Chapter 34 Gene Therapy for Retinal Disease
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
Background: preclinical gene therapy studies
Nonviral gene delivery
There are nonviral methods of delivering nucleic acids to cells, and those include use of physicochemical agents to compact the DNA and/or transport it across the membrane lipid bilayer.1,2 The transport across the bilayer can also be achieved through physical means, including propulsion with a “gene gun” or by electroporation or iontophoresis. Such methods can also be combined with reagents that enhance integration into the host chromosome.3 There are several potential advantages of nonviral approaches: First, they can be used to deliver DNA of unlimited size. Second, there is a smaller chance of detrimental immune response since the only antigen would be the nucleic acid itself plus any protein that is used as a condensation agent. Several studies have demonstrated proof of concept of retinal gene therapy using nonviral DNA delivery1,4,5 and additional studies will reveal the long-term safety, stability, and efficacy of this approach.
Viral vector-mediated gene delivery
Recombinant viruses are genetically neutered or crippled so that they cannot reproduce and cause an infectious disease once they infect a target cell. There is a large list of recombinant viruses that have been tested in the retina (Table 34.1). Different viruses have different attributes and challenges, including cargo capacity, ease of purification, cellular specificity, and immune response. However, a large portion of these have been used to demonstrate efficacy in animal models of retinal disease (Table 34.2).
Table 34.1 Vectors tested for transduction characteristics in animal models. Various retinal cell targets are listed; however, the exact targets depend on the route of administration, dose, species, and modifications to the vector
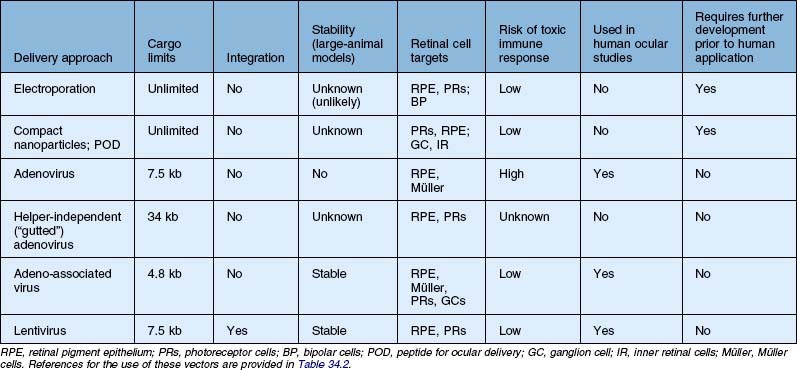
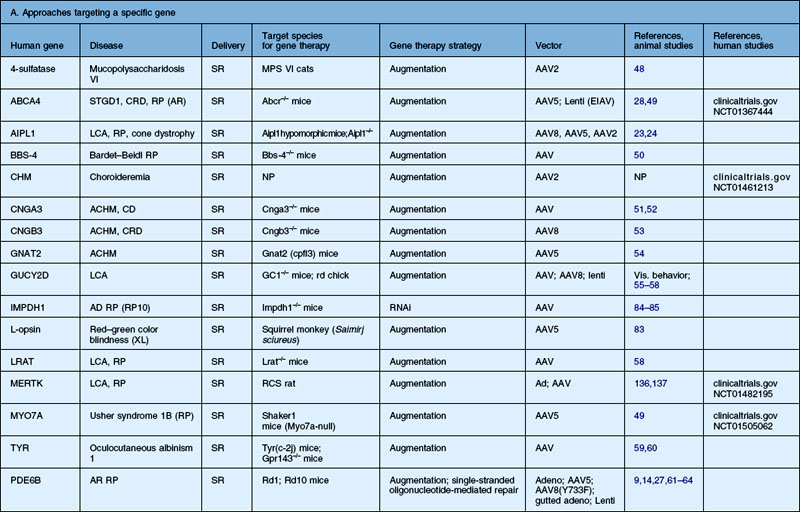
Table 34.2 Recent studies showing proof of concept of retinal gene therapy strategies using (A) selected approaches targeting a specific gene and (B) “generic” approaches that could potentially be used regardless of the disease-causing genetic defect. This list highlights many of the recent studies aiming to treat animal models of retinal degeneration, retinal development anomalies, and retinal neovascularization. Several of these studies have been carried forward to human clinical trial and those references are listed as well
The first recombinant adenovirus vectors, generated from the common respiratory virus, carried deletions in the adenoviral E1, E3 genes, and these Ad type 5 (Ad5) vectors were the first to be evaluated for retinal gene transfer in the differentiated retina.6,7 Adenovirus vectors result in high levels of gene expression within 24–48 hours. When injected subretinally, they target retinal pigment epithelial (RPE) cells efficiently in the adult eye and also Müller cells.6,7 When injected intravitreally, they target Müller cells and cells in the anterior segment, including corneal endothelium, lens and iris epithelium, and cells in the outflow tract (such as trabecular meshwork cells).8 Similar results are found when recombinant adenovirus is injected into the undifferentiated (early postnatal) retina; however, in addition to RPE cells, progenitor cells are targeted in the neonatal mouse eye.9,10 Additional manipulations of adenovirus vectors have yielded reagents that target photoreceptors more efficiently.11,12
A disadvantage of the early generations of E1, E3-deleted adenovirus is that it still carries viral open reading frames. These can enhance its immunogenicity, even in the immune-privileged environment of the eye. These vectors were thus also used to probe the nature of the intraocular immune response. When these vectors were injected subretinally, transgene expression persisted for several weeks to months. However, when injected intravitreally, transgene expression ceased within 2 weeks. Expression could be prolonged by incorporating immune-suppressant molecules, however.13 Efforts were made to generate adenovirus vectors lacking any viral open reading frames, the so-called “gutted” or helper independent vectors, thereby reducing immune clearance and allowing stable transgene expression.14 Such vectors do indeed result in more stable transgene expression than the first-generation vectors and, further, have a much greater cargo capacity than the original adenoviral vectors (Table 34.1).15 They are more difficult to generate, however, and preparations are likely to be contaminated by wild-type adenovirus.
Adeno-associated virus (AAV) vectors do not carry any virus open reading frames (and thus encode any virus-specific proteins) and therefore are generally more favorable from an immunologic standpoint than adenovirus vectors (Table 34.1). There is also an abundant amount of safety data related to AAV administration in animals and in humans, both systemically and intraocularly. Recombinant AAV (rAAV) vectors have an added benefit in that they target a more diverse set of cell types than adenoviral (or other) vectors. Unlike lentiviral vectors, AAV vectors do not integrate, or do so only rarely (Table 34.1). However, since the transgene persists in episomal fashion in the target retinal cells, rAAV vectors result in stable transgene expression. Expression persists for the life of small animals (mice and rats) and at least for many years in large animals and humans.16–19 rAAV vectors are useful for delivering genes efficiently to many types of retinal cells. A disadvantage of these vectors is their relatively limited cargo capacity (a maximum of 4.8 kb) (Table 34.1).
In AAV vectors, the transgene cassette is bordered by the inverted terminal repeats (ITRs) from an AAV2 genome. The original AAV vectors were generated by packaging the transgene cassette ITRs into an AAV serotype 2 capsid and this resulted in “AAV2/2” vectors, i.e., ITRs of an AAV serotype 2 genome packaged into an AAV2 serotype 2 capsid. (Often investigators skip the reference to the ITR serotype and refer to AAV2 vectors instead of AAV2/2 vectors.) More than a dozen AAVs of different serotypes have been described. Cross-packaged rAAVs (for example, rAAV2/5 vectors generated after packaging the AAV2 cis plasmid into AAV serotype 5 capsid) differ significantly from rAAV2/2 vectors with respect to cellular specificity, efficiency of transduction, and onset of transgene expression. While rAAV2/2 targets RPE cells efficiently (and photoreceptors less efficiently), it takes up to 6 weeks for transgene expression mediated by this vector to plateau.20–22 In comparison, rAAV2/5 and rAAV2/8 vectors transduce photoreceptors with much higher efficiency than rAAV2/2 and result in transgene expression within 5–10 days of delivery.20,21 This information is helpful in selecting vectors for particular applications. For example, in a relatively slowly progressive retinal degenerative disease (Leber congenital amaurosis (LCA) due to RPE65 mutations), rAAV2/2 performs well in delivering a therapeutic transgene to RPE cells. For an animal model with a much faster rate of degeneration (e.g., LCA due to AIPL1 mutations23,24), it is necessary to use a vector with a much faster onset of expression and which targets photoreceptors efficiently (such as a rAAV2/5 or 2/8 vector; Table 34.2).
Vectors originally based on the human immunodeficiency virus, lentiviral vectors, have been shown to be safe in animal models and their safety in humans is currently being tested (Table 34.2). A number of groups have since generated vectors based on viruses that were identified in nonhuman species, such as equine Lentivirus.25,26 Transgene expression is stable after lentiviral administration, because these vectors mediate integration into the host chromosome (Table 34.1). Lentiviral vectors target RPE cells efficiently after subretinal injection and, in undifferentiated retina, also target neural progenitor cells. They have thus been used to demonstrate efficacy in animal models of RPE disease, such as LCA-RPE65 or autosomal recessive (AR) retinitis pigmentosa (RP) due to PDE6B mutations27 (Table 34.2). Lentiviral vectors are also attractive in that they can carry a cargo of ~7.5 kb (Table 34.1). Specific modifications also allow lentiviral vectors to target mature photoreceptors.28 The lentiviral envelope can be swapped in order to alter cellular tropism.22 Since lentiviral vectors are integrating vectors, there is a concern about the potential for insertional mutagenesis. To date there has been no report of such an effect after retinal administration in animal models.
Surgical delivery
When a subretinal injection is performed through a small retinotomy, a retinal detachment or “bleb” is raised (Fig. 34.1). Most if not all of the volume of injected material is trapped between the outer retina and RPE as a localized retinal detachment. There is negligible escape of material back through the retinotomy site into the vitreous, as evidenced by the fact that, initially, the size of the bleb does not change once it is formed. There is apparently little pressure differential between the subretinal space and vitreous once the bleb is established and this is especially so when the scleral incisions are closed. In addition, the small-gauge cannulas used for the subretinal injection appear to be self-sealing, especially when a gas tamponade or formed vitreous is present. Since a bleb raised by subretinal injection tends not to expand beyond the border of the initial injection, the volume of distribution of the administered agent is limited, especially when compared to an intravitreal injection or systemic administration. The concentration of the compound contained in the bleb remains high and may even increase as the RPE cells extract free water from the vehicle used to dilute the agent. Limiting the volume of distribution in this manner may serve both to increase the efficiency of drug delivery and to decrease local and systemic toxicities by restricting diffusion of the drug. The location of the original subretinal detachment cannot be appreciated in most species (including humans) after it has flattened.18,29 (The location is often visible in dogs years after the injection, however, because of alterations in reflectivity of the underlying tapetum due to the procedure: Fig. 34.1.)
Since the RPE and Bruch’s membrane are typically not violated when subretinal injection is performed by the pars plana approach, there is further protection against systemic exposure of antigens via the highly vascular choroidal circulation. In addition, there is minimal disruption of retinal vasculature since the placement of the injection cannula is done under direct visualization. Therefore, the integrity of the blood–ocular barrier remains intact when subretinal delivery is performed in this manner. There is in addition an immunologic compartmentalization when antigenic material is delivered to the subretinal space. Limiting exposure to this area may result not only in characteristic immunoprivilege behavior evident with intraocular delivery but, when delivery is confined to the subretinal space, antigenic tolerance can be induced due to immune-deviant response.30,31 This unique property of the subretinal space is of great significance in the delivery of biologics and in gene therapy in particular as the development of antibody/immune response to both viral antigens and foreign transgene products may limit the effectiveness of treatment.
The major differences in surgical methods between various investigators who have carried out gene transfer in humans so far18,29,32–34 involve: (1) use of perioperative systemic corticosteroid therapy; (2) the removal of posterior cortical vitreous prior to subretinal injection; and (3) placement of a gas (air) bubble after the injection is performed. With regard to systemic corticosteroid use, there appears to be no important difference in efficacy. It should be noted that all studies employ the use of topical and periocular corticosteroid to suppress surgical inflammation.
The surgical protocol for the Children’s Hospital of Philadelphia (CHOP) gene therapy trial for LCA2 specifies removal of the posterior hyaloid.18,29 In many instances, a complete posterior vitreous detachment (PVD) is already present despite the young age of the subjects enrolled in this trial. This is not unexpected as vitreous abnormalities, including the presence of debris or posterior separation, are characteristic features of eyes with retinal degeneration. In cases without PVD, the posterior cortical vitreous is engaged with active suction and the hyaloid face is gently separated to create a complete PVD, as evidenced by the presence of a glial ring separating from the optic nerve head (Weiss ring). Once the presence of a PVD is confirmed, the mobilized vitreous is removed as completely as possible, with special attention to pare back any gel in the vicinity of the active sclerotomy sites. This is done both to avoid vitreoretinal traction induced by instruments passing into and out of the eye, and also to prevent vitreous traction which can bend the tip of the 39-gauge (and smaller) subretinal injection cannula. Most investigators recommend the removal of epiretinal membrane (ERM) if present in the macular area in order to prevent interference with the injection cannula and to avoid late-occurring complications such as macular hole resulting from membrane contraction.
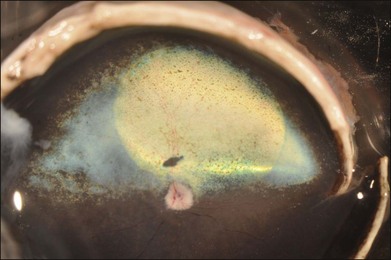
When directing the injection into the posterior pole or macula, the cannula tip is usually placed in the vicinity of the papillomacular bundle. Even in eyes with advanced retinal degeneration, the retina in this area is usually thick enough to allow for successful placement of the cannula tip and the injection into the subretinal space. The cannula tip is positioned so as to avoid direct injury to retinal arterioles. The CHOP protocol specifies that the site of injection be a minimum of 3 mm from the foveal center in order to avoid development of a foveal dehiscence from fluid tracking directly to the central macula in a fistula-like manner. In addition, a small bubble of perfluoro-octane liquid is placed over the fovea to counteract the hydrodynamic force created during the subretinal injection, thereby buttressing this anatomically vulnerable area (Fig. 34.2; video 51 online).
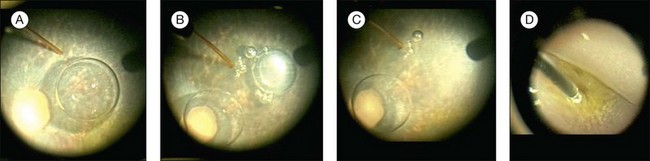
Fig. 34.2 Subretinal injection with protection to the fovea. (A) Subretinal injection cannula is apposed to the retina. Perfluoron had already been layered over the fovea. (B) The injection was initiated. A few small bubbles were expressed initially from the cannula (and are in the subretinal space). (C) The bleb has expanded. (D) The cannula has been removed and the Perfluoron is being removed. The inferior border of the bleb is visible. (See video 51 (online) showing subretinal injection of AAV2-hRPE65v2 in a human.29)
One unique feature of subretinal injection in retinal gene therapy when compared to other surgical indications is that the subretinal fluid, i.e., gene therapy agent, is not evacuated and the retina flattened at the time of fluid–air exchange. This is done in order to maximize the time of exposure to the subretinal injection, something that is not necessary, for example, after the extraction of a subretinal hemorrhage or translocation of the macula. Indeed, the creation of retinal detachment by subretinal injection is of itself a concern with respect to possible retinal toxicity. Fortunately, the extent of injury after acute retinal detachment created by injection of physiologic solutions appears to be small based on both laboratory and clinical data. Resorption of volumes less than 0.45 mL typically occurs in less than 24 hours.16–18,29,35–37 In the CHOP trial, resorption of 0.3 mL of fluid was observed within 6 hours of injection. Bainbridge et al.32 reported a longer period of detachment >24 hours after subretinal injection using a two-step injection technique where vector was administered into a bleb raised first with a physiologic solution. The longer period required for reattachment is likely due to the large volume of injection, i.e., 1.0 mL.32
Development of macular hole is a complication which appears to be unique to the subretinal injection procedure. Although creation of macular hole has been described in subretinal surgery for choroidal neovascularization (CNV), this typically occurs as a CNV is extracted in the presence of an adhesion to the overlying retina. In the CHOP trial, macular hole developed in one patient not during the surgical manipulation but rather several days postoperatively. Although ultimately the ophthalmoscopic appearance of the defect was typical for an idiopathic macular hole, several key differences were noted. First, prior to the development of a full-thickness defect, inner lamellar thinning was demonstrated on optical coherence tomography.18 In addition, intraretinal edema or cystoid macular edema was never present either during evolution of the hole or after hole formation. Finally, a subretinal fluid cuff was never apparent before or after hole formation. In the absence of inflammation, vasculopathy, or acute tissue injury, it was felt that the macular hole was unlikely to have been caused by drug toxicity. The appearance of the lamellar defect was reminiscent of pseudohole caused by ERM contracture. Since the ERM was recognized preoperatively and the posterior cortical vitreous was removed at surgery, it was thought that surgery-induced contraction of the pre-existing ERM was the most likely cause of macular hole formation. A second patient was observed to develop a foveal dehiscence at the time of subretinal injection as fluid channeled directly from the cannula through the fovea through a fistula-like tract.29 This phenomenon has been observed to occur infrequently during subretinal injection in nonhuman primates.38 Occurrence of a foveal dehiscence is presumably induced by intense hydrodynamic stress from injection in the vicinity of the fovea. No ERM was present in this case. Air tamponade with face-down positioning resulted in complete resolution of this induced hole within days. Since instituting a protocol modification specifying a minimum distance from the fovea for injection and use of perfluorocarbon liquid prior to injection, intraoperative foveal dehiscence has not occurred.29
History of retinal gene therapy
Preclinical studies: retinal transduction characteristics of different vectors
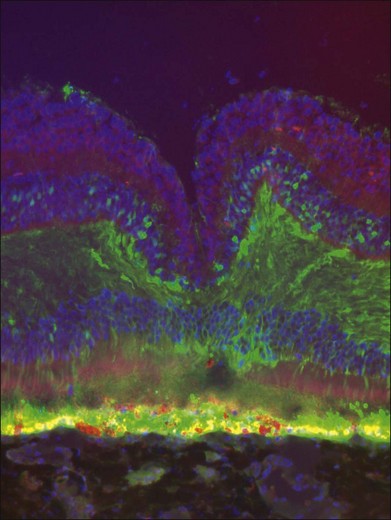
Initial gene transfer studies in animals generally evaluated the safety and stability of delivery of “reporter genes,” genes whose activity in terms of protein products can be evaluated noninvasively or in tissue samples through either histochemical/immunohistochemical measures or through a bioassay. In the early 1990s, these studies often utilized the LacZ (Escherichia coli β-galactosidase-encoding) gene. β-Galactosidase activity can be detected through either histochemical or spectophotometric activity. In the mid to late 1990s, many groups switched to use of green fluorescent protein (GFP), a bioluminescent, intracellular protein normally produced by the jellyfish, Aequorea victoria. A modified version of this protein, enhanced GFP (EGFP), has stronger fluorescence than the original protein, is excited by light in the blue range of the spectrum (498 nm), and has an emission peak at 509 nm, which is in the green portion of the visible spectrum. These absorption/fluorescence characteristics are very similar to those of fluorescein, which is used in the clinic to measure blood vessel integrity in the eye. Thus, EGFP can be measured using the same instruments/optics that are used in the clinic to measure fluorescein (Fig. 34.3). The only difference is that intracellular EGFP does not wash out with time.
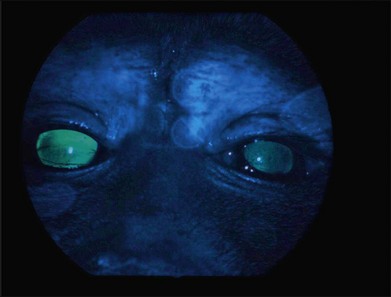
Fig. 34.3 Green fluorescent protein (GFP) is visible through illumination with blue light with an ophthalmoscope in this nonhuman primate that had received subretinal injection of 1E11 vector genomes (vg) AAV2/8.CMV.EGFP in its right eye. The left eye had received subretinal injection of the same material but with a dose that was 2 log units lower than the right eye.39
Evaluation of different vectors
Numerous studies in small and large animals have relied on vector-mediated delivery of reporter genes to elucidate the characteristics of one vector versus another.20–22,39 The transduction characteristics are affected by dose and age (or stage of development) of the animal. In the fetal mouse, one can deliver vector to retinal progenitor cells and carry out “birthday studies” by looking later in adulthood at which types of photoreceptor cells were being “born” at the time of injection.40 Such studies were initially elegantly performed using retroviral vectors, which only target dividing cells.41 Studies on fetal retina AAV-mediated transduction characteristics led to selection of an approach that could be used to reverse blindness in utero in mice.42
The majority of studies published to date involve postnatal delivery. Transduction characteristics are dependent on the surgical approach and the dose. Intravitreal injection of particular recombinant viruses can lead to transduction of ganglion cells and/or Müller glia. For example, intravitreal injection of AAV2 leads to ganglion cell transduction (and expression in the optic nerve, optic chiasms, and brain) in species ranging from mouse to dog to human, whereas intravitreal injection of AAV5 does not.43,44 Intravitreal injection of recombinant viral vectors does not usually result in transduction of the photoreceptors in the outer retina or the RPE (Table 34.1).8,16,17,21
Most vectors target RPE cells efficiently even at low dose (Table 34.1). As the dose is increased and depending on the vector, photoreceptors and Müller cells can also be transduced (Fig. 34.4).39 Cellular transduction characteristics can also differ from species to species. A very active area at present involves engineering of AAV capsids in order to enhance their transduction properties.22,45,46 Efforts are underway to modify the capsid in order to expand its cargo capacity and to modify the capsid to target specific cell types. For example, Greenberg et al. generated lentiviral vectors that target Müller cells specifically after intravitreal injection.47 Recently, some groups have reported that modifications of the AAV capsid will allow it to penetrate from the vitreous through the various retinal cell layers in mice.45 Penetration of that sort has not yet been reported to occur in large-animal models. In smaller animals, it may be an artifact induced by localized retinal detachment produced by the need for a choroidal approach in order to deliver intravitreally or subretinally (see below).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree