Chapter 86 Helminthic Disease
Introduction
Helminths have plagued mankind since before the era of our earliest recorded history.1 It is possible to recognize features of helminthic infections from the ancient writings of Hippocrates, Egyptian medical papyri, and the Bible.2 Nematode worms or roundworms are one of the two major phyla of the Helminths and include the major intestinal human worms (e.g., Ascaris lumbricoides, Trichuris trichiura, Necator americanus, Ancylostoma duodenale, Strongyloides stercoralis), animal worms (e.g., Toxocara canis, Toxocara cati, Ancylosoma caninum, Bayliascaris procyonis), and the filarial worms that can cause lymphatic filariasis (e.g., Wuchereria bancrofti, Brugia malayi) or onchocerciasis (Onchocerca volvulus).1 The other phylum of the Helminths is called Platyhelminths, which is divided into trematodes (e.g., Schistosoma mansoni) and cestodes (e.g., Taenia solium, Taenia saginata). Many kinds of adult worms cannot develop on their own and parasitize human intestine; some in their larval stage can infect humans directly, causing tissue damage that includes the eye.3 It is estimated that almost 1 billion people worldwide, especially in tropical areas and developing countries, are infected with one or more helminths.4 This chapter discusses the most common helminthic diseases that affect the posterior segment of the eye: ocular toxocariasis, diffuse unilateral subacute neuroretinitis (DUSN), ocular onchocerciasis, and cysticercosis.
Ocular toxocariasis
Toxocariasis is a infection caused by the nematode T. canis and less frequently by other roundworms such as T. cati.3,5,6 Systemic effects of Toxocara infestation in the human are termed visceral larval migrans (VLM). VLM is usually a self-limited disease and typically affects young children from 6 months to 4 years of age.5 Findings associated with the disorder range from a mild to moderate eosinophilia in an asymptomatic patient, fever, pallor, anorexia, malaise, hepatomegaly, and transient infiltrates of the lungs in symptomatic VLM to, more infrequently, a fulminating and fatal disease associated with pneumonia, congestive heart failure, or convulsions.3,5,7 Ocular toxocariasis is unusual in adults but is an important cause of visual impairment during childhood, accounting for approximately 1% of all cases of uveitis among all ages in referral centers in the United States and Japan8,9 and 3% of panuveitis in Switzerland.10 Exact correlation between VLM and ocular toxocariasis is not well determined but patients with systemic disease rarely present with ocular involvement. In one study of 245 cases of VLM, only 5% had any indication of ocular disease.11
History
In 1907 Leiper first described the infestation of humans with canine nematodes.12 Later, Wilder, in 1950, described nematode ocular infection.13 In her study, Wilder reviewed the histopathology of 46 eyes primarily enucleated for suspicion of intraocular malignancy and nematode larvae were found in 24 eyes. Two years later Beaver and coworkers identified a Toxocara species larva in a liver biopsy from a child with visceral lesions and eosinophilia, identifying the etiological agent with this previously reported syndrome in children.14 Nichols, in 1956, reported identifying T. canis (Toxocaridae family) in specimens originally studied by Wilder who first thought the worm to be part of Ancylostomidae family.15 These studies demonstrated a common etiology for systemic and ocular diseases. In 1959 Irvine and Irvine16 described histologic findings in a 4-year-old child presenting with strabismus, decreased vision, and retinal detachment. The eye was enucleated for suspicion of a tumor but an eosinophilic abscess with Toxocara canis larva on the pars plana region was demonstrated. This patient differed from cases reported by Wilder since the patient presented with clear media and the retina could be examined.13 In 1960 Ashton17 described four histopathologically proven cases of Toxocara endophthalmitis that presented with a posterior pole granuloma in a distinct fashion that represented a third classic form of this disorder. Duguid, in 1961,18 described a case series of ocular toxocariasis and emphasized the difference between the posterior pole granuloma and diffuse chronic endophthalmitis, suggesting that clinical diagnosis could be perfomed in certain cases. In 1971 Wilkinson and Welch19 reported 41 cases of proven or presumed ocular toxocariasis. Eyes with chronic endophthalmitis and leukocoria were much more likely to have been enucleated, whereas eyes with the posterior or peripheral isolated and visible granulomas were usually diagnosed clinically, and enucleation was not performed to confirm the diagnosis. Since then, several other relatively unusual forms of ocular Toxocara have been described and these are briefly discussed later. Association of T. canis and diffuse unilateral subacute neuroretinitis will be discussed separately in this chapter.
Parasitology
The Toxocara genus comprises 21 species including T. canis which is the most common cause of toxocariasis.3,5,6,20 T. canis is a natural parasite of dogs and occasionally can infest human and other paratenic hosts such as rodents and rabbits.21 Adult worms are cylindrical and long, measuring from 75 to 120 mm. Male worms are longer than female and in their development find five different stages from larvae to adult parasite.20 Adult forms are found only on small intestine of puppies until 6 months of age (definitive host) and pregnant and lactating bitches. In older dogs and humans the cycle is not complete and because of this only the definitive host where worms reach sexual maturation can shed eggs in feces. To understand the pathogenesis of T. canis and the development of VLM and ocular toxocariasis it is necessary to understand the parasite life cycle.
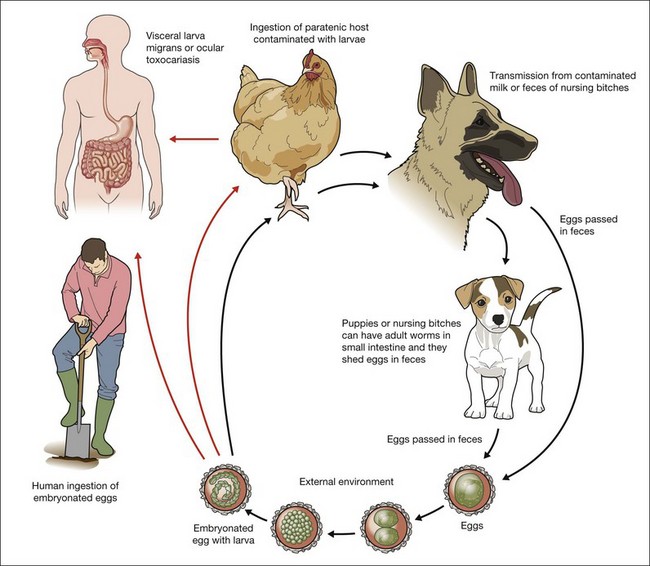
Eggs lacking an embryo are shed in the feces of the definitive host (puppies). The eggs then develop an embryo and become infective in the environment in 2–6 weeks. Following ingestion by dogs or other parasite hosts, the infective eggs hatch and larvae penetrate the intestine wall and are carried by the circulation to a wide variety of tissues (liver, heart, lungs, brain, muscle, eyes). In the human and in adult dogs, this completes the life cycle, and the parasites are usually enclosed by an inflammatory granulomatous response that is primarily eosinophilic, although the larvae may remain viable for years under such conditions.3,5,22,23 During pregnancy, the host response in the infected bitch is altered, and previously encysted larvae may resume migration,23 crossing the placenta and affecting the canine fetus. In puppies, larvae reaching the lungs are coughed up and swallowed and, reaching the small intestine, they quickly mature into adult worms that produce infectious ova that are passed in fecal material. Prenatal infection is the primary route of transmission to puppies, however they can become infected after birth by ingestion of milk or feces from an infected nursing bitch.6,22 The infection rate in puppies may approach 100%24,25 and they start to shed over 200 000 eggs per day in feces from 3 to 6 weeks after initial infestation and may shed eggs until they are about 6 months old when the mature worms die20 (Fig. 86.1).
Pathophysiology
Humans can be infested by ingestion of embryonated eggs or larvae. Eggs can be ingested accidentally after contact with puppies or by geophagia of soil containing embryonated eggs.26–28 Outdoor parks and playgrounds (sandboxes) can be highly contaminated with embryonated eggs of Toxocara, since in this environment people routinely walk their pets.29–33 Under optimal conditions embryonated eggs may remain viable for years.34 Direct contact with untreated puppies is also an important form of transmission. Amaral et al. have shown that perianal hair from stray as well as owned dogs had embryonated eggs in 24% of cases and 99% of the total number of eggs were found in puppies.25 A less frequent method of transmission is the ingestion of larvae from contamined meat. Several studies described infection by eating raw meat or liver from animals such as cows, chicken, and ducks.35,36,37
After ingesting embryonated ova, the parasites mature in the small intestine and transform to second stage larvae which penetrate the intestine wall, enter the lymphatic and portal circulation, and are disseminated to liver and lungs. Then the larvae reach the heart and are disseminated to other organs such as brain, kidney, heart, muscle, and eyes.20 In human beings the parasites are enclosed by an inflammatory granulomatous reaction and the parasites do not evolve further.22 The eosinophilic granuloma comprise the larva enveloped by a central core of eosinophils surrounded by mononuclear cells, histiocytes, epithelioid cells and giant cells. Because of the inability of the parasite to mature into an adult form in humans, a search for T. canis ova in human feces is inevitably unrewarding.38
T. canis can cause either VLM and ocular toxocariasis and the clinical manifestation depends on factors such as host immunological condition, inflammatory response, number of eggs/larvae ingested, frequency of ingestion, and localization of the second stage larvae in human tissues.20 A large amount of ingested embryonated eggs may lead to a rapid increase in eosinophilia and rising antibody levels.20 One study has shown that the degree of systemic eosinophilia in VLM has been correlated with level of exposure to Toxocara larvae.39 A lower exposure to embryonated eggs may produce lower levels of antibodies and eosinophilia and may permit wider larval migration, reaching the eye.20 Intraocular involvement by T. canis tends to occur in young patients, but in individuals who have developed ocular toxocariasis the age of onset is usually older than patients with VLM.7,13,22,40 Mean age of presentation for systemic VLM is 2 years5 versus 7.5 years for ocular toxocariasis.11
Clinical presentations
Although the average age of presentation is around 8 years for ocular toxocariasis,5,11 the age at presentation may range from 2 to 30 years11,41 and individual cases have been reported in adults with age up to 62 years.41,42 Toxocara canis infection in adults and very young children is rare, probably because of better hygiene habits of adults and less contact with puppies, sandboxes, and geophagia.
The ocular involvement is typically unilateral, and bilateral disease is quite rare.20,43 Patients usually present with unilateral visual impairment, leukokoria, and or strabismus in a relatively quiet eye. Careful slit-lamp examination can show cells in the anterior chamber (73%) and vitreous (100%),44 and vitritis causing decrease in visual acuity was reported in 53% in one series.9
The presence of a retinal granuloma is the most common finding in ocular toxocariasis. Wilkinson and Welch,19 in a study with 41 eyes diagnosed with the disease, reported that a peripheral granulomatous mass with relatively clear media was the most common form of presentation, being seen in 18 of 41 (44%) cases.19 Stewart et al.,9 in 2005, described 22 cases of ocular toxocariasis and in 50% of them peripheral granuloma was diagnosed. However, an isolated posterior pole inflammatory mass was the most common form of presentation in a series of 100 cases reported by Hagler et al.45 and in 53% of 30 cases published by Oréfice and coworkers.44
Shields,7 in a review in 1984, divided ocular toxocariasis into nine different categories, including DUSN.7 Despite T. canis being described as one of the possible nematodes to cause DUSN, other roundworms are also causes of DUSN and therefore the disorder will be discussed separately in this chapter.
Peripheral granuloma
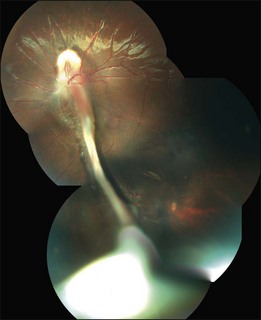
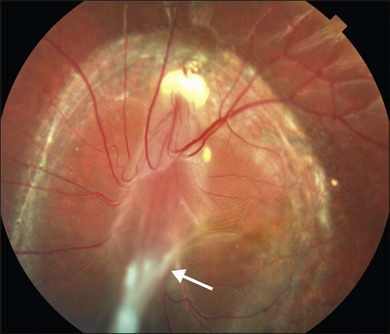
The peripheral granuloma, also described as the peripheral inflammatory mass form of Toxocara endophthalmitis, is usually seen in a quiet eye with varying levels of decreased vision and strabismus.19 The vitreous and anterior chamber show mild to moderate reaction. Intense vitritis can occur, however at diagnosis the vitreous is generally clear. Peripheral granuloma presents as a dense, white inflammatory mass in the periphery of the retina. Localized traction on the retina may result in the production of a typical retinal fold from the periphery to the optic nerve (Fig. 86.2). This mass may be quite localized, spherical, and similar to those observed in the posterior pole. Fibrocellular bands may be observed running from a peripheral inflammatory mass to the more posterior retina or the optic nerve (Fig. 86.3). The prognosis in eyes with peripheral granulomatous inflammation is usually relatively good and visual acuity can be preserved. By the time this diagnosis is made, active inflammation is usually not progressive.19 Ultrasound biomicroscopy study of 15 eyes with peripheral toxocariasis has shown alterations such as vitreous membranes in 86.6%, granuloma in 73.3%, pseudocysts in 53.3%, and thickening of ciliary body in 40% of studied eyes.46 Intra- and epiretinal traction bands can lead to production of both traction and rhegmatogenous retinal detachments, macular displacement and distortion, and optic nerve dysfunction.
Posterior pole granuloma
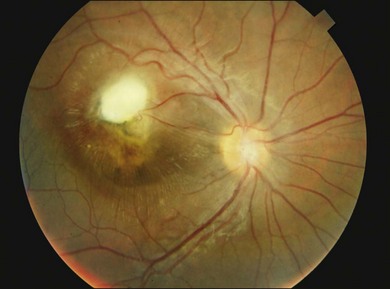
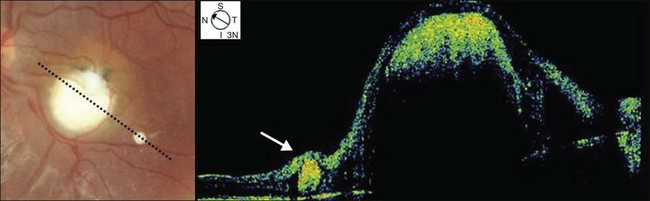
Posterior pole granuloma presents as white or gray spherical intraretinal or subretinal granuloma affecting the posterior pole. Most of the time it is diagnosed in a cicatricial stage when the lesion appears as a well-defined subretinal mass, measuring from 500 to 3000 µm, with no hemorrhage or exudates. Traction bands running from the mass to the surrounding retina may be observed. With cicatricial granulomas, there is very mild or no anterior chamber reaction and a low amount of cells in the vitreous cavity. Perilesional retina can show hyperpigmentation of retinal pigmented epithelium (RPE) and wrinkling of internal limiting membrane (Fig. 86.4). An optical coherence tomography case report has shown that the granuloma presents as a well-defined subretinal mass above the RPE (Fig. 86.5).47 Active disease can show different stages of severity from mild anterior chamber and vitreous reaction to a very intense vitritis as seen in the chronic endophthalmitis form of the disease. During the active phase the posterior pole granuloma is observed as an ill-defined hazy mass surrounded by retinal exudates or hemorrhages.19,20 A granuloma is usually seen in the posterior pole temporal to the optic nerve head; however, the occurrence of optic nerve granuloma has been described.48 Due to posterior pole involvement, patients present with central visual impairment and can have strabismus and leukokoria. Macular lesions may be observed in association with peripheral inflammatory masses and choroidal neovascularization may occur as a late complication.45,49,50
Chronic endophthalmitis
Patients with chronic endophthalmitis are typically younger than those with more localized granulomatous forms of the disease. There is usually relatively clear media.19 Localized granulomatous forms may be a late presentation of an acute form of the disease that had healed without development of retinal detachment or phthisis bulbi. As the vitreous clears it is possible to visualize the retinal lesions.19 The history is usually negative for trauma, intraocular surgery, or bacterial and fungal endophthalmitis. However, a history of playing in sandboxes in public places and geophagia may be obtained.19,27,28 External inspection usually reveals a quiet eye but severe intraocular inflammation can be present. In such cases leukokoria may be seen. Patients present with anterior chamber reaction that can vary from moderate to intense granulomatous reaction and can lead to hypopyon and posterior synechiae. The vitreous shows dense cellular infiltration with hazy media that can clear or develop cyclitic membranes, retinal traction and detachment that can be diagnosed ultrasonographically.51 If vitreous clears, a localized granuloma can be seen. The prognosis in this form of endophthalmitis depends primarily on the degree of intravitreal organization and the development of complications such as retrolental membranes, cataract, glaucoma, retinal detachment, and phthisis bulbi.
Atypical presentations
Atypical forms of ocular toxocariasis have been reported. Most of them had a presumed diagnosis. These include: optic nerve granuloma and optic neuritis;48 motile subretinal nematode,52 which more recently many consider to be an early phase of DUSN; diffuse chorioretinitis;7 conjunctivitis; keratitis; and lens involvement, including motile intralenticular larva and cataract.7,22
Diagnosis
The enzyme-linked immunosorbent assay (ELISA) is the current serum test of choice to detect exposure to T. canis.7 Intraocular fluids (aqueous humor, vitreous) can also be used to detect antibodies in a suspected case of ocular toxocariasis with a negative serum ELISA test.7,41,53 The ELISA test employs antigens secreted by the second-stage larva and recombinant antigens have been produced from them that add even greater specificity to an already-reliable test (approximately 92%). The ELISA has a reasonably high degree of sensitivity as well (approximately 78%), at a titer greater than 1 : 32.54 The immunodiagnostic tests used for VLM are not as reliable for ocular toxocariasis, and eosinophil count is not usually increased in these patients. Centers for Disease Control and Prevention consider serum ELISA titers less than 1 : 32 to be insignificant in the diagnosis of systemic toxocariasis55 but others have stated that a serum titer of 1 : 8 is sufficient to support a diagnosis of ocular toxocariasis if the patient has signs and symptoms compatible with that disorder.45 A positive test may represent a previous contact with the parasite and it does not necessarily mean that the patient suffers from either VLM or ocular toxocariasis. Ellis and coworkers have shown that 23% of 333 kindergarten children in rural North Carolina without signs of ocular toxocariasis exhibited a serum titer ≥1 : 32, and 32% had a titer ≥1 : 16.55 Prestes-Carneiro and coworkers have shown that 14% of 182 ramdomly studied patients in rural Sao Paulo, Brazil, presented positive ELISA tests for T. canis.56 Thus a positive serum titer cannot be used to confirm absolutely a diagnosis of ocular toxocariasis, although the absence of any serologic evidence of Toxocara infestation does not exclude the diagnosis but may assist in reducing the odds of this organism being the cause of ocular disease. Oréfice and coworkers studying 30 cases of presumed ocular toxocariasis found a positive ELISA test in 88% of them.44 Another study has shown that only 45% of patients with clinically diagnosed ocular toxocariasis had titers higher than 1 : 32.54
Cytologic study of aqueous humor or vitrectomy samples may also be helpful in confirming the diagnosis of ocular toxocariasis. The presence of eosinophils in intraocular fluids is consistent with intraocular Toxocara. Remnants of Toxocara organisms have occasionally been recovered from vitrectomy specimens obtained at surgery.57
Differential diagnosis
Retinoblastoma
Retinoblastoma is the most common intraocular malignancy of childhood and also the most important entity frequently confused with ocular toxocariasis.7 The first report of nematode intraocular endophthalmitis by Wilder13 included enucleated eyes for suspicion of intraocular retinoblastoma. Shields and coworkers58 reported that among 500 consecutive patients referred to them with leukokoria 42% had pseudoretinoblastoma and among them 15.6% had a final diagnosis of ocular toxocariasis. Patients with retinoblastoma are usually diagnosed before 2 years of age, being younger than a typical child with ocular toxocariasis. Sporadic retinoblastomas are more frequently confused with ocular toxocariasis since most of them are unilateral, and lack a family history of the malignancy. On ophthalmic evaluation, retinoblastoma usually does not present signs of intraocular inflammation, with clear vitreous and lack of posterior synechiae, cataract or cyclitic membranes, and the tumor presents a growing pattern that does not occur in toxocara granulomas.7 In cases with opaque media the differential diagnosis is harder. Ultrasonography and CT scan may be very valuable in demonstrating an intraocular mass and calcifications of a retinoblastoma, or vitreous organization and tractional membranes, which is more frequently observed in ocular toxocariasis. In cases where the differential diagnosis is difficult, evaluation of intraocular fluids by ELISA or cytology may be of particular importance. If retinoblastoma is suspected, however, biopsy should be avoided and consultation with other experts may be a better path to reliable diagnosis. Risks and concerns relating to sampling retinoblastoma tissue are discussed in the oncology section of this book.
Toxoplasmosis
The typical lesion seen with protozoal Toxoplama gondii infection is focal granulomatous and necrotizing retinochoroiditis. It appears as a whitish or yellowish lesion, slightly elevated and with poorly defined limits.20 During active disease, severe vitritis frequently occurs and in this case a differential diagnosis with chronic endophthalmitis caused by Toxocara can be very difficult. Ultrasonographic study can show vitreous membranes and an elevated mass that are more common in toxocariasis. Serological studies of antibodies to both parasites can be helpful in establishing differential diagnosis.
Other forms of endophthalmitis and uveitis
Bacterial endophthalmitis is frequently related to a recent history of trauma or intraocular surgery. These acute infections produce much more intraocular inflammation than a typical toxocariasis case. Endogenous endophthalmitis is rare, but an indolent infection may be nearly impossible to distinguish from nematode endophthalmitis. In such cases, laboratory diagnostic methods may be of value in determining the etiology. Pars planitis or chronic cyclitis is a condition that typically occurs in an older age group than that in which ocular toxocariasis occurs. Hogan and coworkers59 described a case of pars planitis in which subsequent histologic examination demonstrated that the etiology was T. canis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree