity of the solution, or even photo-sensitization. Some substances can be allergenic at low concentrations and irritant at high doses. The inherent toxicity of the product can cause necrosis of corneoconjunctival cells or induce a cascade of active mechanisms leading to their “suicide” by apoptosis. In both cases the drug will induce secondary inflammatory reactions, with a cicatricial rather than an immunological response. The inflammatory mediators are very similar, whatever the trigger, and the inflammatory infiltrates are not very different. Nevertheless, the often chronic nature of these irritant effects can lead, in the long term, to permanent infiltration by inflammatory cells and fibroblasts. Theoretically, contrary to immunological reactions that require prior sensitization, toxic effects can occur after the first contact. Often, however, they only appear after a cumulative threshold dose, which may only be reached after several weeks, months, or even years. The toxicity of some drugs can be exerted indirectly.3,5 This is the case of antibiotics, antivirals, and corticosteroids, which modify the conjunctivopalpebral microbial flora. These agents favor bacterial or fungal infections by selecting resistant strains and, in the case of corticosteroids and some antivirals, by diminishing local immune defenses. Chronic conjunctivitis or blepharitis can result. Some effects involve tear film modifications, indirectly disturbing the ocular surface. Direct toxicity for conjunctival goblet cells (cytotoxic substances, preservatives, antihistamines, etc.), a reduction in tear production (parasympatholytic and vasoconstrictive agents), or a detergent effect on the lipid phase of the tear film (quaternary ammonia), which is crucial to prevent evaporation, are frequently encountered with a number of drugs, and especially their preservatives. In contrast, corticosteroids and pilocarpine can stimulate meibomian secretion and induce seborrheic blepharitis. Tear outflow obstruction by inflammatory and/or immuno-logical mechanisms (antivirals, adrenomelanic deposits) can also be important disruptive mechanisms for the ocular surface, inducing modifications of tear drainage and the microbial flora. Reflex tearing becomes very difficult to interpret in these circumstances. Delayed wound healing (corticosteroids) and ocular surface deposits (less frequent with the gradual removal of mercury and epinephrine from ophthalmic solutions) are noteworthy effects of iatrogenic toxicity. Adrenomelanic deposits (due to epinephrine oxidation) can appear after a year of treatment, involving the entire ocular surface and possibly obstructing the lacrimal excretory system. Without being dangerous in themselves, they reflect profound modifications of the ocular surface. Finally, it should be remembered that combinations of various mechanisms can occur simultaneously, adding to the complexity of the situation, and hindering the diagnosis and treatment. There have even been reports of fibrosing conjunctivitis secondary to the formation of corrosive complexes of topically delivered mercury salts and systemically administered iodine or bromide!3 Clinical Aspects (Table 8.1) The Eyelids Examination of the eyelids cannot be dissociated from that of the conjunctiva, owing to the contiguity of the two structures, their protective role for the ocular surface, and their contribution to the quality of the tear film by their meibomian secretions. The skin of the eyelids can also disclose abnormalities that permit the diagnosis of clinically nonspecific disorders of the ocular surface. Associated with acute pruritus after eyedrop instillation, urticaria and edema can be typical manifestations of type I hypersensitivity. More frequent is type IV hypersensitivity, the most typical cutaneous manifestation of which is eczema (Fig. 8.1). The eczema can be highly characteristic, especially when it occurs a few days following treatment onset and affects the entire periocular region. It is sometimes far more discreet and difficult to identify when it is delayed or forms only small plaques masked by wrinkles. Eczema must be sought in patients with ocular surface disorders, as it has major diagnostic value. Eczematous lesions near the lid margin can be confused with seborrheic blepharitis. When crusts and scales are some distance from the lashes and meibomian orifices they can point to eczematization and contact allergy, due to a drug or hand-borne substance. The diagnosis can, however, be difficult when eyedrops cause toxic dermatitis or aggravate blepharitis by modifying the local microbial flora. Table 8.1 Local adverse effect of eyedrops
ALLERGIES |
acute, subacute, or chronic |
immediate or delayed |
PSEUDOPEMPHIGOID |
SUBCONJUNCTIVAL FIBROSIS |
FAILURE OF FILTRATION SURGERY |
CHRONIC IRRITATION |
TEAR FILM INSTABILITY |
QUANTITATIVE OR QUALITATIVE DRYNESS |
CONJUNCTIVAL INFLAMMATORY INFILTRATES |
MODIFICATIONS OF THE MICROBIAL FLORA |
PUNCTAL OCCLUSION |
THESAURISMOSIS AND CORNEOCONJUNCTIVAL DEPOSITS |
The Conjunctiva
Besides pruritus and tearing, the conjunctival sign most typical of allergy is chemosis. It is particularly clear and pure in rare type I allergies, but more discreet in delayed hypersensitivity, being masked by inflammatory signs and cutaneous involvement. In delayed hypersensitivity reactions, which are often more chronic (especially when the condition goes undiag-nosed and treatment is continued), the conjunctiva is often the site of vesicles and epithelial punctate erosions, which can be seen with fluorescein or, better still, with rose bengal or lis-samine green.
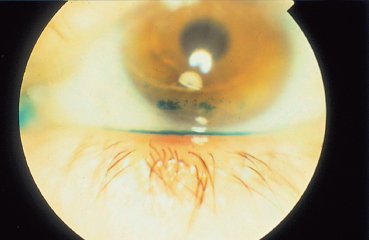
Punctate conjunctival epitheliopathy is often highly evocative of an iatrogenic cause when it predominates nasally (Fig. 8.2) or along the tear meniscus, where the contact time with the drug, between its instillation and lacrimal drainage, is maximal. These locations remain predominant even in diffuse toxic keratoconjunctivitis. In such cases they can also affect areas masked by the eyelids; this distinguishes them from signs of keratoconjunctivitis sicca, in which the lesions are most severe in the palpebral fissure area. However, these signs are not alone sufficient to distinguish immunoallergic conjunctivitis from purely toxic forms due to chronic irritation. Pruritus, contact eczema, and the chronology of events can be valuable diagnostic criteria, but none is really specific as several mechanisms can simultaneously be involved.
Follicular conjunctivitis, in which pruritus is mild or absent, is also frequent, and may or may not be accompanied by superficial keratitis. Immunoallergic mechanisms predominate in such cases.
Drug-induced corneal lesions usually consist of superficial punctate keratopathy; a preferential location on the inferior and/or nasal part of the cornea is often evocative (Fig. 8.3). Im-munoallergic peripheral ulcerative keratitis is rarer. It can respond favorably to corticosteroids, contrary to toxic ulcerations, which can be very extensive or take on deceptive pseudodendritic forms. Keratinization can occur when treatment is continued. Healing of epithelial defects can be delayed or hindered by chronic use of certain eyedrops which are presumed to favor wound healing but in fact are toxic due to their preservatives.
Toxic band keratopathy has also been described, especially during chronic treatment with eyedrops containing mercurial preservatives6. It appears to be due to a purely toxic action of mercury which, by denaturing corneal proteins, may lead to calcium deposits. These deposits are usually close to the limbus, and rarely involve the pupil area.
Presumed Drug-Induced Ocular Cicatricial Pemphigoid
Severe damage to the ocular surface can occur after long-term treatment, especially with antiglaucoma eyedrops (in 25-30% of cases) and topical antivirals. Complications include lacrimal duct and punctal occlusion, subconjunctival fibrosis (Fig. 8.4), symblepharon formation, corneal neovascularization, and ocular surface keratinization (identical to ocular cicatricial pemphigoid). Onset can be very late (mean 15 years after the start of treatment), creating diagnostic difficulties7. Immunopathological examination of conjunctival biopsy specimens only shows a nonspecific inflammatory reaction, and not the characteristic immune deposits in the basement membrane zone (BMZ).
Laboratory Investigations
(see Chapter 2, Focus  4 and 5 and Chapter 3)
4 and 5 and Chapter 3)
Conjunctival Biopsy
Conjunctival biopsy is the method of choice for the diagnosis of some inflammatory, autoimmune and infectious conditions. It also offers key material for research purposes, permitting major breakthroughs in ocular surface biology, especially through the use of immunohistochemistry or molecular biology techniques, and conventional, confocal, or electron microscopy. However, a patient who is suffering may not readily accept a biopsy, and the procedure can be a supplementary source of stress; in addition, it can be a waste of time if a specialized laboratory is not available to process the samples.
Conjunctival biopsy is a particularly useful diagnostic tool for autoimmune conjunctivitis (see Chapter 4), when it shows antibody and complement deposition in the conjunctival BMZ. It can be used, for example, to identify bullous autoimmune conditions such as cicatricial pemphigoid, epidermolysis bullosa acquisita, and bullous pemphigoid, and to distinguish these conditions from iatrogenic cicatrizing conjunctivitis. Perioperative conjunctival biopsies in glaucoma patients treated for long periods have also provided useful information on inflammatory status.8,9
Conjunctival Scraping and Smears
Being simpler and more efficient for the diagnosis of allergic disorders, cytological methods can be used to examine desquamated conjunctival cells and to identify inflammatory cells, some of which can assist the diagnosis. The presence of eosinophils and basophils points to an allergic reaction but is nonspecific, as these cells can also be found in purely toxic syndromes, albeit in small numbers3. Keratinization and epithelial destruction, accompanied by infiltrating lymphocytes and polymorphonuclear neutrophils, are more evocative of toxic causes, but a plethora of combinations can occur.
Impression Cytology
Impression cytology is a simple and noninvasive approach to epithelial cytology. Contrary to conjunctival scraping, this harmless and practically painless test, performed under topical anesthesia and with acetylcellulose filters (Millipore or Gelman; diameter: 13 mm; pore size 0.2 µm) provides a homogeneous cell layer for histological studies, with an intact architecture and preserved cell junctions. However, impression cytology cannot totally replace conjunctival biopsy, which can be used to study the conjunctival architecture (epithelium and chorion) throughout its thickness. Impression cytology can be used to analyze the most superficial layer of the conjunctival epithelium, in which the three principal cell populations are readily identifiable, namely epithelial cells, goblet cells, and Langerhans cells. Immunocytology techniques are used to detect and quantify certain activation markers present on the cell membrane or in the cytoplasm. This type of test is particularly useful in clinically noninflammatory conditions, such as dry eye syndrome refractory to substitutive therapy10 and chronically treated glaucomatous eyes11. In these settings impression cytology is used to detect the presence of a subclinical inflammatory reaction, which is otherwise undetectable but which must be treated to relieve the patient. Langerhans cell counts (increased in conjunctival inflammation), and especially quantification of the percentage of epithelial cells expressing inflammatory markers, are very precise and useful indicators of local immune status. Impression cytology also can be used to diagnose and analyze degenerative and/or toxic conditions of the ocular surface, with markers of apoptosis, for example.
IgE Assay in Tears
IgE assay in tears collected with a glass micropipette or a cellulose strip provides important diagnostic information and theoretically orients the diagnosis toward an allergic disorder involving an immediate hypersensitivity mechanism.12,13 A high level of IgE is very strongly associated with allergic manifestations (seasonal and perennial conjunctivitis, and vernal and atopic keratoconjunctivitis). In chronic conjunctival disorders, IgE assay also often yields interesting results and can rectify an erroneous clinical diagnosis.
One remarkable characteristic of Th1 and Th2 lymphocytes is that activation of one inhibits the other. Thus, when IgE (reflecting stimulation of the Th2 system) is detected in tears, one can deduce that the Th1 system is inhibited. This translates indirectly into abnormally high and easily demonstrable expression of class II antigens on conjunctival epithelial cells sampled by means of impression cytology. Comparative results of impression cytology and lacrimal IgE assay—two relatively simple routine laboratory tests—can differentiate between the two activation pathways and identify true, IgE-dependent allergies (in which allergological investigations and specific treatments are warranted) from nonspecific inflammatory reactions.14
Skin Tests and Conjunctival Allergen Challenge
Allergological skin tests can be of valuable assistance when a drug allergy is suspected. There are several such tests, which can be chosen according to the clinical setting and the type of hypersensitivity suspected. The cutaneous prick test explores immediate allergy: after scarification or dermal microinjection, the culprit allergen triggers abrupt mast cell degranulation and causes an immediate urticarial reaction. This test can be followed by serum assay of specific IgE (radioallergosorbent test [RAST]).
Similar tests can be done after mild skin abrasion: the eczematous reaction, observed after 48 hours, reflects the late phase of an immediate hypersensitivity reaction or contact allergy due to type IV hypersensitivity. Although highly useful, these tests are only meaningful when the results are positive, as the absence of reaction to a given antigen does not rule out the diagnosis of allergy. These tests are underused in the investigation of drug allergies. The results must be interpreted according to the patient’s answers to very precise questions on all drug intakes, their chronology, and all other possible factors of allergy. Finally, these tests pose some problems of interpretation, especially with substances that are more irritant than allergenic, as they are tested in amounts higher than those instilled into the eyes. For example, benzalkonium chloride causes a strong irritant skin reaction in 25-30% of cases, while true allergy is far rarer. In such cases it is extremely difficult to extrapolate the result to the ocular disorder.
Conjunctival allergen challenge consists of instilling a drop of the incriminated ophthalmic solution and noting any immediate or delayed reactions. This is sometimes done unintentionally on reintroduction of eyedrops responsible for an allergic cross-reaction or containing the same chemical (e.g., a preservative). Clinical observations must be carefully interpreted to identify the allergenic drug, and to avoid depriving the patient of effective eyedrops to which he/she is not in fact allergic.
Iatrogenic Drugs
Topical Anesthetics
Considering the large number of patients who receive an eye-drop of topical anesthetic at least once in their life, the incidence of toxic reactions is extremely low. This is mainly due to the small quantity of eyedrops used. The same does not apply to repeated use, either by error (e.g., self-treatment of painful ocular problems) or because of the increasing use of topical anesthetics for surgery of the anterior segment. In the former situation, and occasionally in the latter, toxic reactions can occur, especially on the corneal epithelium. Superficial punctate ker-atopathy is very frequent, even after a single instillation. Toxic lesions are at least partly due to the reduction in blinking, tear film instability (linked to a loss of epithelial microvilli), and corneal dessication.15 Deeper keratopathies can, however, occur, especially in patients with preexisting disorders or after excessive use.
Prescription of a topical anesthetic for a painful disorder inevitably leads to harmful self-treatment, lasting several days or even weeks, and is a serious error that can lead to very severe, sometimes incurable corneal complications.16 Contact anesthetics, when used repeatedly, exert their toxic actions, lead to corneal hypoesthesia, and delay the healing of primary or secondary epithelial lesions. True neuroparalytic keratitis can occur rapidly, with deep corneal involvement, potentially leading to permanent opacification and sometimes neovascularization. As anesthetic efficacy dwindles with repeated use, pain recurs increasingly rapidly and increasingly violently, establishing a vicious circle with aggravation of the corneal lesions and increasingly frequent self-treatment.
Diagnosis is simple when the anesthetic abuse is identified, but can be far more delicate if the patient hides the misuse; it can also be complicated by gradual onset of an inflammatory reaction. An early, fairly characteristic sign is a stromal ring surrounding the initial lesion. The mechanism underlying the stromal ring is not clear: an immunoallergic mechanism has been proposed (the opacity resembles a Wessely’s ring) but remains to be confirmed.
The severity of these lesions, that sometimes necessitate keratoplasty, justifies the absolute rule that anesthetic eyedrops must never be prescribed (or given), even when the patient, who tasted the subjective benefit during the ophthalmological examination, ardently requests a supply.
Mydriatic and Cycloplegic Agents
In addition to its frequent sensitizing actions, phenylephrine is toxic for the cornea, particularly if the epithelium is denuded, creating a risk of endothelial injury. Phenylephrine also has a powerful vasoconstrictive action and triggers the release of melanin pigments in the anterior chamber. These vasoconstrictive properties can cause marked conjunctival hypoxia. A 46% reduction in the PO2 has been reported after a single infusion of 2.5 % phenylephrine.17 This action dwindles during prolonged use, giving way to secondary vasodilation that tends to become more or less permanent. Other mydriatic or cycloplegic eyedrops, besides a danger of pupillary block, carry a risk of irritation and local allergy.
All types of allergic reaction can be triggered by topical antibiotics. In some cases the preservative is directly responsible, but most topical antibiotics are themselves toxic (especially fortified antibiotics) for the conjunctiva as well as the cornea (superficial punctate keratopathy), and the retina after intraocular injection.18 The aminoglycosides are the antibiotics most often responsible for toxic phenomena. Cases of conjunctival necrosis have been described with fortified gentamycin.19 Whitish deposits have been observed after repeated instillation of ciprofloxacin on an ulcerated cornea, but the local tolerability of fluoroquinolones is generally very good.20
Propamidine isethionate (Brolene), which is marketed in the United Kingdom as a simple antiseptic, is in fact a very specific treatment of amebic keratitis. During prolonged use it can induce toxic keratitis, sometimes accompanied by intraepithelial inclusions, and this can make it very difficult to distinguish from an initial amebic infection. If in doubt, treatment should be discontinued, but amebic relapses have been described several months after withdrawal of inadequately effective treatment.
Antivirals
In addition to classical hypersensitivity reactions, antivirals can have toxic effects, particularly when they are not very specific for virus-infected cells. This is especially the case of first-generation antivirals such as idoxuridine (IDU), vidara-bine, and, to a lesser extent, trifluorothymidine, which can cause marked stromal keratitis, delayed corneal epithelial healing, or punctal occlusion. Toxic corneal lesions can be difficult to distinguish from the underlying condition, and the persistence of dendritic keratitis or the onset of stromal opacities can be difficult to interpret. Conjunctival fibrosis has also been described with antiviral drugs.
Steroidal and Nonsteroidal Antiinflammatory Drugs
Corticosteroids mainly have intraocular adverse effects, which are often severe. The main risk is glaucoma and, possibly, cata-ractogenesis. Complications involving the ocular surface are generally milder. However, corticosteroids can aggravate superficial herpetic and fungal infections and significantly delay corneal epithelial healing (by up to 30 %).21 Cases of chronic keratitis, with calcium deposits and band keratopathy in some patients, have been described with topical corticosteroids. Finally, a rebound effect is frequent when topical steroids are used to treat an inflammatory condition. This can lead to chronicity, which can be difficult to distinguish from a relapse of the initial condition.
Nonsteroidal antiinflammatory drugs (NSAIDs) do not have all these drawbacks, but they can cause epithelial punctate keratitis after prolonged use. Although sometimes severe, the keratitis is reversible on treatment cessation.
Antiglaucoma Drugs
Many disorders of the ocular surface have been described after long-term use of drugs used to treat glaucoma (Table 8.1). The most extensively studied setting in this respect is the treatment of chronic open-angle glaucoma, as it often necessitates multiple treatments lasting many years. An initially normal, previously untreated conjunctiva can develop severe inflammatory manifestations with a risk of serious, totally unexpected complications.4
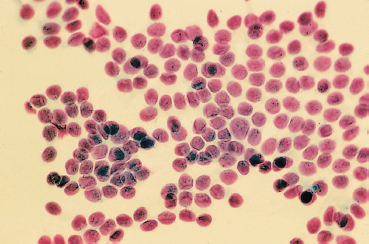
Fig. 8.5 Conjunctival impression cytology in a chronically treated glaucoma patient: note the loss of goblet cells
Like all eyedrops, antiglaucoma drugs can trigger abrupt and sometimes violent allergic reactions a few days after use. Treatment withdrawal is crucial, but the prognosis is generally good. These events occur in 3-5 % of cases and are often simple to diagnose. However, some patients can develop subacute reactions or late-onset manifestations which, for reasons that are poorly understood, only appear after several months of treatment. The subacute reactions are often less severe, and their timing is not particularly evocative of an iatrogenic cause; it is nevertheless important to diagnose the cause in order to avoid prolonging an increasingly toxic topical treatment.
Local irritation is extremely frequent, with a tingling sensation on instillation, foreign-body sensations, discreet conjunctival hyperemia, shortening of the tear break-up time, and corneal and conjunctival epithelial staining by fluorescein, rose bengal, or lissamine green. These minor signs are often attributed to a dryness which is intercurrent or worsened by the drugs used to treat the glaucoma. The prognosis is obviously better than that of the glaucoma itself, and this explains why these adverse effects are often overlooked. Simple beta-blocker monotherapy markedly alters mucosal secretion of the tear film, with a significant reduction in the density of goblet cells, during the first months of treatment22 (Fig. 8.5). Signs of significant cytotoxicity are clearly revealed by electron microscopy.
The most serious adverse effect is cicatrizing conjunctivitis (Fig. 8.4), which begins with a chronic inflammatory reaction, leading to true fibrosis of the conjunctiva, comparable to that observed in autoimmune bullous conditions.7 This very severe but fortunately rare complication occurs in a small percentage of cases, even with simple monotherapy, and onset is highly delayed. The mean time to onset after the beginning of treatment can exceed 15 years, making it extremely difficult to link to a topical treatment which had previously been perfectly tolerated. The pathogenesis of pseudopemphigoid (immuno-allergic or toxic) is still unknown.
Subconjunctival fibrosis is frequent but less severe. Patients treated for long periods, regardless of the nature of the eyedrops, develop fornix foreshortening.23 The fibrosis is sometimes diagnosed after the failure of filtering surgery due to fibrotic scarring at the filtration site. Several studies have shown that long-term use of antiglaucoma eyedrops favors inflammatory and fibro-blastic reactions24,25 (Fig. 8.6). It appears that two-drug therapy lasting more than a year (especially if it includes an epinephrine derivative) and monotherapy lasting more than 3 years are significant risk factors for surgical failure.24
Fig. 8.6 Histological aspect of the conjunctiva after failed filtration surgery: collagen bands surround the filtration zone; the conjunctival epithelium is normal
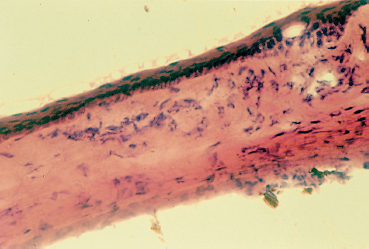
Fig. 8.7 Inflammatory aspect of the rat limbus and peripheral cornea after 1 month of treatment with 0.01 % benzalkonium chloride 4 times a day
Table 8.2 Histopathological analysis of the conjunctiva and trabeculum of patients treated surgically for primary open-angle glaucoma.26a
| Histological Aspect | Untreated | Long-term Monotherapy | Polytherapy |
|---|---|---|---|
| Conjunctiva | |||
| normal | 4/5 | 11/30 | 2/26 |
| fibrosis | 1/5 | 3/30 | 5/26 |
| inflammation | 0/5 | 16/30 | 19/26 |
| Trabeculum | |||
| normal | 4/5 | 13/22 | 6/23 |
| fibrosis | 1/5 | 6/22 | 8/23 |
| inflammation | 0/5 | 3/22 | 9/23 |
Table 8.3 Principal preservatives
| Quaternary ammoniums: benzalkonium chloride, cetrimide, benzododecinium bromide, etc. Amidines: chlorhexidine digluconate Alcohols: chlorobutanol Mercury compounds: phenylmercuric salts Calcium chelators (potentiate quaternary ammoniums): EDTA |
Immunohistochemistry on conjunctival biopsy specimens or impression cytology can reveal signs of inflammation in a very large proportion of patients.8,26 In one study of about 60 patients operated on for glaucoma (Table 8.2), systematic conjunctival examination showed an inflammatory or fibrotic aspect in 63 % of patients treated with a single drug for more than 3 years and in 93 % of patients treated with two or more drugs for at least 1 year. The trabeculum biopsy specimens from glaucoma patients were also infiltrated by inflammatory cells.26a Impression cytology also showed inflammatory signs in nearly two-thirds of glaucoma patients treated for long periods and/or with more than one drug.11
Few clinical studies have compared the toxicity of preservative-free and preservative-containing eyedrops. However, all available data tend to show that preservative-free antiglaucoma eyedrops are significantly less toxic.27,28 These results confirm experimental data obtained with cell cultures and animal models of surgery, which strongly favor preservative-free preparations.29
However, it should be remembered that authentic allergies to active drug substances can also occur (more often with epine-phrine or pilocarpine derivatives than with beta-blockers) and that some eyedrop preparations, such as those containing epinephrine, are themselves toxic, even if preservatives probably amplify their toxicity.
Preservatives (see Chapter 9, Focus  8)
8)
Preservatives are antiseptic substances added to eyedrops to delay microbial growth once the vial is opened. Although used at low concentrations, none are perfectly harmless: on the one hand, they can alter the chemical characteristics or stability of the active drug; on the other hand, they can damage the ocular surface. Their toxicity has long been known—since 1941 in the case of benzalkonium chloride. Quaternary ammonia is the best-studied preservative in terms of its toxicity.30 The toxicity of preservatives is often overlooked; for example, some product descriptions fail to mention the concentration, yet this is crucial for irritants with dose-dependent toxicity.
The two main classes of preservatives are mercury derivatives and quaternary ammonia, followed by chlorhexidine, methyl parahydroxybenzoate (paraben), and alcohol derivatives (Table 8.3). Mercury derivatives (often used in older eyedrops) are gradually being abandoned because of the higher frequency of allergic reactions and especially because of ecological problems linked to the mercury industry.
Benzalkonium chloride is found in almost all antiglaucoma eyedrop preparations, at a concentration that ranges from 0.004-0.02%. This is a toxic compound, not only for its target microorganisms but also for the ocular surface.31–33 It is not only directly cytotoxic for epithelial cells, even at very weak concentrations (as in current eyedrop preparations), but it also possesses surfactant properties, dissolving the lipid phase of the tear film and thereby inducing dryness.
Benzalkonium chloride significantly reduces the tear breakup time at concentrations as low as 0.004 %. At a concentration of 0.005% it is directly toxic for superficial cells, causing epithelial erosions.32 A concentration of 0.007 % only takes 90-100 seconds to lyze 50% of cultured conjunctival epithelial cells!34 At 0.01%, the concentration used in most eyedrops, benzalkonium chloride markedly alters the epithelium and stimulates infiltration of the limbus and conj unctiva by inflammatory cells (Figs. 8.7 and 8.8); corneal epithelial healing is delayed at a concentration of 0.02%. When administered via the anterior chamber or instilled at a concentration of 0.1 %, it destroys the endothelium and causes irreversible corneal edema, as observed during multiple dosing in patients with corneal ulceration. At even higher concentrations (1-2 %), benzalkonium totally destroys the anterior segment of experimental animals in less than a week.33 Benzalkonium toxicity may be potentiated by contact lenses, especially those made of hydroxyethyl methacrylate (HEMA), a material for which it has strong affinity.
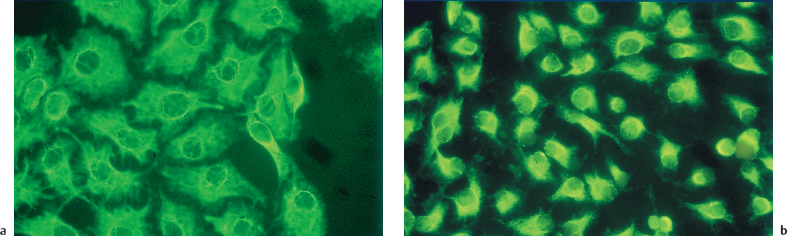
Fig. 8.8 Cultured conjunctival epithelial cells labeled with an anticytokeratin antibody:
a Normal aspect
b Intense cellular retraction after 10-minute contact with 0.01% benzalkonium chloride, despite a 24-hour recovery period in normal culture medium. The cells are apoptotic
The cytotoxic properties of benzalkonium chloride have even been used deliberately to improve the penetration of active drug substances into the anterior chamber. Although from a pharmacokinetic point of view the presence of benzalkonium indeed improves the penetration of the drug substance, comparative efficacy studies of preservative-containing and preservative-free antiglaucoma eyedrops have shown no significant difference.27,35 The fear that antiglaucoma eyedrops without benzalkonium might be less effective therefore seems to be groundless.
Conclusions
Major advances have been made in ocular toxicology, with respect to pathogenic and therapeutic mechanisms. A better knowledge of the mechanisms underlying drug allergy, the demonstration of toxic effects (which can be very discreet but cause severe late complications), and especially the gradual development of preservative-free eyedrops are examples of important advances in a field that was previously restricted to contact allergy. The development of truly nontoxic eyedrops is now a major challenge in ocular pharmacology, together with the search for effective compounds that can safely be instilled for several years onto what is an exquisitely sensitive and reactive surface tissue.
References
5. Bartlett JD, Jaanus SD. Clinical Ocular Pharmacology. Washington, Elsevier, 1995.
20. Leibowitz HM. Clinical evaluation of ciprofloxacin 0.3 % ophthalmic solution for treatment of bacterial keratitis. Am J Ophthalmol 1991; 112(S): 34-47.
30. Rigal D. L’épithélium cornéen. Paris, Masson: 1993: 391-419.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree