CHAPTER 9 General Considerations of Anesthesia and Management of the Difficult Airway
The OLHN surgeon with expertise in rigid laryngoscopy and bronchoscopy, flexible fiberoptic bronchoscopy, and surgical approaches to the airway is uniquely qualified to take the lead surgical role in a team approach with the anesthesiologist to safely manage difficult airway/intubation patients. The goal of airway management is simple: to provide the most expeditious form of management that has the lowest potential for injury and the greatest potential for control of the airway.1
This chapter presents the anesthetic component of complex airway management in a manner that OLHN surgeons can incorporate into their practice. The focus of this chapter is on clinical airway algorithms and the decisions that must be made by the anesthesiologist together with the OLHN surgeon to safely provide anesthesia and airway management for their patient or for a non-OLHN patient seen in joint consultation for airway management. In-depth discussions about the pharmacology and physiology of anesthesiology are beyond the scope of this chapter but can be found in any number of authoritative textbooks of anesthesiology.2,3
Difficult Airway/Intubation: A Multispecialty Problem
Patient Identification
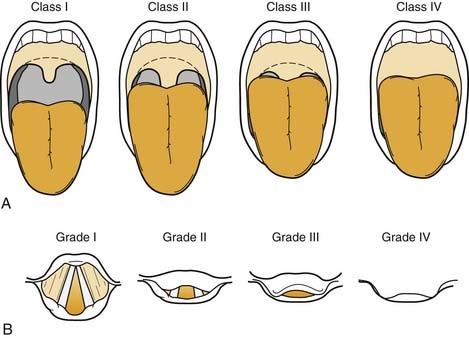
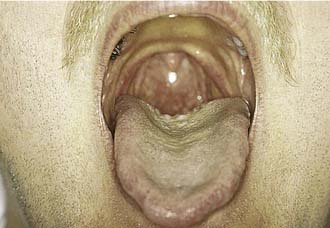
Controversy regarding predictors and definitions of “difficult” exists in both intraspecialty and interspecialty, dependent and independent of practitioner skill, related to specific techniques, and complicated by changing patient pathophysiology.4 Some patients may be anticipated to be difficult to intubate on the basis of a history of difficulty intubation or clinical predictors of difficult intubation. The ASA Practice Guidelines for Management of the Difficult Airway5 reviews some of the historical or physical examination findings possibly suggestive of a difficult intubation. Some of these predictors of anticipated difficulty with conventional direct laryngoscopy (MAC/Miller) include a large overbite, large tongue, narrow mouth opening, or short chin. Various prediction models, such as correlation with Mallampati oral view I to IV to the Cormack and Lehand laryngoscopic view grades I to IV have been proposed, but none offer 100% sensitivity for prediction of a difficult airway (Figs. 9-1 to 9-4).6 Historically, anesthesiology literature cites an incidence of 1% to 3% for unanticipated difficult airway/intubation in patients undergoing general endotracheal anesthesia.5–7 The airway management technique used to define “difficult” in this literature was conventional rigid laryngoscopy (Macintosh or Miller blades).
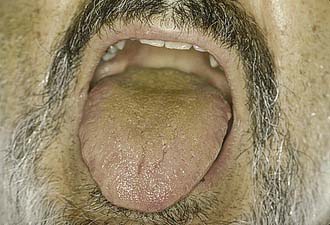
Figure 9-4. Postoperative evaluation of patient in Figure 9-3. Note that without the tongue blade, the patient has a Mallampati class IV airway and should have been considered to be anticipated difficult intubation with conventional laryngoscopy.
Despite advances in airway management techniques and refinement of difficulty predictors, the cited 1% to 3% incidence of unanticipated difficulty has not changed and is still defined by conventional laryngoscopy.6–9 In an institution where approximately 25,000 general endotracheal anesthetic procedures are done annually, there are potentially 250 to 750 unanticipated difficult airway/intubations per year. Assuming that a full-time practicing anesthesiologist would encounter one unanticipated difficult airway/intubation per year, then, based on ASA membership (which represents 90% of practicing anesthesiologists), there are potentially 30,000 to 90,000 unanticipated difficult airway/intubations annually in the United States. However, these numbers may underestimate the true incidence, because anesthesiologists may not recall the more common “near misses” as vividly as they recall the smaller number of actual difficult airway/intubations in which the outcome was suboptimal. On a national and international level, the scope of this problem and its impact on patients, practitioners, and the health care system is sufficient to warrant vigorous efforts to identify and implement solutions.
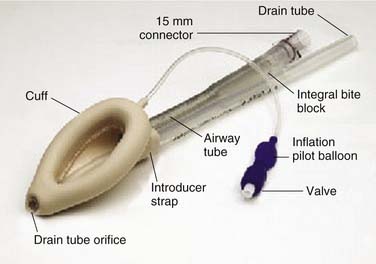
In addition to those patients who have unanticipated difficult airway/intubations on initial presentation, there are cohorts of patients who have anticipated complex airway management; these patients can be successfully managed by a variety of innovative and specialty-specific techniques. Some of these techniques (laryngeal mask airway [LMA]10 and Combitube)11 are readily available (Figs. 9-5 and 9-6), require minimal practitioner education or training, and are inexpensive; other techniques (fiberoptic bronchoscope, surgical airway, specialized rigid laryngoscopes, and fluoroscopic-assisted intubation) are available primarily in specialty centers, may require extensive practitioner skill, and may be relatively expensive. For patients who have undergone head and neck surgery and have visible or hidden implants (e.g., laryngeal stents, thyroplasties), specific considerations for airway management may be unknown to future providers (e.g., thyroplasty patients might require smaller endotracheal tubes than anticipated), thus compromising patient safety and increasing practitioner risk for adverse events.
Consequences of Difficult Airway Management
Three studies specifically demonstrated the consequences of difficult airway/intubation management on liability exposure.12–14 An analysis of approximately 5000 claims filed in the Maryland legal system over a 15-year period in which one or more anesthesiologists were named as defendants revealed that insertion of an endotracheal tube was the sixth most common medical procedure leading to a liability claim. Most of these claims also included other members of the operating room team (e.g., otolaryngologists, general surgeons, nurse anesthetists, orthopedic surgeons, plastic surgeons, cardiac surgeons, dentists, or nurses) as defendants. One claim (in 1994) resulted in a jury award of $5 million.15 In a 1992 loss analysis study conducted by the Physicians Insurers Association of America (PIAA), files from 43 physician-owned malpractice insurance companies (representing approximately 2000 anesthesiologists nationally) ranked “intubation problems” as the third most prevalent misadventure (behind “tooth injury” and “no medical misadventures”). The average paid indemnity for 175 of 339 files was $196,958.15 The ASA Committee on Professional Liability closed claims study found that respiratory events were the most common cause of brain damage and death during anesthesia, with difficult intubation being the likeliest category for risk reduction. The median payment for respiratory claims was $200,000.12
To put these statistics in perspective, the following must be considered.
Advances in Monitoring
As a previous section made clear, there are serious legal and financial consequences of failed airway management. Fortunately, adverse outcomes related to poor airway management have decreased in frequency with the standardization of anesthetic monitoring. The American Society of Anesthesiology Standards for Basic Anesthetic Monitoring statement was first released in 1986 and further amended in 1998 and 2005.17 These standards call for continual evaluation of the patient’s oxygenation, ventilation, circulation, and temperature during all administered anesthetics. Practically speaking, the standard mandates continuous oxygen analysis of the anesthetic circuit, pulse oximetry, end-tidal capnometry, tidal volume measurement, electrocardiography, and temperature, as well as intermittent (no less frequent than every 5 minutes) measurement of arterial blood pressure and heart rate. In addition, the routine use of the neuromuscular blockade (NMB) monitor (also called the “twitch” monitor) to assess degree of muscle paralysis and return of muscle strength after pharmacological reversal of paralyzing agents significantly contributed to improved patient safety. Compared with earlier eras, this document made the important leap of elevating pulse oximetry and capnometry to standards of care, thus allowing more rapid, accurate recognition of oxygen desaturation and rapid recognition of previously unrecognized esophageal intubation.
The consequences of this change were staggering. Respiratory system adverse events (including airway mishaps) accounted for 36% of claims in the ASA Closed Claims Project data set for the 1970s, but this percentage decreased to 14% of claims in the 1990s after implementation of the routine use of capnometry and pulse oximetry.18 Claims related to death or brain injury likewise had a similar drop in the percentage that was attributable to respiratory adverse outcomes. Caused in part by continuous pulse oximetry and capnometry, anesthesiology has been a leader in the patient safety movement in the past decade.
Standard Anesthetic Pharmacology
Paralytic Agents
The major advantage of succinylcholine is its very fast onset of action. Paralysis sufficient for endotracheal intubation can be reliably produced within 45 to 60 seconds. Another advantage is its short duration of action, because clinical paralysis usually dissipates within 5 minutes of an intubating dose. It has been thought that this quick return of strength would allow resumption of spontaneous respirations if positive-pressure ventilation were not successful. However, a past study shows that deleterious oxygen desaturation may occur before resumption of spontaneous respirations.19 In addition, the small percentage of patients who are pseudocholinesterase deficient will have prolonged paralysis after administration of succinylcholine. Vigilant use of the NMB monitor has led to increased diagnosis of patients with atypical cholinesterase activity, which varies with the population but is cited to be 1 : 2800 in the general population in the United States, with a 1 : 1 male/female ratio.3 Confirmatory blood laboratory diagnosis is made by determining the patient’s dibucaine number. Succinylcholine has also been identified as the most common muscle relaxant trigger for malignant hyperthermia (MH).20 Primary contraindications for the use of succinylcholine include known or suspected MH, increased intracranial pressure, increased intraocular pressure, and elevated potassium. Although not contraindicated in patients with pseudocholinesterase deficiency, administration should be monitored with the NMB monitor to verify full return of strength before extubation.
Standard Induction versus Rapid Sequence Induction
An important question to ask before this point is whether the patient is at risk for aspiration of gastric contents into the airway, an event that can be potentially catastrophic. The patient’s risk of aspiration of gastric contents helps determine whether the patient should be managed with rapid-sequence induction (RSI) and intubation or with a nonrapid sequence of events. The increased risk of aspiration is due to the presence of gastric contents and is the reason anesthesiologists are concerned about the length of time patients have been without food or drink before surgery. The ASA has published guidelines for preoperative fasting that are based on the time required for gastric emptying in healthy patients.21 As surgeons, your familiarity with these guidelines can prevent the delay or cancellation of elective surgery.
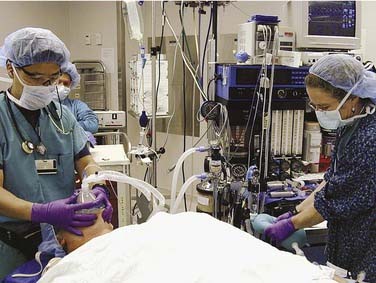
In patients without increased risk of aspiration, a controlled and stepwise approach is taken with induction and intubation. After monitoring and preoxygenation, general anesthesia is induced. Once the patient is unconscious, positive-pressure mask ventilation is performed (Fig. 9-7). Only after successful mask ventilation is established is a paralyzing agent given. This stepwise approach to the airway increases patient safety because, even if intubation cannot be performed successfully, it is known that the patient can be mask ventilated and oxygenated while the paralytic wears off or alternative intubation techniques are readied. The ability to ventilate a patient is more crucial than the ability to intubate a patient, and bag-valve-mask ventilation is a lifesaving skill that every anesthesiologist must master. After successful mask ventilation, the paralytic is given, and intubation is performed after the paralytic takes effect.
The risk of an RSI is that intubation may not be successful and the ability to mask ventilate the patient has not been previously established. The most dangerous result of failed RSI could be a paralyzed patient who cannot be ventilated or intubated (see Case 8).
Difficult Airway/Intubation: Advances in Airway Devices and Techniques
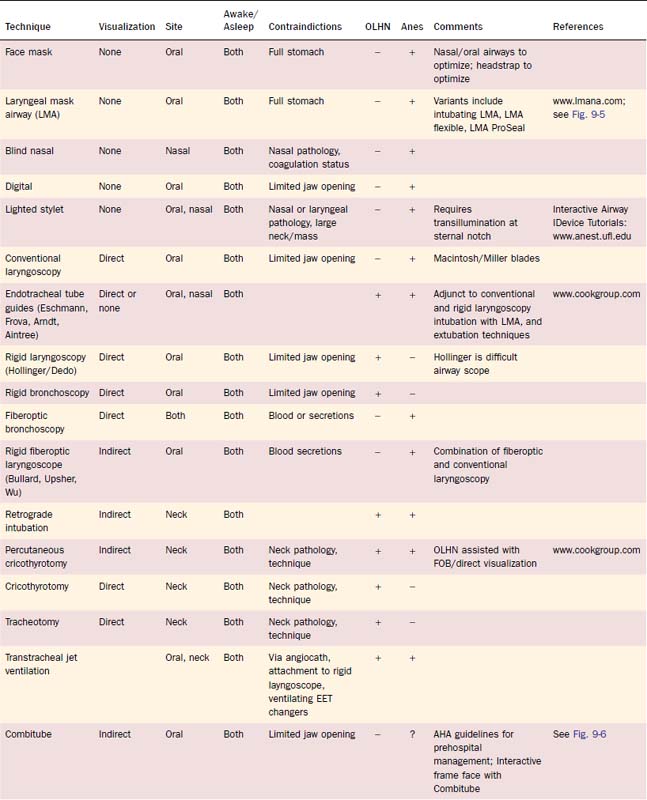
In parallel with organizational responses to difficult airway management, individual practitioners responded with inventions of new airway devices and with innovative combinations of existing techniques. In Table 9-1, a brief review of many of these devices is presented, with identification of primary use by anesthesiology and of OLHN. For a more in-depth discussion of the devices and appropriate techniques, refer to Airway Management: Principles and Practice22 and references identified in Table 9-1.
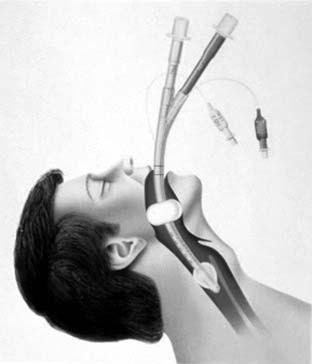
In the early 1990s, OLHN surgeons had the following airway management techniques: awake and asleep surgical airway, awake fiberoptic bronchoscopy, and asleep rigid laryngoscopy and bronchoscopy (Fig. 9-8).
Indisputably, the most significant invention in the recent history of airway management was the LMA (see Fig. 9-5).10 Although introduced into the United States in the early 1990s as an alternative to elective face mask ventilation (Fig. 9-9) in general anesthetics not requiring intubation, the value of the LMA as a rescue device for the most devastating situation “cannot intubate/cannot ventilate” quickly became realized. In the 1993 ASA Practice Guidelines for Management of the Difficult Airway, the LMA was the airway device that routed the Difficult Airway Algorithm into the significant branch points. In the 2003 amended Guidelines, the LMA was promoted as a first choice device for cannot-ventilate rescue options.23
In the past decade, for elective difficult airway management, the “family” of LMAs (Classic, ProSeal, Flexible, and Fastrach) has had a major impact on the decision branches with the Difficult Airway Algorithm. Specifically, many practitioners decide that if they can mask the patient with an LMA, definitive placement of an endotracheal tube (ETT) can be achieved with either fiberoptic bronchoscopy or ETT exchanger assistance. Backup plans, however, must be in place, because these techniques can be difficult and unsuccessful, as described in Case 7.
Table 9-1 briefly presents many of the advances in airway management devices discussed previously and throughout this chapter. References, including links to more comprehensive websites, are provided for additional information. Cases 1 to 10 span 10 years of experiences with difficult airway patients and advancements of airway management techniques.
The Johns Hopkins Departments of Anesthesiology and Critical Care Medicine and Otolaryngology—Head and Neck Surgery Airway Management Initiative and Formation of a Hospital-Wide Airway Service
Conceptual Framework
The Johns Hopkins Medical Institutions Departments of ACCM and OLHN Surgery responded to the release of the ASA guidelines by developing an Airway Management Initiative that has evolved over the past 15 years into the present-day Airway Management Service, education program, and hospital-wide difficult airway management response system. For the first 6 years, both departments provided financial support. However, in 1999, the Legal Department issued the following statement regarding the Airway Management Initiative and multidisciplinary Airway Service that resulted in full financial support by The Johns Hopkins Medical Institutions24:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree