11 
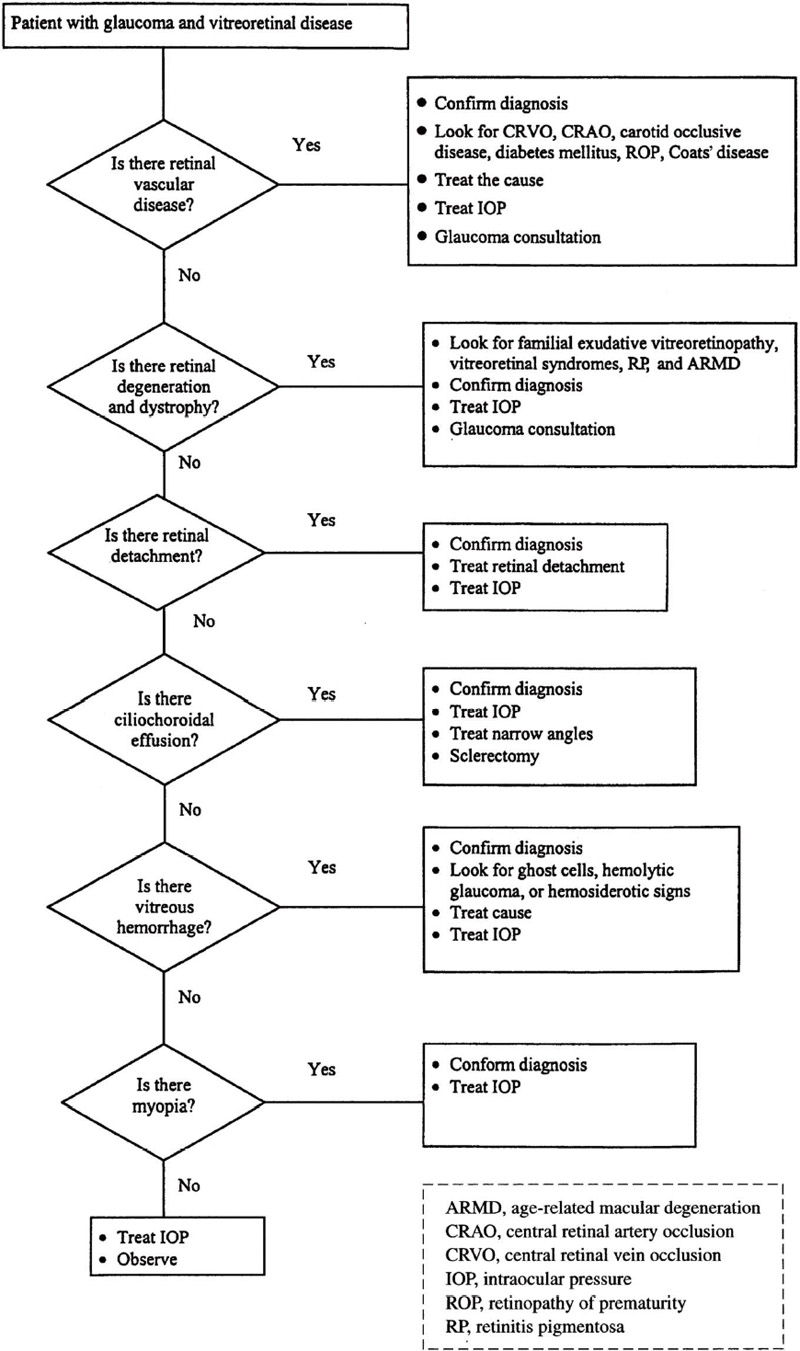
There is an intimate association between several vitreoretinal diseases and glaucoma (Table 11–1). Both primary and secondary forms of glaucoma may be encountered. The glaucomatous process may start out as an episode of elevated intraocular pressure (IOP), which may subside after a short course or may persist indefinitely. This discussion follows the outline shown in Table 11–l and the diagnostic considerations are shown in Figure 11–1.
Retinal vascular diseases |
Retinal venous occlusion |
Retinal arterial occlusion |
Carotid occlusive disease |
Diabetes mellitus |
Retinopathy of prematurity (ROP) |
Coats’ disease |
Retinal degeneration and dystrophy |
Familial exudative vitreoretinopathy |
Vitreoretinal syndromes (Stickler’s syndrome) |
Retinitis pigmentosa (RP) |
Age-related macular degeneration (ARMD) |
Others |
Rhegmatogenous retinal detachment (Schwartz syndrome) |
Ciliochoroidal effusion |
Vitreous hemorrhage |
Myopia |
Figure 11–1. Management of a patient with glaucoma and vitreoretinal disease.
RETINAL VASCULAR DISEASES
Definition
What Retinal Vascular Diseases Are Commonly Associated with Glaucoma?
Excluding postoperative glaucoma, retinal vascular diseases represent the most common causes of secondary glaucoma. Ischemic retinovascular diseases give rise to glaucoma through different mechanisms leading to angle closure from neovascularization of the anterior segment, ciliary body rotation, and contracture of retrolenticular tissue. These diseases include each of the six retinal vascular diseases listed in Table 11–1. In addition, open-angle glaucoma can arise in the absence of angle neovascularization in patients with retinopathy of prematurity, diabetes mellitus, and retinal venous occlusions (Table 11–2).
Epidemiology and Importance
How Common Is Glaucoma in Association with the Retinal Vein Occlusion?
Several studies have clearly demonstrated that the incidence of primary open-angle glaucoma (POAG) in patients with central retinal vein occlusion (CRVO) is much higher than in the normal population.1 Between 10 and 50% of patients with CRVO have been found to have an open-angle glaucoma.2,3
The frequency of secondary nonrubeotic angle-closure glaucoma after CRVO is unknown, although a few cases have been reported. Possibly this condition is rarely diagnosed rather than rare.4
Retinal venous occlusions |
Branch retinal vein occlusion (BRVO) |
Hemiretinal vein occlusion (HRVO) |
Central retinal vein occlusion (CRVO) |
Retinal arterial occlusions |
Central retinal artery occlusion (CRAO) |
Ophthalmic artery occlusion |
Carotid occlusive disease |
Proliferative diabetic retinopathy (PDR) |
Retinopathy of prematurity (ROP) |
Coats’ disease |
Other retinal and choroidal vascular diseases |
Choroidal hemorrhage |
Choroidal hemangioma |
Sturge-Weber syndrome |
Sickle cell retinopathy |
Syphilitic retinal vasculitis |
Radiation retinopathy |
The reported rate of neovascular glaucoma in CRVO ranges from 4% to approximately 50%.5 The Central Vein Occlusion Study Group found that anterior segment neovascularization developed in 16% of 714 eyes.6 The strongest predictors of development of iris or angle neovascularization were initial visual acuity and amount of nonperfusion seen on fluorescein angiography. If the initial visual acuity is worse than 20/200, prognosis for vision is poor (80% remain at that level or worse); the primary concern is the prevention of neovascular glaucoma by careful attention to the development of iris or angle neovascularization and prompt treatment if it occurs. Because more than one-third of initially perfused eyes with poor vision develop nonperfusion and iris/angle neovascularization, visual acuity is more important than the initial fluorescein angiogram in determining the prognosis and clinical management of the patient.6
How Common Is Glaucoma in Association with Central Retinal Artery Occlusion?
Development of neovascular glaucoma has been reported in approximately 15% of patients with central retinal artery occlusion (CRAO). In a prospective study of 33 consecutive patients, 18.2% developed neovascularization of the iris and 15.2% went on to develop neovascular glaucoma.7
How Common Is Glaucoma Associated with Carotid Occlusive Disease?
Carotid occlusive disease is a condition in which there is low blood flow through the ophthalmic artery due to blockage in the carotid artery. This condition accounted for 13% of all cases of neovascular glaucoma in one series.8
How Common Is Glaucoma in Association with Diabetes Mellitus?
Several large epidemiologic studies have noted an association between diabetes and open-angle glaucoma.9 The reported prevalence of rubeosis iridis in diabetic patients ranges from 0.25 to 20%.10 In diabetic patients with rubeosis iridis, the reported incidence of neovascular glaucoma ranges from 13 to 22%.10–12
How Common Is Glaucoma in Association with Retinopathy of Prematurity?
Glaucoma associated with retinopathy of prematurity (ROP) often occurs at approximately 3 to 6 months of age. However, angle-closure glaucoma may occur later in childhood, so continued observation is necessary.13,14 It is estimated that this complication may occur in as many as 30% of eyes with advanced retinopathy of prematurity.15
How Common Is Glaucoma In Association with Coats’ Disease?
In one series, glaucoma was present in 36 out of 62 eyes (58%) with Coats’ disease.16
Diagnosis and Differential Diagnosis
How Is Glaucoma Diagnosed in Patients with CRVO?
When a patient presents with acute loss of vision, shallow anterior chamber, elevated IOP, and a hemorrhagic retinal fundus picture, the diagnosis of CRVO complicated with angle-closure glaucoma should be suspected. Unilateral shallowing of the anterior chamber may be secondary to anterior lens dislocation, uveal effusion, choroidal tumors, choroidal hemorrhage, and choroidal and ciliary body detachment, and can follow panretinal photocoagulation.4 Careful examination with indirect ophthalmoscopy is helpful to make the correct diagnosis. Ultrasonography may be necessary if there is media opacity.
Neovascular glaucoma associated with a CRVO usually occurs within 3 months and has been called “90-day glaucoma.” A patient often presents with poor vision, a severely elevated IOP, an open angle with neovascularization of the iris and angle, and a hemorrhagic retinal fundus picture. It is important to perform gonioscopy in order to detect neovascularization of the angle.
How Is Glaucoma Diagnosed in Patients with CRAO?
A patient who suffers an acute central retinal or ophthalmic artery occlusion presents with sudden visual loss, an edematous white retina except in the foveal region, giving a “cherry red spot” appearance, and sluggish flow through the retinal arterioles. After a few weeks the fundus may appear normal; however, the vision remains poor. These patients may develop neovascular glaucoma.
How Is Glaucoma Diagnosed in Patients with Carotid Occlusive Disease?
Patients with carotid occlusive disease will initially present with a low IOP, chronic aqueous flare, and midperipheral dot-blot retinal hemorrhages. Some patients may progress to neovascular glaucoma from iris neovascularization due to ocular ischemia.
How Is Glaucoma Diagnosed in Patients with ROP?
The diagnosis of glaucoma in a small infant with ROP is difficult.17 When a young child with a history of prematurity presents with a shallow anterior chamber and elevated IOP, the diagnosis of glaucoma associated with ROP should be entertained. Contracture of the retrolental mass in ROP can cause forward displacement of the lens-iris diaphragm with progressive shallowing of the anterior chamber and eventual angle-closure glaucoma.14 Developmental abnormalities in the anterior chamber may contribute to glaucoma in these patients. In addition, chronic retinal detachment may lead to retinal ischemia and neovascular glaucoma.
How Is Glaucoma Diagnosed in Patients with Coats’ Disease?
Coats18 described a childhood exudative retinopathy in which the primary feature was hemorrhage and exudation from the retinal blood vessels in the subretinal space. Coats’ disease was later described as a unilateral, exudative retinopathy resulting from telangiectasia of retinal vessels.19 This disease usually affects young boys before the age of 8 and occurs unilaterally.16 Glaucoma associated with Coats’ disease was secondary to angle closure, with IOP’s ranging from 21 to 60 mm Hg.16 In one study, 11% of patients with Coats’ disease presented with painful glaucoma of sudden onset due to angle closure.16 The mechanism of angle closure in these patients appears to be due to forward displacement of the lens-iris diaphragm. Neovascular glaucoma may also develop and result in a blind, painful eye.20
Treatment and Management
How Is Glaucoma Associated with CRVO Treated?
When CRVO is associated with transient angle-closure glaucoma, the treatment should be medical because the angle returns to normal depth over a few weeks. Unlike primary angle-closure glaucoma, for which iridectomy is the definitive therapy, the treatment of choice for secondary angle-closure glaucoma is medical.4 Treatment with cycloplegics and aqueous suppressants is recommended. Miotics such as pilocarpine cause a forward shift of the iris lens diaphragm and may worsen the angle closure.
The Central Vein Study Group recommended that patients be followed monthly or bimonthly depending on their visual acuity for the first 6 months to examine the iris and angle for any development of neovascularization.6 It is important to be aware that angle neovascularization can occur without pupillary margin involvement in CRVO, implying the necessity of screening gonioscopy.21 Panretinal photocoagulation (PRP) was recommended promptly at the first sign of definite neovascularization, but not prophylactically.6 Prophylactic PRP did not totally prevent anterior segment neovascularization. Prompt regression of anterior segment neovascularization in response to PRP was more likely to occur in eyes that have not been treated previously.22 Because of the widespread areas of nonperfusion, posterior segment neovascularization rarely occurs with ischemic CRVO. In 10 of 117 eyes in patients with iris/angle neovascularization, the new vessels failed to regress in response to photocoagulation, and neovascular glaucoma developed in the eye that could not be controlled medically.6 The treatment of neovascular glaucoma is discussed in Chapter 14.
How Is Glaucoma in Relation to Retinal Arterial Occlusion Managed?
Although PRP has been proposed as a treatment for prevention of neovascular glaucoma in patients with CRAO, in one study, five of six patients with neovascularization of the iris went on to develop neovascular glaucoma despite PRP.7 However, ablation of the ischemic retina, which may be the source of angiogenic factors, must be undertaken. If the fundus view is adequate, then thorough PRP should be undertaken promptly. Cryotherapy or transscleral diode laser is useful if the view is poor.
How Is Glaucoma in Relation to Carotid Occlusive Disease Managed?
Carotid occlusive disease may ultimately result in neovascular glaucoma. The role of carotid endarterectomy in preventing neovascular glaucoma is unclear. There are cases of resolution of ocular ischemic findings, but others could not document improvement or stabilization of vision.23
How Is Glaucoma in Association with Diabetes Managed?
Management of open-angle glaucoma in association with diabetes is similar to its management in nondiabetic patients. In is prudent to avoid epinephrine and prostaglandin agents in patients with diabetic macular edema.24–28 One should avoid using oral glycerin to acutely reduce IOP in diabetic patients. Oral isosorbide does not raise blood sugar and is preferable. Advanced diabetic retinopathy with widespread retinal ischemia may result in neovascular glaucoma. The ischemic retina, which is presumed to be producing angiogenic factors, must be treated for anterior segment neovascularization to regress. PRP is the easiest and least invasive method; however, this method is not possible if the view to the posterior segment is obscured by cataract or vitreous hemorrhage. Retinal cryotherapy, which does not require a posterior pole view, may be performed as an office procedure. Any retinal detachment that is present must be surgically repaired followed by laser photocoagulation.
How Is Glaucoma in Relation to ROP Managed?
Management of glaucoma in cases of ROP is difficult.17 Lens extraction, alone or combined with vitrectomy, has proven helpful in patients with angle closure.13,29 Cycloplegics may initially be helpful in some cases of angle closure related to pupillary block; however, most cases require lensectomy and vitrectomy for definitive cure.30 When a chronic retinal detachment is present, neovascular glaucoma often complicates ROP.31 Treatment may include seton devices and cyclophotocoagulation; however, enucleation may be necessary if the eye becomes blind and uncomfortable.
How Is Glaucoma Associated with Coats’ Disease Managed?
Multiple sessions of photocoagulation or cryotherapy of the peripheral retinal telangiectasias is the preferred treatment of eyes with Coats’ disease.32 Angle-closure glaucoma associated with Coats’ disease is extremely difficult to treat. Success has been reported using vitreoretinal techniques in order to reattach the retina, destroy the extensive retinal telangiectasias, and drain the subretinal exudates.20,33,34
Future Considerations
What Future Therapies May Be Available to Treat Glaucoma Associated with the Retinal Vascular Disorders?
Antiangiogenic drug delivery may be used during or independently of surgery to reduce neovascularization. Endocyclophotocoagulation has become a useful adjunct in treatment of chronic glaucoma.
RETINAL DEGENERATION AND DYSTROPHY
Definition
How Is Glaucoma Associated with Retinal Degenerations and Dystrophies Defined?
Familial exudative vitreoretinopathy, vitreoretinal syndromes such as Stickler’s syndrome, retinitis pigmentosa (RP), and age-related macular degeneration (ARMD) are associated with glaucoma (Table 11–1).
Epidemiology and Importance
How Common Is Glaucoma Associated with Retinal Degenerations and Dystrophies?
The incidence of angle-closure glaucoma with ARMD (secondary to massive substantial hemmorhage) is rare. The prevalence of POAG in patients with RP has been reported to range from 2 to 12%,35 whereas angle-closure glaucoma has been reported to be 1.03% in patients with RP over age 40.36 In one study of 39 patients with Stickler’s syndrome, 10% had ocular hypertension.37
Diagnosis and Differential Diagnosis
How Is Glaucoma in Association with Age-Related Macular Degeneration Diagnosed?
Age-related macular degeneration (ARMD) is a common disorder characterized by decreased visual acuity with drusen in the macula occurring in elderly patients. Late forms of this disorder affect approximately 7% of individuals over the age of 75.38 Patients with exudation may experience massive vitreous, subretinal, or suprachoroidal hemorrhage. Rapid expansion of the subretinal or suprachoroidal space leads to an increase in the posterior segment volume and anterior displacement and rotation of the lens-iris diaphragm, which may result in angle-closure glaucoma. This diagnosis should be suspected in the elderly patient presenting with angle-closure glaucoma and forward displacement of the lens-iris diaphragm without the anticipated shallow chamber in the fellow eye. Ghost cell glaucoma has also been reported following vitreous hemorrhage from ARMD.39
How Is Glaucoma in Relation to Retinitis Pigmentosa Diagnosed?
Retinitis pigmentosa (RP) is an inherited retinal degeneration characterized by nyctalopia, attenuated retinal arterioles, and presence of pigmentary changes in the retinal periphery. The diagnosis of glaucoma in the presence of RP can be challenging because both diseases may give a similar field defect. Because most forms of RP are rod-cone degenerations, a generalized constriction of the visual field is seen, which makes detecting glaucomatous scotomas difficult. In addition, the waxy pallor observed in RP can make evaluation of the optic nerve head difficult.
How Is Glaucoma in Relation to Stickler’s Syndrome Diagnosed?
Stickler’s syndrome, or hereditary arthro-ophthalmopathy, is characterized by arthritis, cleft palate, midfacial hypoplasia, and ocular defects including radial perivascular lattice degeneration, vitreous degeneration, glaucoma, cataracts, and frequent retinal detachments.40 Phelps41 noted that several of his patients with Stickler’s syndrome had mild to moderate elevation of IOP. The anterior chambers were open without obvious structural malformation. The diagnosis of glaucoma is difficult because lens opacities and high myopia impair the view of the optic disc. In addition, the discs are often tilted, and cupping invariably is shallow. Areas of retinal degeneration may cause visual defects similar to those of glaucoma, and IOP may be the only reliable criterion on which to base treatment.41
Treatment and Management
How Is Glaucoma Associated with ARMD Treated?
Angle-closure glaucoma secondary to exudative complications of ARMD may be treated medically with cycloplegia and aqueous suppressants. Cycloplegia may reverse angle-closure glaucoma in this and other posterior segment causes of angle closure, whereas miotics may exacerbate the condition.
How Is Glaucoma Associated with RP Treated?
There is a tendency to treat patients with RP earlier with aqueous suppressants than might otherwise be done in the absence of a definitive diagnosis of glaucoma because of the difficulty in interpreting the visual fields and the optic nerve heads. Because posterior subcapsular cataracts are common in patients with RP, miotic therapy may lead to decreased visual acuity.
How Is Glaucoma Associated with Stickler’s Syndrome Treated?
The treatment of glaucoma in Stickler’s syndrome is mainly medical. The high IOPs respond well to antiglaucoma medications. Miotics should be avoided, as they may reduce vision in the presence of an axial lens opacity or may induce retinal detachment in these highly susceptible patients.42
Future Considerations
What Future Therapies May Be Available for Glaucoma Associated with Retinal Degenerations and Dystrophies?
Gene therapy may alter the course of inherited retinal degenerations and dystrophies. Genetic testing may also help diagnose these conditions earlier.
RHEGMATOGENOUS RETINAL DETACHMENT (SCHWARTZ SYNDROME)
Definition
How Is Glaucoma Associated with Rhegmatogenous Retinal Detachment Defined?
Most patients with a retinal detachment have decreased IOP in the affected eye as compared to their fellow eye. This phenomenon is attributed to increased uveoscleral outflow associated with a retinal break. The magnitude of the decrease is directly proportional to the size of the detachment.43 Schwartz syndrome refers to unilateral elevation of the IOP associated with retinal detachment.44
What Is the Mechanism of Glaucoma in Schwartz Syndrome?
A retinal break allows communication between the subretinal space and the anterior chamber. Photoreceptor outer segments with few inflammatory cells obstruct the trabecular meshwork to cause open-angle glaucoma.45
Epidemiology and Importance
How Common Is Glaucoma Associated with Retinal Detachment?
Phelps and Burton46 examined 817 cases of retinal detachment and found that open-angle glaucoma was present in 4%, and an additional 6.5% had elevated IOP without glaucomatous damage. They also discovered that that 2.1% of 817 patients with retinal detachment had features consistent with Schwartz syndrome.46
Diagnosis and Differential Diagnosis
How Is Schwartz Syndrome Diagnosed?
Features of Schwartz syndrome include unilateral elevation of IOP, rhegmatogenous retinal detachment, iridocyclitis, and normalization of the IOP following retinal detachment repair.47 The probable etiology of this condition is obstruction of the trabecular meshwork with photoreceptor outer segments.45
Treatment and Management
How Is Glaucoma Associated with Schwartz Syndrome Treated?
The management of this condition is aqueous suppression and repair of the retinal detachment. In the majority of cases, glaucoma medications may be discontinued within a few weeks of reattachment of the retina.48
Future Considerations
How Have Recent Advancements Contributed to the Treatment of Schwartz Syndrome?
With the advancement of surgical techniques for retinal detachment repair, the reattachment rates for chronic retinal detachments have improved dramatically. In addition, with the advent of vitrectomy for retinal detachment repair, the majority of diffused photoreceptor elements may be removed from the eye at the time of the operation.
CILIOCHOROIDAL EFFUSION (UVEAL EFFUSION SYNDROME)
Definition
How Is Glaucoma Associated with Ciliochoroidal Effusion Defined?
Angle-closure glaucoma is a known complication of ciliochoroidal effusion. These eyes are nanophthalmic and hyperopic due to a short axial length. The sclera in these eyes are thicker than normal and contain unusually disordered collagen fibrils.49 Uveal effusion may result from either a reduced scleral permeability to proteins or vortex vein compression. Uveal effusion causes forward rotation of the lens-iris diaphragm, inducing angle-closure glaucoma.
Epidemiology and Importance
How Common Is Glaucoma Associated with Uveal Effusion Syndrome?
This syndrome usually occurs in patients with small, hyperopic eyes in the fourth to sixth decades of life. This is a rare condition.
Diagnosis and Differential Diagnosis of the Problem
How Is Glaucoma in Association with Uveal Effusion Syndrome Diagnosed?
In the setting of an elevated IOP, shallow anterior chamber, and narrow angle in a small, hyperopic eye, one should consider angle closure due to ciliochoroidal effusion. These eyes are highly hyperopic due to their short axial length. One should perform a fundus exam to look for a choroidal detachment. An exudative retinal detachment may be observed.
Treatment and Management
The management of glaucoma with uveal effusion syndrome is complex. Angle-closure glaucoma may be treated topically with cycloplegics, aqueous suppressants, and corticosteroids.50 Miotics may improve some cases but worsen others.51 Laser iridotomy and peripheral iridoplasty may be tried to eliminate any component of pupillary block. The choroidal effusion is treated with drainage sclerotomies in abnormally thickened sclera.52,53
Future Considerations
Future studies may better define the surgical management of these patients. Better diagnostic instruments such as biomicroscopic ultrasonography may help when the diagnosis is not clear.
VITREOUS HEMORRHAGE
Definition
How Is Glaucoma Associated with Vitreous Hemorrhage Defined?
Vitreous hemorrhage may cause glaucoma by three mechanisms: ghost cell glaucoma, hemolytic glaucoma, and hemosiderotic glaucoma.
What Is Ghost Cell Glaucoma?
Campbell et al54 described a form of glaucoma in which degenerated red blood cells (ghost cells) develop in the vitreous cavity and subsequently enter the anterior chamber where they temporarily obstruct aqueous outflow.
What Is the Mechanism of Ghost Cell Glaucoma?
When red blood cells are present in the vitreous cavity by various mechanisms (trauma, surgery, diabetes, etc.) they degenerate into ghost cells in a matter of weeks. Ghost cells are tan- or khaki-colored, spherical, less pliable structures that have thin walls and appear hollow except for clumps of denatured hemoglobin, called Heinz bodies. These ghost cells do not readily pass through the trabecular meshwork. Thus, once they gain access into the anterior chamber via a disrupted anterior hyaloid face, they accumulate in the trabecular meshwork and cause elevation of IOP.
What Is Hemolytic Glaucoma?
Hemolytic glaucoma is a condition in which the trabecular meshwork is blocked by hemoglobin-laden macrophages.
What Is Hemosiderotic Glaucoma?
Hemosiderotic glaucoma is a rare type of glaucoma associated with a longstanding vitreous hemorrhage that may lead to trabecular meshwork damage secondary to iron accumulation.
What Is the Etiology of Vitreous Hemorrhage?
The most common causes of spontaneous vitreous hemorrhage are diabetic retinopathy, and retinal break with or without detachment, and retinal vein occlusion.55,56 Trauma and surgery are commonly associated with vitreous hemorrhage.
Epidemiology and Importance
What Is the Incidence of Glaucoma Associated with Vitreous Hemorrhage?
The incidence of spontaneous vitreous hemorrhage is approximately 7 cases per 100,000 population.57 The incidence of glaucoma associated with vitreous hemorrhage is not known.
Diagnosis and Differential Diagnosis
How Is Ghost Cell Glaucoma Diagnosed?
Patients with ghost cell glaucoma present with pain, perilimbal injection, and an elevated pressure with a history of a vitreous hemorrhage. There are ghost cells visible in the anterior chamber. Ghost cells are spherical, khaki-colored cell walls of the red blood cell. They are produced 1 to 2 weeks following a vitreous hemorrhage when the red blood cells degenerate. Ghost cells clog the trabecular meshwork because they are less rigid than the normal red blood cell. An anterior chamber paracentesis may be performed to examine the fluid for ghost cells if the diagnosis is uncertain.
How Is Hemolytic Glaucoma Diagnosed?
Hemolytic glaucoma is a condition in which the trabecular meshwork is blocked by hemoglobin-laden macrophages. Because the breakdown of the red blood cell results in both free hemoglobin and ghost cells, it is likely that ghost cell glaucoma coexists with hemolytic glaucoma.
How Is Hemosiderotic Glaucoma Diagnosed?
Hemosiderotic glaucoma is a rare type of iron-induced glaucoma associated with a long-standing vitreous hemorrhage. Iron released from the hemoglobin of the red blood cell is toxic to ocular structures, which may result in cataract, iris discoloration, and reduced retinal function. Damage to the endothelial cells of the trabecular meshwork results in glaucoma. Most cases of hemosiderotic glaucoma result from iron-containing foreign bodies. The diagnosis is difficult to make clinically.
Treatment and Management
How Is Ghost Cell Glaucoma Treated?
The first step in treating ghost cell glaucoma is to medically reduce the pressure using aqueous suppressants. Topical steroid treatment may reduce inflammation caused by the elevated IOP; however, some authors feel that ghost cell glaucoma is not an inflammatory disease because the primary problem is obstruction of the trabeculum with ghost cells. If medical therapy is not successful, then a thorough vitrectomy is indicated to remove all the ghost cells as well as the remaining vitreous hemorrhage. During vitrectomy, it is important to trim the inferior vitreous base as much as possible, as this location harbors many red blood cells that may continue to produce ghost cells even after vitrectomy.
How Is Hemolytic and Hemosiderotic Glaucoma Managed?
The treatment of hemolytic glaucoma follows the standard medical therapy of glaucoma. If this therapy will be unsuccessful, a paracentesis with irrigation of the anterior chamber may be required.58
Treatment of hemosiderotic glaucoma follows the standard therapy of glaucoma as well. Filtering surgery may be required, as permanent damage to the trabecular meshwork may occur.
Future Considerations
What Future Therapies Are there for Glaucoma Associated with Vitreous Hemorrhage?
Experimental nonsurgical treatment options involve improvement of physiologic clearance mechanisms to accelerate fibrinolysis, liquefaction, hemolysis, and phagocytosis.57
What Are the Future Concerns About These New Therapies?
There may be an increased incidence of ghost cell glaucoma with the advent of enzymatic vitreolysis of vitreous hemorrhage.
MYOPIA
Definition
How Is Glaucoma Associated with Myopia Defined?
The relative risk of open-angle glaucoma was found to increase sequentially as the level of refractive error studied was shifted away from hyperopia toward higher levels of myopia, becoming three times greater for high myopia as compared to hyperopia.59 In addition, there is an increased frequency of myopia among patients who have POAG, ocular hypertension, and low-tension glaucoma.60
Are Myopes More Susceptible to Glaucoma?
Perkins and Phelps60 suggested that myopic eyes are more susceptible to the effects of raised IOP than are nonmyopic eyes and that myopes with ocular hypertension have a particularly high risk of the development of field defects. In the Blue Mountains Eye Study, glaucoma was present in 4.2% of eyes with low myopia and 4.4% of eyes with moderate to high myopia compared to 1.5% of eyes without myopia.61 This study has confirmed a strong relationship between myopia and glaucoma in that myopic subjects had a twofold to threefold increased risk of glaucoma compared to that of nonmyopic subjects. The risk was independent of other glaucoma risk factors and IOP.61
What Is the Mechanism of Glaucoma in Myopia?
Nesterov and Katnelson62 postulated that in myopic eyes, the ciliary body is in a relatively posterior position in relation to the canal of Schlemm so that it has less mechanical advantage in widening the spaces of the trabecular meshwork during accommodation. Tomlinson and Phillips63 found a statistically significant positive correlation between ocular tension and axial length. They proposed two possible explanations: Either the raised tension is an important factor in producing a large axial length, or the raised tension is produced by a large axial length. Although unlikely in the adult eye, the raised IOP in glaucoma may stretch the globe and cause myopia, especially in congenital and juvenile glaucoma.
The myopic eye tends to have a larger optic cup with a greater cup-to-disc ratio than emmetropic eyes, and it is possible that these anatomic features predispose the disc to pathologic changes from raised IOP.60 Fluorescein angiographic studies have suggested a reduced choroidal blood flow in myopia,64 and the amplitude of the ocular pulse is lower in myopes than in emmetropes or hypermetropes.65,66 The circulation to the optic disc may also be more susceptible to raised IOP.
Epidemiology and Importance
How Common Is Glaucoma in Association with Myopia?
In a population based survey of 2,403 individuals in Israel, increased IOP was observed in high myopes compared to emmetropes and hypermetropes.67 In one study, compared with the normal population, the glaucoma population contained about half as many hypermetropic eyes but four times as many myopic eyes.60 In one series of 68 patients between the ages of 10 and 35 years, myopia was present in 59% of the ocular hypertensives and 73% of the POAG patients.68 In a population-based study of an Australian white community, the overall glaucoma prevalence was reported to be 3.0%.69
Diagnosis and Differential Diagnosis
How Is Myopia Defined?
Myopia, or nearsightedness, is generally classified as axial or refractive. Axial myopia occurs because the eye has a longer axial length, whereas refractive myopia occurs because of the higher refractive properties of the cornea and lens. The vast majority of myopes are axial in nature. In the general population, the prevalence of myopia is approximately 25%.68
What Is the Differential Diagnosis for Glaucoma in Association with Myopia?
Friedman70 showed that an eye of large volume with thin scleral walls will experience greater stress on its scleral walls than a normal eye at the same IOP. During ocular tension measurements, the plunger reading would seemingly indicate a lower IOP in the myopic eye and a higher IOP in the hyperopic eye than actually exists.70 The optic disc in myopia is larger with a larger cup-to-disc ratio, making evaluation of the optic disc difficult when attempting to make a diagnosis of glaucoma. In addition, tilted discs, which are more common in myopia, may give visual field defects that mimic glaucomatous scotomas. Myopic retinal degeneration is common in patients with high myopia. Myopic retinal degeneration may produce visual field defects that mimic glaucoma.
Treatment and Management
How Is Glaucoma in Association with Myopia Treated?
Because the optic nerve head in myopic eyes may be structurally more susceptible to the effects of raised IOP, appropriate treatment should be initiated once the diagnosis is made.60,68,71 Similar to patients with POAG, medical therapy should be instituted first and should consist of topical aqueous suppressants and oral carbonic anhydrase inhibitors. Miotics should be avoided due to the higher risk of retinal detachment in these eyes. If the pressure continues to be elevated despite maximal medical therapy, then argon laser trabeculoplasty or filtering surgery should be considered. Young myopic males are also at risk for pigmentary glaucoma, which may respond to peripheral iridotomy.
Because of many confounding variables in making a diagnosis of glaucoma in patients with myopia, factors such as optic disc appearance, visual field, and IOP should be carefully monitored for progression.
Future Considerations
What Are the Future Considerations in Glaucoma in Association with Myopia?
There is ongoing research in finding a genetic link between glaucoma and myopia, which may help us better understand the relationship between these two common disorders.
Acknowledgment
This work was supported in part by grants from Research to Prevent Blindness.
References
18. Coats G: Forms of retinal disease with massive exudation. R Lond Hosp Rep 1907;17:440–525.
19. Reese AB: Telangiectasia of the retina and Coat’s disease. Am J Ophthalmol 1956;42:1–8.
21. Browning DJ, Scott AQ, Peterson CB, Warnock J, Zhang Z: The risk of missing angle neovascularization by omitting screening gonioscopy in acute central retinal vein occlusion. Ophthalmology 1998;105(5):776–784.
24. Classe JG: Epinephrine maculopathy. J Am Optom Assoc 1980;51(12):1091–1093.
37. Spallone A: Stickler’s syndrome: a study of 12 families. Br J Ophthalmol 1987;71(7):504–509.
46. Phelps CD, Burton TC: Glaucoma and retinal detachment. Arch Ophthalmol 1977;95(3): 418–422.
53. Johnson MW, Gass JD: Surgical management of the idiopathic uveal effusion syndrome. Ophthalmology 1990;97(6):778–785.
56. Burner RW, McPherson AR: Spontaneous vitreous hemorrhage. Ann Ophthalmol 1982;14(3): 268–270.
57. Spraul CW, Grossniklaus HE: Vitreous hemorrhage. Surv Ophthalmol 1997;42(l):3–39.
62. Nesterov AB A, Katnelson L: Intraocular pressure. Moscow: MIR, 1978.
66. Perkins ES: The ocular pulse. Curr Eye Res 1981;l(l):19–23.
71. Chihara E, Liu X, Dong J, et al: Severe myopia as a risk factor for progressive visual field loss in primary open-angle glaucoma. Ophthalmologica 1997;211(2):66–71.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree