8 
Definition
How Is Glaucoma Associated with Inflammation Defined?
Inflammatory ocular conditions like keratitis, episcleritis, scleritis, and uveitis may cause elevated intraocular pressure (IOP) by compromising the outflow of aqueous humor. Whether or not increased secretion of aqueous plays any role is still controversial.1,2 The anterior chamber angle may be open or occluded. The IOP may be elevated acutely and/or chronically. This chapter discusses each inflammatory condition separately.
GLAUCOMA ASSOCIATED WITH KERATITIS
Definition
How Is Glaucoma Associated with Keratitis Defined?
IOP elevation may be encountered in any active corneal inflammatory condition.3 If the elevated IOP is causing optic nerve damage and visual field loss, then the diagnosis of glaucoma secondary to corneal inflammation can be made. This implies that the elevated IOP does not necessarily lead to optic nerve damage, that is, glaucoma. This further implies that elevated IOP associated with corneal inflammation does not always need to be lowered with antiglaucoma agents. After treatment and resolution of the underlying inflammatory condition, the IOP may return to the normal level without having caused any damage to the optic nerve.
What Is the Cause of the Elevated IOP in Keratitis?
Anterior uveitis often accompanies keratitis. Inflammatory cells may occlude the trabecular meshwork and thus decrease aqueous outflow. Posterior synechiae may develop as a result of inflammation and cause pupillary block. Peripheral anterior synechiae may cause IOP elevation, not only acutely but also chronically, even after the inflammation subsides.3
After chemical burns, the IOP may rise rapidly initially secondary to shrinkage of the outer coats of the eye, followed by a second slow phase of IOP elevation probably mediated by an intraocular release of prostaglandins.4,5 IOP elevation usually occurs if the keratitis involves the corneal stroma, such as infectious keratitis secondary to herpes simplex,6 bacterial and fungal keratitis, and an immune response to Treponema pallidum.6–10 A shallow anterior chamber may develop in keratomycotic malignant glaucoma.11 Fungi can also penetrate the cornea and directly invade the trabecular meshwork.12
Direct involvement of the trabecular meshwork by the underlying inflammatory disease process is the main cause of elevation of the IOP.8,13 Increased corneal thickness secondary to corneal edema may lead to closure of the anterior chamber angle. Formation of anterior synechiae with secondary angle closure may also be encountered. Mechanisms of IOP elevation can also include secondary iridoschisis,14 keratouveitis,6 and open-angle glaucoma superimposed on old inflammatory changes, involving endothelialization and glassy membrane in the angle, which are refractory to medical treatment. This condition usually requires filtering surgery.8 Reversible angle-closure glaucoma associated with anatomically small anterior segments responds well to iridectomy.8,10 Sundmacher and Neumann-Haefelin15 found that secondary glaucoma was present in all herpes simplex keratitis patients with corneal endothelial disease and/or anterior uveitis who had herpes simplex virus isolated from the aqueous. Similarly, adenovirus type 10 has been associated with keratoconjunctivitis, pharyngitis, and transient increase in IOP.16
Epidemiology and Importance
How Often Is Keratitis Associated with Increased IOP?
Keratitis by itself is not commonly associated with elevated IOP. However, when herpes simplex keratitis was associated with uveitis, almost a third of the patients developed IOP elevation and 10% had glaucomatous damage.6
About 40% of patients with herpes zoster ophthalmicus can have uveitis, and more than 10% of those patients may have associated glaucoma.17 Another study showed that every third patient (five out of 14) with herpes zoster keratouveitis had IOP elevation.18 Combination of keratitis and IOP elevation may be seen in patients with acute primary angle-closure glaucoma. These patients may develop secondary keratouveitis and keratic precipitates.19 Possible mechanism of the inflammation is likely profound endothelial damage from elevated IOP. Hypopyon is also likely to be present in these eyes,19–21 secondary to ischemic iris necrosis.
Diagnosis and Differential Diagnosis
How Is Glaucoma Associated with Keratitis Diagnosed?
An accurate history is important to find the etiology and to differentiate between an acute and a chronic condition. Slit-lamp examination facilitates evaluation of possible corneal infiltration and anterior chamber reaction. Subsequent use of fluorescein helps in evaluating the corneal surface. Gonioscopy should be performed, if possible.
What Is the Differential Diagnosis of Glaucoma Associated with Keratitis?
In a patient presenting with keratitis and IOP elevation, the following conditions should be differentiated: corneal abrasion,22 recurrent erosion syndrome,23 herpes simplex keratitis, herpes zoster keratitis, and angle-closure glaucoma causing corneal edema. Leprosy may need to be excluded when a patient presents with glaucoma, keratitis, and bilateral uveitis.24
Treatment and Management
How Is Glaucoma Associated with Keratitis Managed?
Treatment of keratitis associated with increased IOP is directed against the underlying etiology and includes administration of aqueous suppressants. One of the suggested therapeutic regimens for herpes simplex keratitis associated with glaucoma consists of antivirals, steroids, cycloplegics, and aqueous suppressants.15 The treatment can be difficult, especially if angle closure with synechiae is present. In these cases, filtering surgery8 or implantation of a drainage device might be necessary.
Future Considerations
In stromal keratitis induced by herpes simplex virus (HSV), the immune response contributes to corneal scarring and neovascularization. To analyze the efficacy of topically applied cyclosporin A (CsA) in patients with HSV keratitis, Heiligenhaus and Steuhl25 performed a prospective study in 18 patients with HSV stromal keratitis. The authors treated eyes with CsA eyedrops and acyclovir ointment. Keratitis resolved with CsA treatment in 10 of 14 patients with nonnecrotizing keratitis and in two of four with necrotizing keratitis. One-third of the patients treated with CsA developed persistent or progressive inflammation and required combined CsA and corticosteroids. The only complication from the use of CsA was toxic epitheliopathy. HSV stromal keratitis can be treated successfully with CsA eyedrops, especially in nonnecrotizing keratitis. In patients with steroid-induced glaucoma, herpetic corneal ulcers may be primary candidates for use of the CsA.25 Further studies will evaluate the use of topical CsA in the treatment of herpetic keratitis associated with glaucoma.
GLAUCOMA ASSOCIATED WITH EPISCLERITIS
Definition
How Is Glaucoma Associated with Episcleritis Defined?
Intraocular pressure elevation is not seen frequently with episcleritis. Similar to all inflammatory conditions, elevated IOP does not always cause optic nerve damage and visual field loss, and does not always need to be lowered with IOP-lowering agents. After observation and/or treatment, the underlying inflammatory condition usually resolves, and the IOP typically returns to normal level without damaging the optic nerve.
Epidemiology and Importance
How Often Does Episcleritis Present with Elevated IOP?
Episcleritis is rarely associated with elevated IOP.26,27 Few cases have been reported in the literature.28,29 Glaucoma was found in 9% (9 out of 100) of the eyes with episcleritis in a recent retrospective study of patients with episcleritis, and none of the patients had a history of glaucoma.30 Four of those nine patients were using topical steroids for recurrent episcleritis of the initial exam. Glaucoma was more common in patients with recurrent episcleritis but was infrequent with bilateral involvement.30 Possible elevation of the episcleral venous pressure does not seem to influence the IOP, but this is still controversial.26,28,31
Diagnosis and Differential Diagnosis
How Does Episcleritis Present?
Episcleritis is a benign condition that presents with acute onset of redness, mild discomfort, and infrequently mild pain. It is often recurrent and is bilateral in every third patient with episcleritis. It occurs in young adults32 and appears to be twice as common in females.30 Visual acuity is usually not affected; the redness is commonly limited to a scleral sector, but occasionally can be diffuse. Conjunctival and episcleral vessels are engorged and can be moved over the sclera. The cornea is usually clear, and the anterior chamber is quiet.32,33 Episcleritis is mostly idiopathic; however, an associated systemic disease is found in a third of the cases,26,30,32 the most common being atopy.30 Rosacea can also cause episcleritis.
What Is the Differential Diagnosis of Glaucoma Associated with Episcleritis?
Inflammation of the sclera and episclera may be associated with systemic diseases. Sainz et al29 evaluated ocular complications and specific systemic disease association in 266 patients (358 eyes) with different types of scleritis and episcleritis; 37% of patients with scleritis developed decrease in vision, 42% had an associated anterior uveitis, 14% developed peripheral ulcerative keratitis, 13% developed glaucoma, 17% formed cataracts, and 6% had fundus abnormalities. More than half of the patients (57%) had an associated disease. These findings were more typical for necrotizing scleritis. In contrast, only 2% of patients with simple and nodular episcleritis developed some degree of visual loss, 11% had an associated anterior uveitis, 4% developed glaucoma, and 2% formed cataracts. Every third patient had a specific disease association.29
Treatment and Management
How Is Episcleritis Treated?
Treatment of episcleritis is directed at the underlying cause if one is identified (i.e., elimination of irritants and application of mast cell stabilizers for atopy, doxycycline for rosacea).30 Because episcleritis is a self-limited condition, nonspecific treatment is not necessary if the patient is asymptomatic. Idiopathic symptomatic episcleritis can be treated with cold compresses, iced artificial tears,30 and topical vasoconstrictors. Treatment of episcleritis with topical flurbiprofen has not been shown to be more effective than placebo.34 Treatment of recurrent episcleritis with low-dose oral nonsteroidal antiinflammatory drugs (NSAIDs) is effective30 and has not been associated with IOP elevation.
Topical treatment with the relatively new NSAID aminothiazole hydrochloride 0.l%35,36 was effective in reducing inflammation in mild episcleritis within the first week of administration of the drug. It was not effective against severe episcleritis. There Was no effect on IOP.37
How Is Glaucoma Associated with Episcleritis Managed?
There are different mechanisms for elevated IOP in a patient with episcleritis. Each condition is managed accordingly.
How Is Steroid-Induced Elevation of IOP Managed?
Corticosteroids are always effective in reducing the symptoms of episcleritis,34,38,39 but IOP should be monitored weekly and therapy with steroids should not be continued longer than a few weeks30,32 (see also Chapter 15).
How Is Elevated IOP with Open Anterior Chamber Angle Managed?
There are almost no reported causes in the literature of an association of episcleritis with IOP elevation. Watson40 did not find any evidence of glaucoma in 192 eyes of 117 patients with episcleritis. Harbin and Pollack28 described two patients with episcleritis and IOP elevation. One of the four eyes had anterior uveitis. The authors assume that in the other 3 eyes the inflammation was limited to the angle structures, especially the trabecular meshwork, causing impairment of the aqueous outflow without involving the iris and ciliary body vasculature. The low facility of outflow implies that the increased episcleral venous pressure was not the mechanism of IOP elevation. These patients respond to aqueous suppressants and treatment with topical steroids.28
How Is Elevated IOP Associated with Angle Closure Managed?
Acute angle closure is a rare complication of episcleritis. These eyes should respond to standard therapy for acute angle closure with neodymium:yttrium-aluminum-garnet (Nd:YAG) laser iridotomy or peripheral iridectomy41 (see also Chapter 5).
How Is Preexisting Open-Angle Glaucoma Associated with Episcleritis Managed?
Patients with preexisting open-angle glaucoma have a higher chance of being steroid responders and should therefore be monitored closely when treated with topical steroids.42 Among comparable age groups, the steroid-induced IOP elevation was always greater in glaucomatous than in normal eyes.43
Future Considerations
The development of soft steroids that will treat inflammation without elevation of IOP will greatly aid in the management of episcleritis. Recent arrivals in this group are loteprednol and rimexolone.
GLAUCOMA ASSOCIATED WITH SCLERITIS
Definition
How Is Glaucoma Associated with Scleritis Defined?
As in all inflammatory conditions associated with IOP elevation, glaucoma associated with scleritis must meet the criteria of glaucomatous damage: glaucomatous optic nerve, visual field loss, and IOP elevation developing secondary to scleritis.
How Does Scleritis Present?
Scleritis is a serious and damaging ocular condition. It usually presents with severe pain, radiating to the orbit, forehead, cheekbone, and teeth. The patient complains of photophobia and insidious decrease of vision. The eye is red due to injection of scleral, episcleral, and conjunctival vessels. The sclera may be affected only in one sector or diffusely. As opposed to episcleral vessels, the affected scleral vessels cannot be moved with a cotton swab. The scleritis can be recurrent and is associated with a systemic disease in approximately 50% of cases. It can be divided into anterior and posterior scleritis.32,44
Epidemiology and Importance
How Often Is Scleritis Associated with Elevated IOP?
Most studies have found that 12% to 13% of patients with anterior scleritis present with glaucoma;29,32,41 20% of those eyes may also have associated keratitis.26 Elevated IOP was detected in 18.7% of eyes with rheumatoid scleritis.27 However, 50% of eyes enucleated for scleritis also had glaucomatous optic neuropathy.41 Episcleritis normally does not progress to scleritis, except in the case of herpes zoster, which may start as an episcleritis and reappear 3 months later as a scleritis at the same site.26
Wilhelmus et al45 reviewed 92 enucleated eyes with histopathologic evidence of scleral inflammation. They also examined 114 eyes from 81 patients with scleritis. Almost half of the enucleated eyes and every fifth eye in examined patients showed evidence of increased IOP, the most common causes of which were damage to the trabecular meshwork from iridocyclitis, overlying corneoscleral inflammation, or peripheral anterior synechiae. Other causes included topical corticosteroid use; angle neovascularization in enucleated, but not in clinically observed, eyes; and posterior scleritis with secondary angle closure.
Sainz and co-workers46 found that in 42% of patients with scleritis, inflammation may extend to the anterior uveal tract and cause anterior uveitis, with consequent development of glaucoma in 19% of eyes in that group.46 Posterior scleritis is typically not associated with glaucoma, but angle closure secondary to choroidal effusion has been reported in the literature.45–51 In 5% of patients with scleritis, the elevated IOP can be related to the administration of corticosteroids used to treat the underlying condition.
Why Is the Intraocular Pressure Elevated in Scleritis?
There are several mechanisms for raised pressure in patients with scleritis. These patients may present with any of the following four clinical features:
ELEVATION OF IOP WITH OPEN ANTERIOR CHAMBER ANGLE
The IOP may be elevated in the presence of an open anterior chamber angle. This may be caused by (1) increased resistance to outflow from inflammation in the trabecular meshwork;45 (2) preexistent abnormalities in outflow pathways that are impaired by perilimbal inflammation and edema; (3) abnormal steroid response;45,52 and (4) elevated episcleral venous pressure.53
Acute uveitis causing damage to the trabecular meshwork and occlusion of the trabecular meshwork by inflammatory cells was the main mechanism of IOP elevation in pathologic studies of the eyes with glaucoma and scleritis.45 Discontinuation of steroids decreased IOP in the eyes with induced ocular hypertension due to abnormal steroid response.
Wilhelmus and associates45 reported that 4 out of 92 eyes enucleated for scleritis had an open angle and did not demonstrate any trabecular meshwork abnormalities. They were thought to have primary open-angle glaucoma. All these eyes had lymphocytic cuffing around the intrascleral outflow channels with perivasculitis of the anterior uvea, which may have caused increased resistance to aqueous flow. The possible increase of the episcleral venous pressure has been argued as a cause of the elevated IOP.45
ELEVATION OF IOP ASSOCIATED WITH ANGLE CLOSURE
Angle closure secondary to peripheral anterior synechiae may cause elevated IOP. However, this is unlikely to occur in scleritis without concurrent uveitis.45 Acute angle closure may occur in eyes with posterior scleritis secondary to choroidal effusion and forward rotation of the iris-ciliary body.47–51
PUPILLARY BLOCK SECONDARY TO POSTERIOR SYNECHIAE
Concurrent uveitis may cause formation of posterior synechiae with seclusio pupillae and iris bombé.
NEOVASCULAR GLAUCOMA
Neovascular glaucoma can occur in eyes affected by scleritis. In a series of 92 eyes enucleated for scleritis, 14 eyes (15%) had a neovascular membrane covering the anterior chamber angle.45
Diagnosis and Differential Diagnosis
How Is Glaucoma Secondary to Scleritis Diagnosed?
Typically, the diagnosis is based on clinical examination.26 In addition to routine laboratory blood tests, skin tests, radiologic tests, electrodiagnostic tests, and tissue biopsy for uveitis workup, patients should have documented damage to the optic nerve secondary to the IOP elevation from scleritis.
What Is the Differential Diagnosis of Glaucoma Associated with Scleritis?
The differential diagnosis of scleritis is crucial for early and effective treatment. The recognition of necrotizing scleritis is particularly important because it is frequently associated with ocular complications and a bad ocular prognosis. Scleritis associated systemic vasculitis indicates its generalization with potential development of lethal systemic complications. Only the early diagnosis and an adequate aggressive therapy can preserve ocular functions and the patient’s life. Conventional steroid therapy generally fails to control the inflammatory activity in necrotizing scleritis. However, application of nonsteroidal immunosuppressive drugs has been shown to control the vasculitic conditions in the majority of cases, to improve ocular prognosis, and to reduce mortality.54 The dry eye syndrome secondary to Sjögren’s syndrome or medication use must be differentiated and treated appropriately to avert sight-threatening complications and to alleviate substantial discomfort.55
Acute rheumatic fever in a young child with fever, sore throat, joint pains, and malaise can present together with scleritis, uveitis, and glaucoma. Patients may have an increased antistreptolysin-O antibody titer. Testing for antistreptococcal antibody is indicated in diagnosing rheumatic fever complicated with scleritis, uveitis, and glaucoma.56
Treatment and Management
Treatment of glaucoma secondary to scleritis is often difficult and complicated. It usually includes topical and systemic corticosteroids and NSAIDs. Aqueous suppressants are applied to lower the IOP. Discontinuation of steroids that are used to treat the underlying scleritis is not always feasible even if an abnormal steroid response is suspected. If medical therapy completely fails to control the IOP, trabeculectomy or implantation of a drainage device may be necessary.
Acute angle closure can be treated with standard therapy regimen (see also Chapter 5). Formation of posterior synechiae can be prevented with the use of mydriatics. Iris bombé is treated initially with mydriatics. If this is unsuccessful, laser pupilloplasty can be tried, or a peripheral iridotomy/iridectomy can be placed.
Future Considerations
Identification and description of inflammatory mechanisms will be necessary for the development of new medications that will control intraocular inflammation without affecting the IOP.
GLAUCOMA ASSOCIATED WITH UVEITIS
Definition
What Is Uveitic Glaucoma?
Glaucoma can be called uveitic glaucoma only when the glaucomatous optic nerve damage, glaucomatous visual field defects, and IOP elevation associated with uveitis have been documented. More specific terms such as hypertensive uveitis can be used for conditions not meeting all the aforementioned criteria.57
What Is the Etiology of Glaucoma Associated with Uveitis?
Elevated IOP commonly complicates any type of uveitis. The inflammatory cells and mediators, as well as the corticosteroids used in the treatment of the uveitis, all contribute to the pathogenesis of uveitic glaucoma by altering the anatomy of the anterior chamber and its angle and by influencing aqueous production and outflow. These changes disrupt the homeostasis of IOP control. Changes in the angle can be acute, such as in secondary angle closure with pupillary block glaucoma, or chronic, such as combined steroid-induced and secondary open-angle glaucoma.57
Uveitic glaucoma may occur by acute angle closure due to iris bombé caused by posterior synechiae, chronic angle closure due to peripheral anterior synechiae, open-angle glaucoma due to obstruction and/or inflammation of the trabecular meshwork, or any combination of the above. If secretory hypotony develops, it may mask compromised outflow. Corticosteroids for treatment of the uveitis may cause an elevation of IOP. Thorough differential diagnosis of the pathophysiologic mechanisms involved in uveitic glaucoma is absolutely essential for successful management of these conditions.58 In uveitic glaucoma the optic neuropathy is caused by an increased IOP, which by itself is elevated due to complications caused by inflammation of the uvea. In addition, inflammatory conditions, such as sarcoidosis, can cause direct damage to the optic nerve.59
Does Uveitis in a Glaucomatous Eye Always Mean Uveitic Glaucoma?
No, even though the differentiation between uveitic glaucoma and glaucoma with uveitis can be difficult, it is important to consider angle closure or open-angle glaucoma and its subtypes in the differential diagnosis of uveitic glaucoma. Pseudoexfoliation, neovascularization, and laser or intraocular surgery may cause secondary inflammation due to disturbance of the blood–aqueous barrier.
Epidemiology and Importance
How Common Is Uveitic Glaucoma?
The epidemiology of uveitic glaucoma is difficult to define because different investigators define glaucoma and uveitis differently in their studies. The frequency of glaucoma in uveitis differs among the different uveitis entities. Anterior uveitis represents 2760 to 45%61 of all uveitis syndromes, achieving 0.2 to 0.4% lifetime cumulative incidence in the general population.62 Human leukocyte antigen HLA-B27–positive acute anterior uveitis is associated with a more serious prognosis when compared to HLA-B27–negative acute anterior uveitis.63
Panek and associates64 noted that 20 out of 76 patients with chronic uveitis developed secondary glaucoma, and among 24 patients with acute uveitis secondary glaucoma was found in three patients. The authors concluded that their results confirmed the concept that secondary glaucoma is a management problem in patients with chronic rather than acute uveitis. In another study, out of 340 children with anterior uveitis 46 (13.5%) developed secondary glaucoma.65
Is the Elevated IOP More Often Associated with a Closed
or Open Anterior Chamber Angle?
The elevated IOP in eyes with uveitis is more often associated with open anterior chamber angles than with angle closure. Frequently the elevated IOP is caused by clogging of the trabecular meshwork with inflammatory cells without developing peripheral anterior synechiae, and rarely by precipitates on the trabecular meshwork without signs of inflammation.66,67 IOP elevation may be caused by obstruction of aqueous outflow by serum components,68 or swelling of the stroma,69 or inflammation of the endothelium of the trabecular meshwork. Glaucoma commonly complicates herpetic uveitis by causing trabeculitis with obstruction of the trabecular meshwork with inflammatory cells.6,70,71 However, closed anterior chamber angles can also be encountered. Several mechanisms of angle closure are possible:
PRIMARY ANGLE CLOSURE
Primary angle closure occurs primarily in predisposed eyes with narrow angles. It is usually difficult to establish what is the primary event.
SECONDARY ANGLE CLOSURE
Peripheral Anterior Synechiae
Inflammatory cells accumulating in the drainage angle of the anterior chamber adhere to the trabecular meshwork and peripheral iris causing peripheral anterior synechiae (PAS). Iris neovascularization that may develop from chronic inflammation can also contribute to the formation of PAS. This leads to occlusion of the drainage angle and thus elevation of the IOP.
Forward Rotation of the Ciliary Body
Edema of the ciliary body may rotate the lens-iris diaphragm forward. The anterior chamber becomes shallow and the angle closes.49
Pupillary Block and Posterior Synechiae
Formation of inflammatory adhesions between the iris sphincter and the lens, or the vitreous in the aphakic patient, or the intraocular lens (IOL) in the pseudophakic patient causes the interruption of aqueous flow from the posterior to the anterior chamber.72
Diagnosis and Differential Diagnosis
How Is Glaucoma Associated with Uveitis Diagnosed?
The diagnosis of active inflammation is based on detection of inflammatory cells in the aqueous and/or vitreous. Flare in the anterior chamber is evidence of vascular incompetence and is usually chronic.73 Careful gonioscopy is absolutely essential for making the correct diagnosis. The presence of precipitates on the trabecular meshwork and scattered peripheral anterior synechiae even in the absence of obvious uveitis is a sign of ocular inflammation.66
What Is the Differential Diagnosis of Glaucoma Associated with Uveitis?
Adenoma of nonpigmented epithelium of the ciliary body can present as anterior uveitis and glaucoma.74 Uveitis glaucoma hyphema syndrome75,76 together with lens-induced uveitis and phacolytic glaucoma77 should be included in the differential diagnosis.
Severe corneal edema in aphakic eye can develop secondary to neovascular glaucoma.78 Nonpenetrating trauma to the eye can induce nonspecific intra-cameral inflammation with cells, flare, fibrin, and increased IOP.79
Rarely, inflammatory glaucoma may present with acute onset of IOP elevation and inflammatory precipitates on the trabecular meshwork in otherwise quiet eyes. This condition is called Grant’s syndrome. Ultrasound biomicroscopy can be useful for imaging inflammatory precipitates in the angle. These patients do not respond well to typical IOP lowering agents but have excellent response to topical steroids. This condition is often bilateral and tends to recur. It may have a systemic association with sarcoidosis. Grant’s syndrome should be considered as a possible diagnosis of elevated IOP in clinically quiet eyes.66,67
Treatment and Management
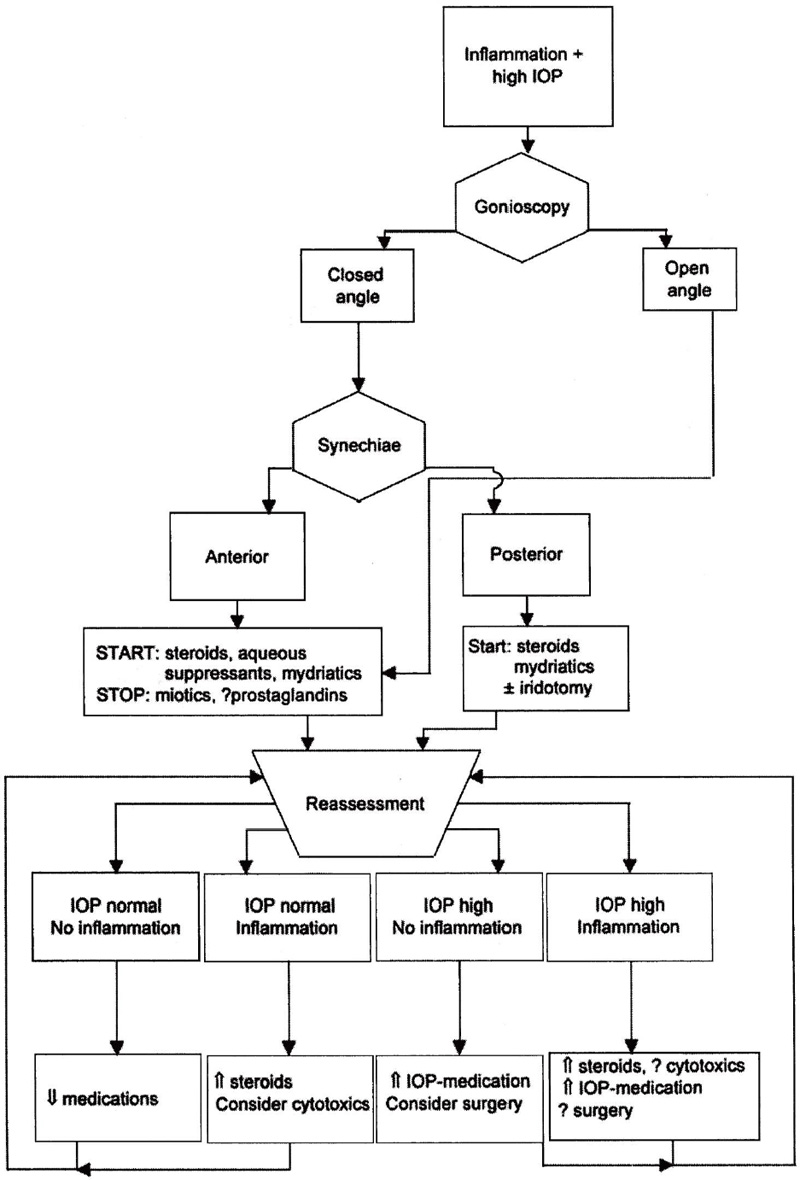
Treatment is simultaneously aimed at the underlying uveitis, preventing future complications that may cause IOP elevation and/or vision loss, and at lowering the IOP if necessary to prevent glaucomatous optic neuropathy. Decreased inflammation in the trabecular meshwork can increase aqueous drainage and thus lower IOP. The reverse, however, might be true if the ciliary body recovers its secretory function with decreasing inflammation. Medical and surgical options are available (Fig. 8–1).
What Is the Medical Therapy?
There are several options for the medical therapy of this condition:
CORTICOSTEROIDS
Corticosteroids are the mainstay of therapy for uveitis. Topical steroids are effective for anterior segment inflammation but not against posterior and intermediate uveitis in the phakic eye. However, they also have the potential to raise the IOP (see also Chapter 15) and cause ocular complications such as fungal keratitis.80
Dexamethasone and prednisolone are the most potent topical corticosteroids available, but have an increased risk of IOP elevation. Fluorometholone and medrysone are weaker steroids, but are less likely to produce IOP elevation. Newer “soft steroids” like loteprednol and rimexolone seem to have the properties to treat ocular inflammation effectively with less risk of affecting the IOP.81,82
Periocular injections provide effective treatment for posterior and intermediate uveitis. Hydrocortisone subconjunctival injection dosages range from 50 to 125 mg. Methylprednisolone depot is available for subconjunctival injection in 40 to 80 mg/0.5 mL concentrations. Triamcinolone suspensions are injected periocularly in 10- to 40-mg doses. Dexamethasone suspension may be injected with an initial dose of 2 to 4 mg. The blood–ocular barrier limits the intraocular penetration of systemic corticosteroids.73
Figure 8–1. Management of uveitic glaucoma. IOP, intraocular pressure.
NONSTEROIDAL ANTIINFLAMMATORY DRUGS
Therapy with NSAIDs is an important adjunct to application of topical, periocular, or systemic steroids. The amount of steroids and thus the rate of side effects can be significantly lessened with topical or oral use of NSAIDs.83
MYDRIATIC AND CYCLOPLEGIC AGENTS
These agents are used to prevent posterior synechiae by dilating the pupil at regular intervals and thus keeping the pupil in motion. They also relieve ocular discomfort by relaxing the ciliary muscle. Homatropine 5% provides good mydriasis and adequate cycloplegia. Its duration of action is much shorter than that of atropine and scopolamine and will therefore keep the pupil much better in motion. Alternatively, a combination of tropicamide 1% and phenylephrine 2.5% can be given.84 Cyclopentolate may be contraindicated in eyes with uveitis because it has been shown to be a chemoattractant to inflammatory cells in vitro.85
AQUEOUS SUPPRESSANTS
Aqueous suppressants such as beta-blockers,86 topical carbonic anhydrase inhibitors, and α2-adrenergics are mainly used to lower the IOP in uveitic glaucoma. Carteolol was found to be effective for the treatment of secondary glaucoma associated with endogenous uveitis.87
What Medications and Procedures Should Be Avoided?
Miotics (parasympathetic drugs) should be avoided because they disrupt the blood–aqueous barrier and thus promote the intraocular inflammation.88 Induced miosis would increase the surface area of the iris coming in contact with the lens, and thus may cause posterior synechiae and iris bombé. Concurrent ciliary body spasm may cause pain and blurred vision.
Prostaglandins such as latanoprost have been shown to cause anterior uveitis and therefore should probably be avoided in the treatment of uveitic glaucoma.89 However, concurrent use of NSAIDs, such as diclofenac, might prevent disruption of the blood–aqueous barrier and maintain the IOP lowering effect.90
Caution should be exercised in patients with a history of glaucoma, previous surgery, or a predilection for uveitis when treated with Nd:YAG laser posterior capsulotomy. These patients may be at risk of developing ciliochoroidal effusions. Symptoms usually resolve after steroid treatment.91
Sympathetic uveitis may develop after filtering procedures on blind, painful eyes. It was the condition of the eye undergoing a filtering procedure that caused the sympathetic uveitis, and not the procedure itself. When glaucoma is absolute, the risk is much higher. Filtering surgery on blind, painful eyes thus presents increased danger for the fellow eye.92
Is Immunosuppressive Therapy Useful?
Immunosuppression with antimetabolites (azathioprine, methotrexate), alkylators (cyclophosphamide, chlorambucil), and/or CsA is sometimes necessary to treat the ocular inflammation.93,94
What Is the Surgical Therapy?
Surgery should be avoided in an eye with active inflammation.95 But when medical therapy fails, surgery is indicated. Iridoplasty, laser peripheral iridotomy, or peripheral iridectomy is recommended, when mydriatics fail to break posterior synechiae that are causing pupillary block. In cases of uncontrolled open- and closed-angle glaucoma, the following surgical options are available:
FILTERING SURGERY
Traditional filtering surgery has a low success rate in eyes with uveitic glaucoma. Failure of trabeculectomy most often results from fibroblast proliferation and subconjunctival fibrosis.96 In patients with uveitis, excessive inflammatory response increases the risk of bleb failure.97 Normal aqueous seems to inhibit subconjunctival fibroblast proliferation,98,99 whereas aqueous100–102 and conjunctiva97 in uveitic eyes contain an increased number of T lymphocytes, which modulate wound healing. Success rates are higher when concurrent antiproliferative agents are used.96,103,104
Trabeculectomy with intraoperative application of mitomycin C for control of uveitic glaucoma achieved an IOP of 21 mm Hg or lower without glaucoma medications in 75%.105
DRAINAGE DEVICES
Use of a drainage implant may be indicated as a primary procedure in glaucoma associated with uveitis.106 Trabeculectomy was successful in four out of five eyes after 1 year, and in three out of four eyes after 2 years. Success rates for Molteno implants were defined as four out of five eyes in the same study and remained stable over 2 years. When significant inflammation is present, aqueous drainage devices are more likely to control IOP.107
CYCLODESTRUCTIVE PROCEDURES
If medical and surgical therapy fail to control IOP, it may be necessary to ablate parts of the ciliary body to reduce aqueous inflow. Techniques for cyclodestruction that have been tried in the past include diathermy, electrolysis, and beta irradiation. Cyclocryotherapy is also available, although this technique has significant limitations. Newer techniques utilize laser energy (Nd:YAG or diode laser) via a transscleral approach. The disadvantages include the inability to precisely quantify the destruction of the ciliary processes and damage to adjacent tissue. Transpupillary cyclophotocoagulation minimizes these problems, but is limited to the small number of eyes in which adequate gonioscopic visualization of the ciliary processes can be achieved. An alternative approach for aphakic eyes is intraocular cyclophotocoagulation, utilizing an endophotocoagulator through a pars plana incision. Depending on the status of the eye, visualization for this technique can be accomplished either by the transpupillary route or with an endoscope.108 Cyclodestructive procedures need to be repeated more frequently when compared to drainage device procedures. However, patients with drainage devices are more likely to have other types of adverse ophthalmic events than patients who had a cyclodestructive procedure.109
Cyclodestructive therapy works best if the drainage angle is partially open. Postoperatively, increased inflammation, however, might close the angle further. It can cause phthisis bulbi more often than any other surgical procedure.
How Are Specific Uveitic Syndromes Handled?
FUCHS’ IRIDOCYCLITIS
This is a syndrome that includes stellate keratic precipitates, heterochromia iridis, cataract, and a chronic anterior uveitis without anterior synechiae formation. The condition is typically unilateral. Fine blood vessels, which have a tendency to bleed very easily, but do not represent neovascularization, can be seen in the angle. Rubeosis iridis and rubeosis of the anterior chamber angle have also been reported.110 Chronic anterior segment ischemia seen in Fuchs’ heterochromic iridocyclitis can be possible mechanism predisposing these eyes to neovascularization.111
The prevalence of glaucoma in this condition varies from 6.3 to 59%.57 The IOP is elevated secondary to reduced outflow facility. Glaucoma is usually treated with aqueous suppressants. The iritis does not respond well to corticosteroids, which by itself may induce IOP elevation in steroid responders. Most studies show that surgical intervention is required in approximately half of the patients;112,113 26.2% of patients with Fuchs’ heterochromic uveitis had glaucoma. Half of these patients presented with glaucoma on initial examination. The risk of glaucoma in patients with Fuchs’ heterochromic uveitis is 0.5% per year, decreasing significantly after 15 years. Causes of IOP elevation include inflammation with peripheral anterior synechiae, rubeosis, lens-induced angle closure, and recurrent spontaneous hyphema. Most patients have chronic open-angle glaucoma. Cataract surgery may precipitate glaucoma. The failure rate of glaucoma drainage surgery was 55.5%, and antimetabolites are needed for filtering surgery.114
POSNER-SCHLOSSMAN SYNDROME
(GLAUCOMATOCYCLITIC CRISIS)
This condition is usually unilateral.115,116 Recurrent episodes of IOP elevation are usually asymptomatic. The angle remains open. The anterior chamber reaction is minimal. Corneal edema is usually present. Patients with Posner-Schlossman syndrome may have peptic ulcers and other gastrointestinal disorders.117 Usually individuals 20 to 50 years old are affected. This condition is self-limited and resolves spontaneously regardless of treatment. But aqueous suppressants and topical steroids may be indicated,57 and patients rarely require filtering surgery.
JUVENILE RHEUMATOID ARTHRITIS (JRA)
Juvenile rheumatoid arthritis is a general name for arthritic conditions in children and is relatively rare.118 Secondary glaucoma is the most devastating complication of chronic uveitis65 and can be seen in 14 to 27% of children.119–123 The mechanism of glaucoma in JRA is either a pupillary block95 and/or development of PAS with progressive angle closure.118 Secondary open angle with obstruction of trabecular meshwork has been reported.124 Because steroids are used in the treatment of JRA uveitis, steroid-induced glaucoma may occur. Medical management with topical and systemic aqueous suppressants initially controls IOP in about half of the patients, with only a third controlled later in the course of the disease.125 Traditional filtering surgery is usually not very successful.65,96,120 The use of antimetabolites with filtering surgery or shunting devices can improve the outcome.57,64,105,107,126 In one study trabeculodialysis has been successful in more than half of the eyes after 2 years.127
INTERMEDIATE UVEITIS (PARS PLANITIS)
Intermediate uveitis is defined as a clinical condition with usually bilateral inflammation of the peripheral retina (“snow banking”) and vitritis.128 It has been associated with sarcoidosis, multiple sclerosis, Lyme disease, and tuberculosis, but often remains idiopathic. Glaucoma occurs in 7 to 8% of adult patients with intermediate uveitis60,129,130 and in 15% of children.131,132 The IOP is elevated probably due to synechiae formation, rubeosis, and corticosteroid-induced glaucoma.133
Management includes topical aqueous suppressants. Cyclocryotherapy131,134,135 has been used. Implantation of a drainage device might be necessary for uncontrolled glaucoma.
BEHÇET’S DISEASE
This disorder presents with acute hypopyon, iritis, aphthous and genital ulcers, and erythema nodosum in young adults.136,137 Ocular involvement is found in 83 to 95% in males and 67 to 73% in females; the male to female ratio is l.78:l.138
Glaucoma usually occurs from obstruction of the trabecular meshwork by inflammatory cells and synechiae formation,136 and sometimes neovascularization.139–143 All patients initially respond to steroids, but steroids appear to be deleterious to visual prognosis and the patients eventually require cytotoxic-immunosuppressive drugs such as chlorambucil and cyclophosphamide after “resistance” to steroids develops. Glaucoma is treated with aqueous suppressants, filtration surgery, or drainage implants. The prognosis is poor. Only one of four patients had useful visual acuity after 10 years.138,144
VOGT-KOYANAGI-HARADA (VKH) SYNDROME
Patients with VKH usually present with bilateral decreased vision and general symptoms such as headache, nausea, vomiting, and hearing loss. Bilateral panuveitis with serous retinal detachments and underlying choroidal infiltrates are seen. Perilimbal vitiligo is common.145 Typically, affected individuals are 20 to 50 years of age and of Asian or Native-American descent. Uveitic glaucoma occurs in almost every third patient with VKH syndrome.146–148 The IOP is usually elevated due to secondary open-angle glaucoma. Pupillary block due to posterior synechiae is also encountered frequently.146–148 Rarely, angle closure secondary to choroidal effusion with anterior rotation of the ciliary body can be seen.149 The main treatment consists of systemic corticosteroids. In severe cases or in patients who cannot tolerate high doses of steroids, cytotoxic agents or immunosuppressives such as CsA might be indicated.147,150 The possibility of steroid-induced glaucoma must always be kept in mind. Glaucoma is treated medically with aqueous suppressants. If surgery is necessary, drainage devices offer a greater degree of success than trabeculectomies even with antiproliferative agents.147,148
SYMPATHETIC OPHTHALMIA
This entity consists of bilateral granulomatous panuveitis after injury to one eye. Uveitic glaucoma develops in 43% of the eyes with sympathetic ophthalmia.151 Filtering procedure on blind painful uveitic glaucoma eyes may provoke sympathetic ophthalmia in the fellow eye.92 Mechanisms of uveitic glaucoma in sympathetic ophthalmia include angle closure secondary to pupillary block and iris bombé,152 angle closure due to a thickening of the iris and ciliary body by cellular infiltration,153 PAS and infiltration of the outflow pathway by inflammatory cells.151 The main treatment for sympathetic ophthalmia consists of administration of topical and systemic corticosteroids and occasionally cytotoxic or immunosuppressive agents. Filtering surgery or implantation of a drainage device might be necessary.
SARCOIDOSIS
Ocular manifestations of sarcoidosis most commonly include bilateral chronic granulomatous uveitis with mutton fat keratic precipitates, iris nodules, and synechiae.154 Ocular manifestations in systemic sarcoidosis occur in 38% of patients.154 Granulomatous uveitis occurs in 52154 to 74% of patients with ocular sarcoidosis. Glaucoma develops in 10.9 to 25.5% of patients with sarcoidosis uveitis.154,155 Blacks with sarcoid uveitis have a higher risk than whites of developing glaucoma and blindness.154
Impairment to outflow in sarcoid uveitis arises from posterior and anterior synechiae, neovascularization of the angle and the iris, steroid-induced ocular hypertension,156 and inflammatory precipitates and changes in the trabecular meshwork.66 The aqueous level of angiotensin-converting enzyme (ACE) may be elevated in patients with uveitis suspected to have sarcoidosis and may need to be measured when serum ACE level and other laboratory and radiologic findings are negative.157 Association of elevated serum levels of ACE in patients with uveitis of unknown etiology suggests the diagnosis of ocular sarcoidosis.158 Sarcoidosis can cause direct damage to the optic nerve and mimic glaucomatous optic neuropathy.59 Glaucoma associated with sarcoid uveitis is treated with corticosteroids and aqueous suppressants. Filtering surgery or implantation of a drainage device is indicated if IOP cannot be medically controlled. Steroids should be used pre- and postoperatively. Pupillary block is treated with laser iridotomy or surgical iridectomy.
HERPETIC UVEITIS
Herpes Simplex Herpes simplex infection may present as superficial keratitis, disciform keratitis, necrotic stromal keratitis, neurotrophic ulcer, and retinitis. Approximately 5% of all uveitis cases in adults are associated with herpes simplex infection.64 Increased IOP in ocular herpes infection associated with uveitis varies from 28 to 40%, but only 10% have secondary glaucoma.6,64 Disciform and necrotic stromal keratitis is more commonly associated with increased IOP. In severe cases the incidence can reach 80%.6 Obstruction of the trabecular meshwork with inflammatory products, trabeculitis,6,13 and angle closure71 most commonly cause IOP elevation in herpetic uveitis. The initial management of elevated IOP is directed at controlling viral replication. Oral and/or topical acyclovir and topical trifluridine are most effective because they penetrate the ocular tissues easily. Topical cycloplegics are helpful to control ciliary spasm, and topical corticosteroids may be used if inflammation is severe or persists despite the antiviral treatment. Topical steroids alone can reactivate or aggravate the herpetic infection. Therefore, they should never be used without initial antiviral coverage. IOP usually returns to normal levels when the inflammation subsides. Aqueous suppressants are effective if IOP needs to be controlled. About 10% of patients who have persistent IOP elevation despite medical treatment may require surgery.6
Herpes Zoster Ocular involvement occurs in two-thirds of patients with herpes zoster ophthalmicus. This might include conjunctivitis, superficial keratitis, stromal keratitis, neurotrophic keratitis, uveitis, scleritis, retinitis, choroiditis, and optic neuritis. Elevation of IOP and glaucoma occurs in 16 to 50% of cases associated with corneal involvement of keratitis and uveitis.17,64,159 Decreased outflow facility due to trabeculitis and inflammatory debris in the trabecular meshwork is thought to cause the IOP elevation.160,161
Oral acyclovir given early in the course of the disease seems to reduce the risk of complications, such as uveitis and associated elevated IOP.162 Later in the course of the disease, uveitis is no longer due to viral replication, but rather to ischemia.163 Uveitis and IOP should then be treated with topical corticosteroids, mydriatics, and aqueous suppressants as indicated.
Future Considerations
Mycophenolate mofetil (MMF), a potent selective uncompetitive and reversible inhibitor of ionisine monophosphate dehydrogenase involved in purine synthesis is currently under investigation. It is an immunosuppressive and steroid-sparing agent, which can be used to treat ocular inflammatory disease. In a recent study mycophenolate mofetil was given 1 g twice daily in conjunction with steroids, as a steroid-sparing agent, or as an additional agent with CsA, or instead of CsA or azathioprine.164 The addition of MMF to immunosuppressive regimens improved symptoms and allowed reducing the dose of prednisone in most patients. MMF may become a useful immunosuppressive drug for controlling ocular inflammation without significant side effects.
References
7. Tavs LE: Syphilis. Major Probl Clin Pediatr 1978;19:222–256.
8. Grant WM: Late glaucoma after interstitial keratitis. Am J Ophthalmol 1975;79:87–91.
10. Lichter PR, Shaffer RN: Interstitial keratitis and glaucoma. Am J Ophthalmol 1969;68:241–248.
11. Kuriakose T, Thomas PA: Keratomycotic malignant glaucoma. Indian J Ophthalmol 1991;39:118–121.
26. Watson PG, Hayreh SS: Scleritis and episcleritis. Br J Ophthalmol 1976;60:163–191.
28. Harbin TSJ, Pollack IP: Glaucoma in episcleritis. Arch Ophthalmol 1975;93:948–950.
40. Watson PG: Management of scleral disease. Trans Ophthalmol Soc UK 1966;86:151–167.
42. Becker B, Mills DW: Corticosteroids and intraocular pressure. Arch Ophthalmol 1963;70:500.
47. Benson WE: Posterior scleritis. Surv Ophthalmol 1998;32:297–316.
55. Michelson PE: Red eye unresponsive to treatment. West J Med 1997;166:145–147.
58. Ritch R: Pathophysiology of glaucoma in uveitis. Trans Ophthalmol Soc UK 1981;101:321–324.
59. Beardsley TL, Brown SV, Sydnor CF, et al: Eleven cases of sarcoidosis of the optic nerve. Am J Ophthalmol 1984;97:62–77.
70. O’Connor GR: Recurrent herpes simplex uveitis in humans. Surv Ophthalmol 1976;21:165–170.
72. Schlaegel TFJ: Complications of uveitis. Int Ophthalmol Clin 1977;17:65–74.
78. Rodgin SG: Neovascular glaucoma associated with uveitis. J Am Optom Assoc 1987;58:499–503.
90. Miyake K, Ota I, Maekubo K, et al: Latanoprost accelerates disruption of the blood-aqueous barrier and the incidence of angiographic cystoid macular edema in early postoperative pseudophakias. Arch Ophthalmol 1999;l17:34–40.
98. Herschler J: The inhibitory factor in aqueous humor. Vision Res 1981;21:163.
120. Chylack LTJ, Bienfang DC, Bellows AR, et al: Ocular manifestations of juvenile rheumatoid arthritis. Am J Ophthalmol 1975;79:1026–1033.
123. Kanski JJ: Uveitis in juvenile chronic arthritis. Clin Exp Rheumatol 1990;8:499–503.
125. Kanski JJ: Juvenile arthritis and uveitis. Surv Ophthalmol 1990;34:253–267.
128. Aaberg TM: The enigma of pars planitis [editorial]. Am J Ophthalmol 1987;103:828–830.
131. Giles CL: Pediatric intermediate uveitis. J Pediatr Ophthalmol Strabismus 1989;26:136–139.
132. Giles CL: Uveitis in childhood—part II. Intermediate. Ann Ophthalmol 1989;21:20–22.
136. Michelson JB, Chisari FV: Behçet’s disease. Surv Ophthalmol 1982;26:190–203.
137. Michelson JB, Friedlaender MH: Behçet’s disease. Int Ophthalmol Clin 1990;30:271–278.
140. Nussenblatt RB: Uveitis in Behçet’s disease. Int Rev Immunol 1997;14:67–79.
145. Sugiura S: Vogt-Koyanagi-Harada disease. Jpn J Ophthalmol 1978;22:9–35.
147. Moorthy RS, Inomata H, Rao NA: Vogt-Koyanagi-Harada syndrome. Surv Ophthalmol 1995;39:265–292.
153. Samuels B: Glaucoma and sympathetic ophthalmia. Arch Ophthalmol 1999;117:1031–1039.
155. Jabs DA, Johns CJ: Ocular involvement in chronic sarcoidosis. Am J Ophthalmol 1986;102:297–301.
157. Weinreb RN, Sandman R, Ryder MI, et al: Angiotensin-converting enzyme activity in human aqueous humor. Arch Ophthalmol 1985;103:34–36.
164. Larkin G, Lightman S: Mycophenolate mofetil. A useful immunosuppressive in inflammatory eye disease. Ophthalmology 1999;106:370–374.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree