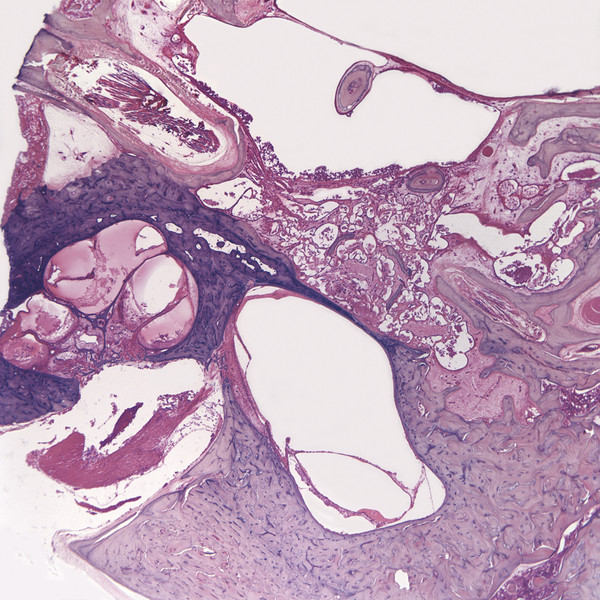
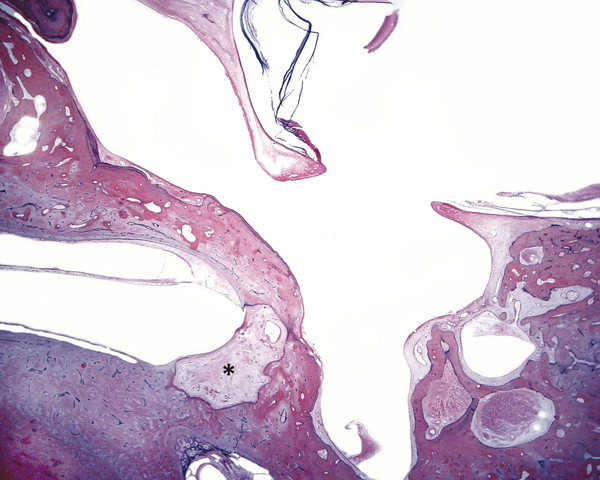
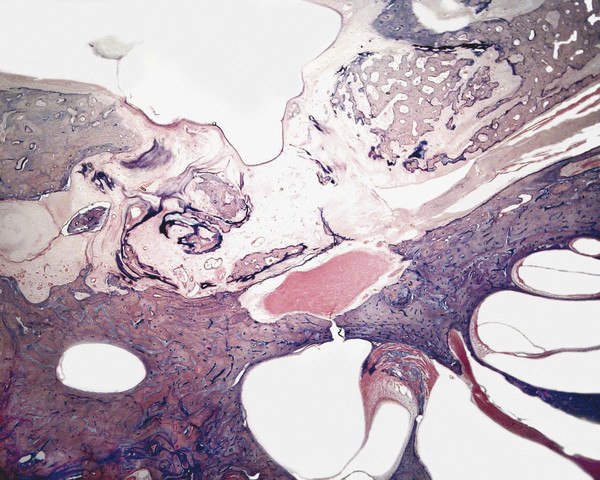
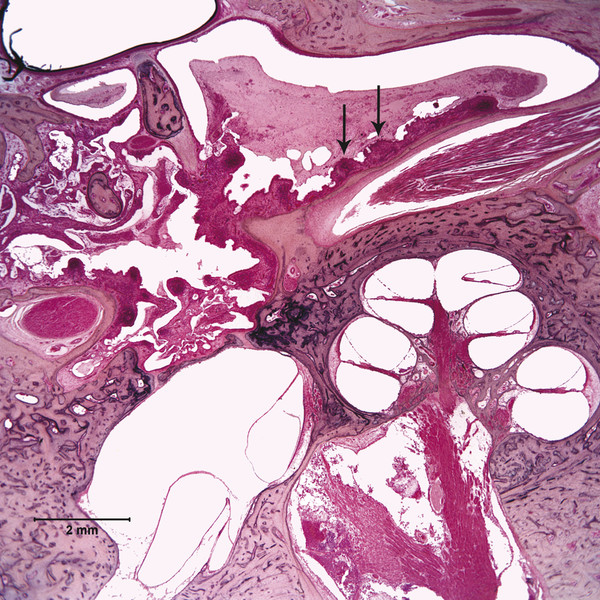
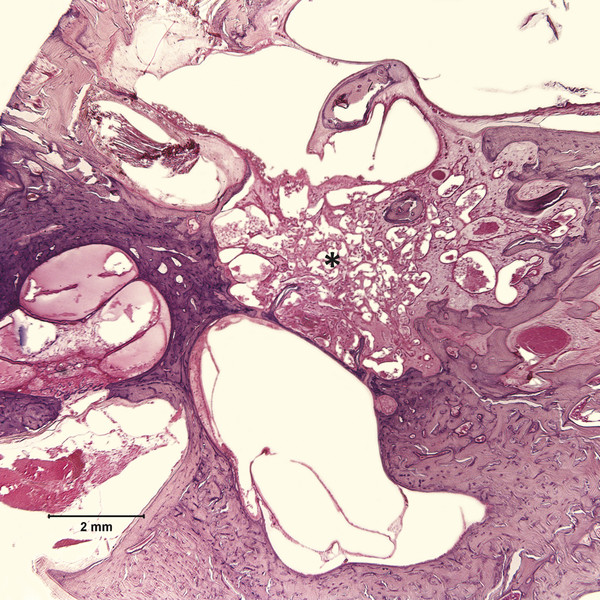
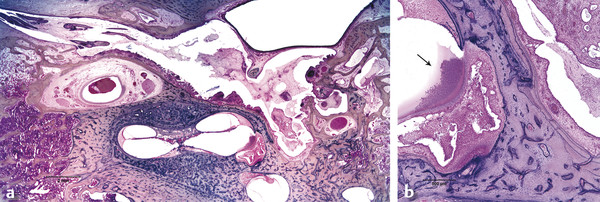
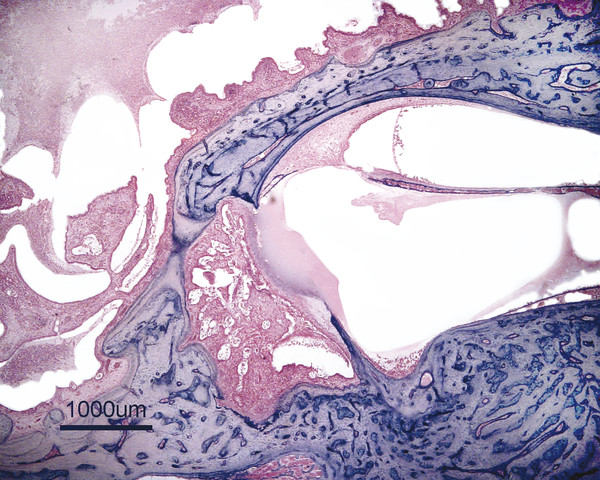
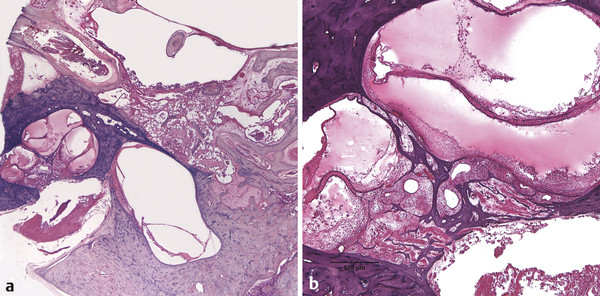
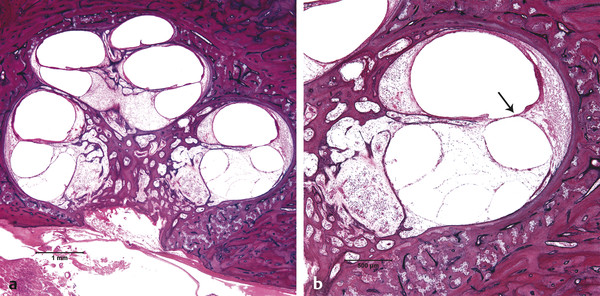
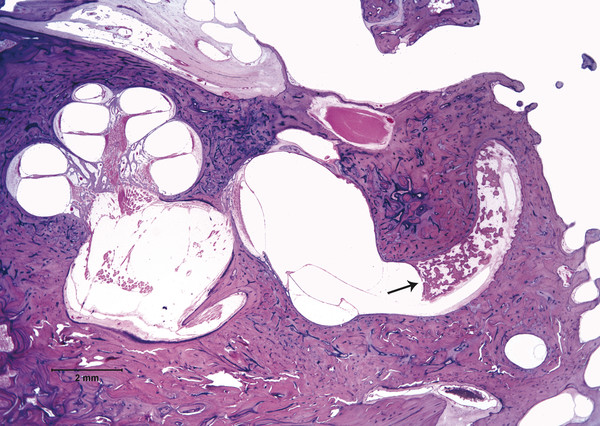
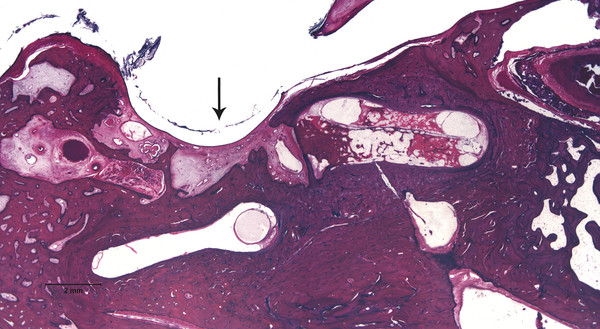
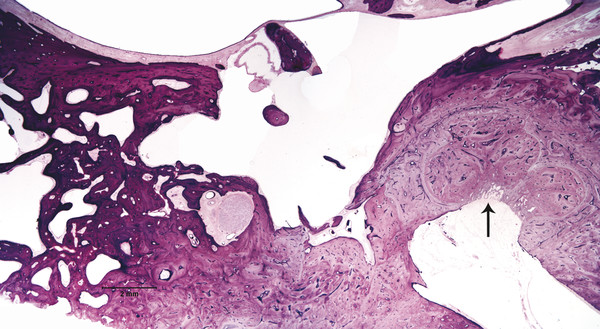
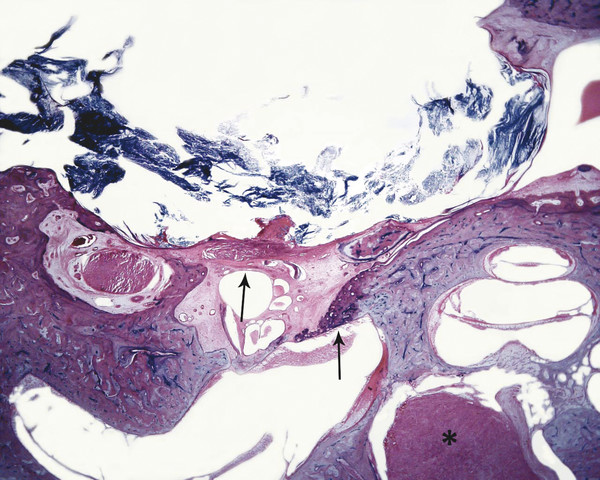
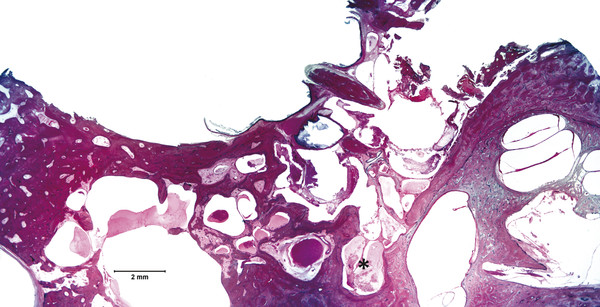
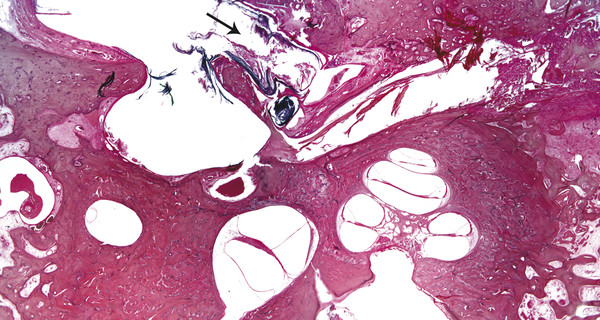
Otitis media is an inflammation of the middle ear cleft. It has been suggested that all types of otitis media represent different stages of a continuum of events, with the acute stage leading to a chronic stage.1 In addition, one form of otitis media may resolve or lead to a more chronic stage of the disease. Although otitis media with effusion is characterized by the presence of a clear fluid in the middle ear cleft, there is no evidence of secretory cell proliferation at an earlier stage. Over time, the subepithelial space is widened by edema, vascular dilation, and the infiltration of host defense cells. With the reabsorption of watery components of the fluid, the increased density of goblet cells, and the formation of secretory glands, the middle ear effusion becomes mucoid and there are chronic inflammatory changes in the middle ear cleft. Chronic otitis media (COM) has been clinically defined as a chronic discharge from the middle ear in the presence of a perforation of the tympanic membrane. Histopathologically, COM is defined when the inflammatory process has entered a chronic phase and permanent alteration of middle ear structures is seen (▶ Fig. 6.1; ▶ Fig. 6.2).2 The pathologic changes in the tympanic membrane due to COM include perforation, retractions, myringosclerosis, pseudo-cystic spaces, and hemorrhage.3 Fig. 6.1 This was an 18-month-old patient with chronic otitis media and seropurulent labyrinthitis. She had otogenic meningitis. Her right ear showed residual mesenchyme and chronic changes, including granulation tissue, fibrocystic changes, and cholesterol granuloma in the middle ear cleft. Note that the tympanic membrane was intact. Fig. 6.2 This was an 82-year-old patient with a history of bilateral tympanic membrane perforation. Her right ear showed tympanosclerosis in the middle ear and tympanic membrane. The middle ear mucosa and subepithelial space were thickened. Granulation tissue was seen around the round window membrane (*). The histopathological changes of the middle ear in COM are similar in the temporal bones with and without perforation of the tympanic membrane.4 The most frequently encountered pathological tissues in the middle ear of patients with COM are granulation tissue; osteitis; fibrous thickening of the mucoperiosteum; fluid; cholesteatoma; and cholesterol granuloma;2 with granulation tissue being the most common.3 Throughout the course of COM, osseous changes, including osteitis, necrosis, reabsorption, and osteoneogenesis, are occurring in the middle ear cleft (▶ Fig. 6.3).2 The bony involvement parallels the severity of the inflammatory process. In the generalized form of active COM, although all three ossicles can show chronic changes, the incus is the most commonly involved, being followed in frequency by the stapes, and then the malleus.2 Fig. 6.3 This was a 75-year-old patient with a long history of chronic bilateral ear disease with frequent discharge since the age of 22 years. From age 72 years, he noticed episodic vertigo, which subsequently recurred. Histopathology evaluation showed chronic changes, such as ossicular erosion, new bone formation, and tympanosclerosis in the middle ear cavity. The clinical definition of COM that requires a tympanic membrane perforation and/or drainage from the middle ear excludes a large number of cases that show chronic pathologic changes in the middle ear cleft. Paparella, Schachern, and Cureogluintroduced the concept of chronic silent otitis media on the basis of histopathologic evidence of chronic infection in the middle ear cleft in the absence of obvious ear-related symptoms and changes in the tympanic membrane (▶ Fig. 6.4).5 In 1999, Jaisinghani, Paparella, Schachern, and Le studied 150 temporal bones from 97 patients with COM to correlate the presence of middle ear pathologies with the changes in the tympanic membrane.3 Almost half of the temporal bones with middle ear pathology had no associated tympanic membrane pathology. Fig. 6.4 This was a 10-month-old patient with otogenic meningitis. Histopathologic examination of his ears showed bilateral mucopurulent otitis media, polypoid middle ear mucosa (arrows), and purulent labyrinthitis. Fibrocystic changes can be seen in the facial recess and around the stapedial footplate. Note that the tympanic membrane is intact. It has been stated that the incidence of COM associated with an intact tympanic membrane was four times more common than the incidence of COM with a perforated membrane.5 In addition, the otopathological changes of the middle ear cleft in COM are similar in patients with and without perforation of the tympanic membrane.5 In 1986, Paparella, Goycoolea, Bassiouni, and Koutroupas reported 10 temporal bones from 5 patients below the age of 2 years who died of Hemophilus influenzae meningitis.6 None of the patients studied had documented clinical manifestations of any ear pathology before the onset of meningitis. Temporal bone findings included acute inflammatory disease as well as chronic pathologies such as granulation tissue, fibrosis, and mononuclear cell infiltration. These chronic irreversible pathologies in the middle ear and the presence of labyrinthitis in all cases indicated that the otitis media had been present for some time, suggesting meningitis of otogenic origin (▶ Fig. 6.5). Fig. 6.5 This was an 18-month-old patient with otogenic meningitis. Histopathologic examination showed bilateral chronic otitis media with chronic changes, such as fibrocystic structures and granulation tissue as well as cholesterol granuloma in the middle ear cavity and seropurulent labyrinthitis (*). Because the chronic pathology is undetected, there is a lack of clinical treatment of otitis media, increasing the risk of complications due to the underlying problems (▶ Fig. 6.6).5 As a result of the silent middle ear infection process, intratemporal problems, such as endolymphatic hydrops, labyrinthitis, facial palsy, and sensorineural hearing loss, and/or intracranial complications, such as meningitis or brain abscess, can be seen.5,7 The clinician should, therefore, be aware that an intact tympanic membrane does not necessarily preclude the presence of pathologic changes in the middle ear cleft. Fig. 6.6 (a) This was a 10-month-old patient with silent otitis media and otogenic meningitis. The tympanic membrane is intact. Histopathologic examination of his ears showed bilateral mucopurulent otitis media, polypoid middle ear mucosa, and purulent labyrinthitis. Note the presence of granulation tissue around the round window membrane. The mucopurulent secretions crossed the round window membrane into the scala tympani and the patient ended up with purulent labyrinthitis. (b) A higher magnification of the round window niche, which facilitates the accumulation of pus in the middle ear site (arrow). The inner ear is intimately associated with the middle ear both functionally and anatomically. Functional changes of the middle ear mechanics affect the transmission of sound to the inner ear, whereas areas of anatomical connections allow for the passage of microorganisms, cytokines, toxins, or drugs from the middle to the inner ear, with potentially serious sequelae. The round window membrane, which is the only soft tissue barrier between the middle and inner ear and which lies within a niche and adjacent to the sinus tympani, facilitating the accumulation of pus, is vulnerable to toxic substances within the middle ear (▶ Fig. 6.6). Inner ear sequelae that include labyrinthitis (▶ Fig. 6.1), endolymphatic hydrops (▶ Fig. 6.1),8 outer hair cell losses, and as a result, sensorineural hearing loss and vestibular disturbances can occur in both acute otitis media (AOM) and COM.9,10 In addition, a high-frequency sensorineural hearing loss can be clinically significant in cases of COM given that this denotes perilymphatic inflammation. The basal turn of the cochlea is the most common location of the pathologic changes in the inner ear during this middle ear interaction (▶ Fig. 6.7). This results in a temporary threshold shift, which may become permanent over time. Cureoglu et al showed loss of outer and inner hair cells, especially in the basal turn of the cochlea, and a decrease in the areas of the stria vascularis and spiral ligament in the basal turn of the cochlea of temporal bones with unilateral COM.11 Fig. 6.7 This was another case involving a 10-month-old patient with otogenic meningitis. His left ear showed mucopurulent otitis media with polypoid middle ear mucosa as well as purulent labyrinthitis. Note that the basal turn of the cochlea is the most common location for pathologic changes in the inner ear during middle ear/inner ear interactions. This is characterized by sensorineural hearing loss and vertigo that usually shows partial or complete recovery. It is usually a reaction to exotoxins rather than actual bacterial invasion (▶ Fig. 6.6).12 The presence of vertigo in a patient with otitis media may indicate the presence of serous labyrinthitis. If it is diagnosed and treated before suppuration starts, the damage in the inner ear will be reversible. The histopathological markers of serous labyrinthitis can be described as endolymphatic hydrops, eosinophilic fluid staining due to increased proteinaceous content, and sero-fibrinous strands in the labyrinthine spaces without any associated host inflammatory cell invasion.13 The most common underlying problem of circumscribed labyrinthitis is cholesteatoma erosion. The erosion makes the membranous labyrinth sensitive to environmental changes such as temperature or pressure. The inflammation is localized to a bony surface of the labyrinth without the invasion of endosteum. If the patient starts to have sensorineural hearing loss in addition to a positional vertigo, treatment should be initiated for serous labyrinthitis to prevent suppurative labyrinthitis. Hispathologically, suppurative labyrinthitis (▶ Fig. 6.8) has three stages; however, any intracranial complication may occur at any stage, and the patient may end up with a complete loss of labyrinthine functions.14 Fig. 6.8 (a) This was an 18-month-old patient with chronic otitis media. She had otogenic meningitis. Her right ear showed residual mesenchyme and chronic changes, including fibrocystic changes and cholesterol granuloma in the middle ear cleft. (b) A higher magnification of the inner ear showing seropurulent labyrinthitis and inner ear damage. During the first stage, purulent effusion fills the perilymphatic spaces but spares the endolymph. The main symptoms are severe vertigo and hearing loss at this stage. During the second stage, polymorphonuclear lymphocytes (PMLs) dominate in the area with fibrillar precipitates, especially in the perilymphatic space (▶ Fig. 6.9). Endolymphatic hydrops is also seen. This stage begins around 2 weeks after the onset of infection. Although vertigo subsides, sensorineural hearing loss progresses. Fig. 6.9 (a) This was a 29-year-old patient with a diagnosis of acute leukemia. He had a history of dizziness and fluctuant hearing loss. Histopathologic exam showed bilateral fibrous labyrinthitis and profound endolymphatic hydrops. (b) A higher magnification of the basal turn of the cochlea showing severe organ of Corti damage, hair cell losses, reduced ganglion cells, and stria vascularis atrophy (arrow). The third stage represents chronic labyrinthitis: fibroblasts predominate and small capillaries occur as a result of increased angiogenesis. Eventually osteogenesis may occur (in an animal model, around 2 months). After being stimulated by infection or inflammatory irritation following breakdown of cells, mesenchymal cells, which are located around capillaries, the undersurface of the basilar membrane, and endosteum that lines the perilymphatic space, rapidly differentiate into fibroblasts that produce collagen fibers, endothelial cells that form capillaries, and osteoblasts. The osteoblasts form trabeculae (lamellar or spongy bone) that join together and incorporate small blood vessels within them. Then, haversian canals form from the condensation of lamellar bone, which could be difficult to distinguish from the normal bone around it. Pathologic new bone formation within the otic capsule in response to inflammatory and/or destructive processes is called labyrinthine ossificans, which represents the terminal stage of repair of labyrinthitis. At early stages, only scala tympani of the basal turn of the cochlea is involved. Later, all parts of the inner ear are involved. In humans, it may take years to have a complete labyrinthitis ossificans (▶ Fig. 6.10; ▶ Fig. 6.11; ▶ Fig. 6.12). Fig. 6.10 This was a 49-year-old patient with acute leukemia. Histopathologic examination of her ears showed labyrinthitis ossificans in the lateral semicircular canal (arrow) and hydropic changes in the inner ear. Fig. 6.11 This was a 74-year-old patient with a history of chronic otitis media dating back to childhood. At age 36 years, he experienced dizziness and total deafness in the right ear. He had a canal wall-down mastoidectomy (arrow) in both ears. At age 50 years, reactivation of chronic otitis media in his left ear occurred and caused vertiginous spells. In this section, labyrinthitis ossificans in the basal turn of the cochlea and granulation tissue at the round window niche can be clearly seen. Fig. 6.12 This was a patient with recurrent episodes of chronic suppurative otitis media. Note the complete ossification of the cochlea (arrow). Granulation tissue is the most common pathology encountered in chronic ears. It consists of vascularized connective tissue with a component of acute inflammatory exudation. The most common sites for granulation tissue are the epitympanum, mesotympanum, round window niche, and mastoid antrum (▶ Fig. 6.13).2,3,5 During the formation of granulation tissue, the mucous membrane is thickened by edema, submucosal fibrosis, and infiltration with chronic inflammatory cells. Later, mucosal edema may progress to the formation of polyps.1 With maturation, granulation tissue becomes more fibrous and less cellular, with increased collagen and reticular formation.2 Granulation tissue may lead to adhesions or tympanosclerosis in the middle ear.1 This activity, if unchecked, may lead to serious complications, including the destruction of the inner ear. Fig. 6.13 This was an 86-year-old patient with a history of congenital deafness. Histopathologic examination showed bilateral chronic otitis media and cholesteatoma. The left ear showed granulation tissue, cholesterol granuloma, tympanosclerosis, otosclerosis (arrow), acoustic neuroma (*), and serous labyrinthitis. Cholesteatoma can be described as a keratinized squamous epithelium in the middle ear cleft (▶ Fig. 6.14). It is classified as congenital or acquired based on its presumed etiology. The pathogenesis of cholesteatoma, outlined in Chapter 4, is multifactorial. Fig. 6.14 This was an 83-year-old patient with carcinoma of the bladder. He had a history of bilateral decreased hearing beginning at the age of 60 years. Histopathologic examination showed bilateral chronic otitis media and cholesteatoma. His left ear showed chronic changes and cholesteatoma with seropurulent secretion in the mesotympanum and sinus tympani in the middle ear (*). With acquired cholesteatoma, the pathogenesis is unclear and remains controversial. The most accepted theory is the invagination theory (also known as the retraction pocket theory) (▶ Fig. 6.15). Eustachian tube dysfunction creates negative pressure and causes loss of structural support for the tympanic membrane; eventually atelectasis results in retraction pockets (▶ Fig. 6.16). Keratin accumulates within these pockets and progresses to cholesteatoma (▶ Fig. 6.17). Fig. 6.15 This was a 50-year-old patient with metastatic carcinoma of the breast. Histopathologic examination of the ears showed bilateral chronic otitis media. The left ear showed chronic changes, including cholesteatoma, cholesterol granuloma, and a perforated tympanic membrane. Note the complete retraction of the tympanic membrane into the middle ear cavity and the accumulation of cholesteatoma in the anterior part of the retraction as well as a cholesteatoma mass in the anterior middle ear cavity (arrow).
6.2 Chronic Silent Otitis Media
6.3 Middle Ear/Inner Ear Interaction
6.3.1 Serous (Toxic) Labyrinthitis
6.3.2 Circumscribed (Localized) Labyrinthitis
6.3.3 Suppurative Labyrinthitis
6.4 Pathological Changes in the Middle Ear Cleft in COM
6.4.1 Granulation Tissue
6.4.2 Cholesteatoma
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


