CHAPTER 80 Free Tissue Transfer
A multidisciplinary team approach, especially for patients with neoplastic disease, is commonly employed to maximize both the oncologic and reconstructive efforts. Postoperative management is often labor intensive and requires additional team members to achieve maximal rehabilitative potential. Although significant additional training and resources are required to perform free tissue transfer for head and neck reconstruction, the diversity of available tissues for harvest allows for the replacement of “like tissue with like tissue” in most instances, with a focus on functional and cosmetic issues. Currently, more than 40 identified sites in the human body are candidates for the harvest of tissue for transfer, with new sites being added regularly.1 It becomes the reconstructive surgeon’s mandate to determine the optimal flap for free tissue transfer for each patient and the anticipated defect. The advantages of free tissue transfer in the reconstruction of these complex defects include the following:
Specific circumstances exist in which reconstruction using free tissue transfer has become the standard of care in most communities. These include oromandibular composite defects (particularly defects involving the anterior mandibular arch),2,3 subtotal or total pharyngoesophageal defects,4,5 and defects resulting from resection of recurrent carcinoma failing organ preservation nonsurgical therapy (combined chemotherapy and radiation therapy).6 These situations have traditionally been the most challenging in head and neck reconstructive surgery and have been associated with high complication and failure rates. The introduction of free tissue transfer for reconstruction of these defects has dramatically improved the overall course for these difficult patients.
Historical Perspectives
The first clinical application of free tissue transfer was a 1959 case report by Seidenberg and colleagues,7 as a transfer of a free jejunal segment for cervical esophageal reconstruction. In 1961, Hiebert and Cummings8 transferred and revascularized a portion of the gastric antrum for pharyngoesophageal reconstruction. Other reports of the clinical application of free tissue transfer techniques began to appear in the literature in the early 1960s. The first reports of oral cavity reconstruction were made in the early 1970s by Kaplan and associates,10 who utilized a free groin flap, and Harii and coworkers,9 who described the microvascular distant transfer of a deltopectoral flap. Panje and associates11 at the University of Iowa are credited with being the first otolaryngologists to report the use of free tissue transfer for the reconstruction of the oral cavity, in 1976. Subsequent years saw rapid implementation of free tissue transfer in head and neck reconstruction by both plastic surgeons and otolaryngologists. Pioneers such as Panje, Urken, Hayden, and Sullivan worked to establish head and neck microvascular reconstructive surgery as an important part of the specialty of Otolaryngology—Head and Neck Surgery.
Taylor and associates12 reported the first vascularized bone graft using a fibula for reconstruction of long bone injuries in 1975, and Hidalgo13 adapted the osteocutaneous fibula flap for mandibular reconstruction in 1989. Dye-injection studies proved a reliable vascular supply to the ilium from the deep circumflex iliac artery for vascularized bone transfer.14,15 Cadaver dissections identified the vascular branch of the circumflex scapular artery to scapula, allowing vascularized scapula transfer.16 Throughout the 1980s, there was a progressive development and refinement of these and other vascularized bone flaps for oromandibular reconstruction, significantly expanding the availability of this technique.
Indications for Free Tissue Transfer
Free tissue transfer is just one of many options available to the reconstructive surgeon. Well-established methods of primary closure, skin grafting, local flaps, and pedicled fasciocutaneous or musculocutaneous flaps all remain viable and necessary tools in the surgeon’s armamentarium. The indications for free tissue transfer continue to evolve with experience and as additional donor site options have emerged. The etiologies of defects that may require free tissue transfer are listed in Box 80-1. The advantages of free tissue transfer over less sophisticated techniques have likewise become more apparent with greater experience; they are listed, along with the method’s disadvantages, in Box 80-2. The tremendous versatility offered by multiple potential donor sites can test the surgeons’ imagination for new reconstructive methods. The result has been a progressive evolution of new ways to tackle old problems. Certainly, the driving force behind the expansion of free tissue transfer has been the potential for dramatically improved functional outcomes and a remarkable success rate in the head and neck of more than 90%.17–19 For some situations, such as reconstruction of the oral cavity, free tissue transfer offers a significant improvement from a functional and cosmetic standpoint over previously available techniques, particularly for composite defects. In other situations for which no reconstructive options had been available, such as large skull base and scalp defects, free tissue transfer offers excellent reconstructive possibilities. Clinical situations commonly requiring free tissue transfer for reconstruction in the head and neck are listed in Box 80-3.
As with any new surgical procedure or technique, a significant learning curve exists.20 As a result, these procedures have been available primarily in major academic medical centers, and multiple fellowship training programs have been developed to safely convey the necessary knowledge and experience through advanced training. From a medical center perspective, a high level of investment in personnel and equipment is necessary. Anesthesia support is critical, and additional training and experience for dedicated microsurgical operating room nurses are advised as well. The postoperative management of patients undergoing free tissue transfer is certainly more complex, and the demands on personnel are more intensive, in comparison with those for most patients undergoing head and neck surgery. The overall cost of free tissue transfer has been examined and compared with the cost of more conventional (pedicled flap) techniques. The higher operative costs for free tissue transfer resulting from longer operating room times and instrumentation are offset by the significantly shorter lengths of hospitalization. The resulting overall cost of free tissue transfer is either comparable to21 or less than22 the cost of pedicled flap reconstruction.
Donor Site Options
Although more than 40 donor sites for free tissue transfer have been described,1 only a handful have been found to be consistently useful in the routine reconstruction of head and neck defects. The dominant free tissue transfer flaps in use today are shown in Table 80-1. In fact, we have found that the radial forearm fasciocutaneous, radial forearm osteocutaneous, and fibula osteocutaneous free flaps are utilized in more than 80% of cases of head and neck reconstruction. Disa and colleagues23 reported that forearm, fibula, rectus, and jejunum flaps accounted for 92% of free flaps they used for head and neck reconstruction.23 This distribution will certainly vary according to the makeup of the surgeon’s practice as well as the surgeon’s preferences and experiences. Nonetheless, it is important for the reconstructive surgeon to be familiar with a full array of flaps to manage the more difficult or unusual situations. Although several flap options are available for most defects, the surgeon must choose one on the basis of critical characteristics such as those listed in Box 80-4. Additionally, a thorough donor site evaluation and assessment of long-term donor site morbidity must be considered. A complete discussion of each of these flaps is beyond the scope of this chapter; however, some general comments are warranted.
Soft Tissue Flaps
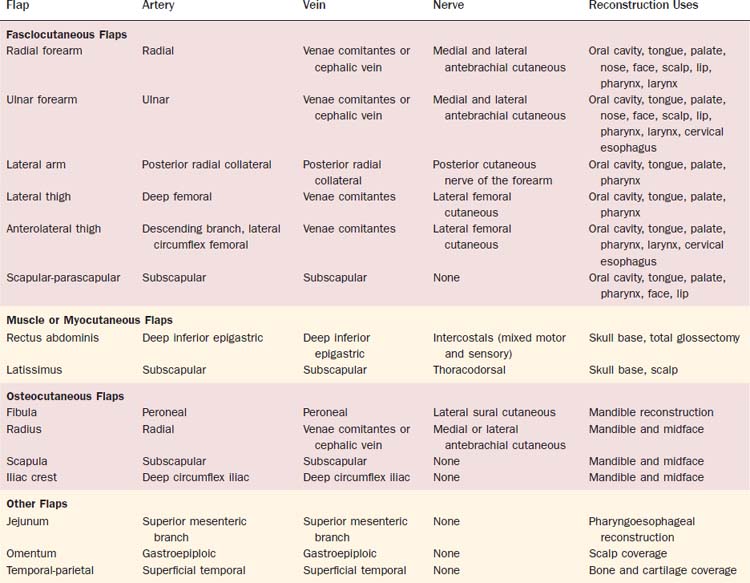
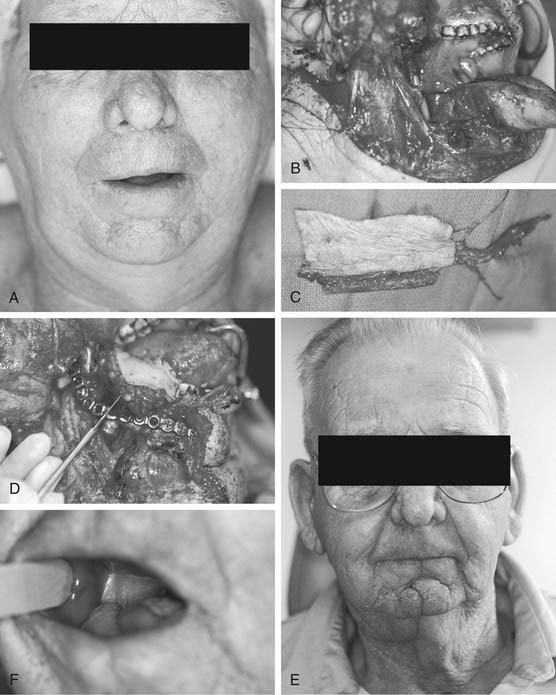
For many cutaneous, mucosal, and soft tissue defects in the head and neck, the radial forearm fasciocutaneous free flap has proved to be the most versatile and reliable. Forearm skin is available in large quantities, is thin and pliable, and has excellent sensory capability that is ideal for oral cavity reconstruction (Fig. 80-1). The vascular pedicle (1) is long, (2) has a favorable vessel caliber, (3) can easily be harvested concurrently, and (4) exhibits acceptable functional morbidity at the donor site. However, as many as 12% of people have an incomplete superficial palmar arch and poor communication between the deep and superficial arches, precluding the safe harvest of the radial artery without causing hand ischemia.
The lateral arm flap is similarly thin and pliable with sensory capability but is limited in skin quantity, has a much shorter pedicle, and has vessels of small caliber.24–29 There is no risk of vascular compromise to the hand, and the donor site can usually be closed primarily. The scapular and parascapular fasciocutaneous flaps produce the largest volume of skin but are quite bulky, have no sensory capability, and have intermediate pedicle length with large vessel caliber.30–34 Although the donor site may be closed primarily, the donor site location (back) for these two flaps requires intraoperative patient repositioning, which precludes simultaneous resection and harvest.
In order to maximize color match, eliminate tissue bulk, and streamline the process of simultaneous flap harvest, other donor site choices, such as the anterolateral thigh flap, have been popularized.35 The anterolateral thigh flap, based on the perforators from the descending branch of the lateral circumflex femoral artery, has been used extensively, mostly in Japanese and Taiwanese centers, as a flap of choice for defects requiring more soft tissue bulk than a radial forearm free flap can provide (see Table 80-1). Peripheral vascular disease is not a contraindication to the use of the anterolateral thigh flap, because the profundus femoris arterial system is not usually affected by atherosclerotic changes. A pedicle length up to 14 cm could be obtained by ligation of the transverse branch of the lateral circumflex femoral artery and the branch to the rectus femoris muscle. This may allow access to the neck vessels contralateral to the defect, if the ipsilateral neck has no suitable vessels. A skin paddle as wide as 12 cm may be harvested, while still allowing primary closure of the donor site. Overall, this flap may allow harvesting of a skin surface area up to 25 cm wide and up to 40 cm long as a single paddle or as multiple skin paddles.36
The two main reasons that the anterolateral thigh flap has been slow in gaining popularity in the United States are the thickness of the subcutaneous tissue of the thigh in the Western population and the highly variable vascular anatomy of the flap. However, the negligible donor site morbidity, primary closure of the donor site, and significant experience gained by microvascular surgeons in harvesting and thinning the flap have led to popularization of the anterolateral thigh flap in many U.S. centers.35–39 Some writers describe using a handheld Doppler probe to identify perforators preoperatively, before designing the skin paddle.37
Muscle and Myocutaneous Flaps
Muscle and myocutaneous flaps have found a niche in the reconstruction of very large defects that require significant bulk. The most common of these defects are found at the skull base, where large portions of the facial skeleton, paranasal sinuses, facial skin, and palate may be missing. The rectus abdominis flap may be harvested as a muscle or myocutaneous flap with the patient in a supine position, a long vascularized pedicle of large caliber vessels providing adequate reach to the defect site in most instances.40–44 The skin of the rectus myocutaneous flap is useful for lining the nasal and oral cavities of the defect, and the muscle provides the needed bulk to replace the missing facial skeleton. Donor site morbidity is minimal, although abdominal hernias may occur as a result of a weakening of the abdominal wall due to the partial harvest of the anterior rectus sheath.
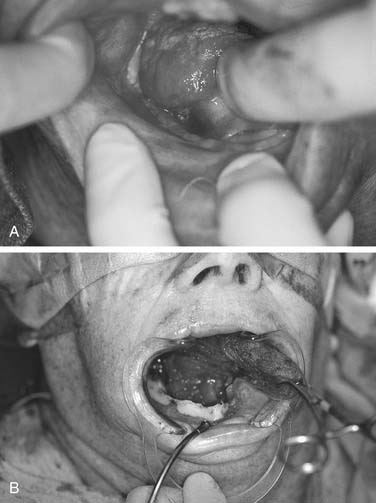
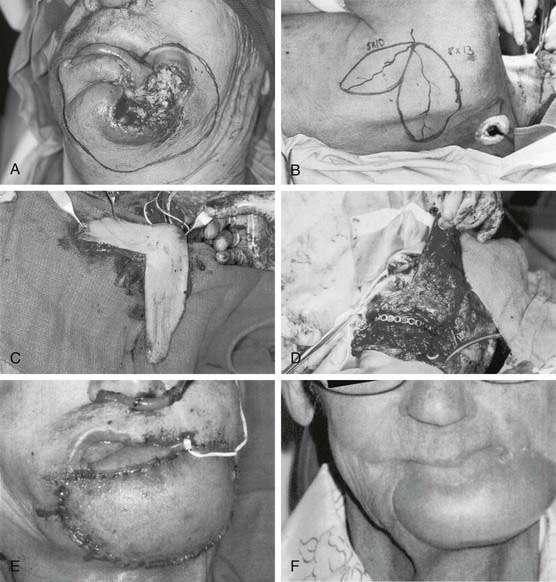
The latissimus flap may be harvested as a muscle or myocutaneous flap based on the subscapular vascular system.45,46 Originally described as a pedicled flap, the latissimus flap has been found to be much more versatile as a single-stage, free tissue transfer. The large amount of muscle available for harvest is very useful for very large soft tissue defects, including the skull base and scalp. The subscapular system has additional versatility, with the option for vascularized bone (scapula bone or rib). The vascular pedicle has large vessel caliber and a length of 6 to 10 cm, and the donor site is repaired primarily without the need for a skin graft. The major disadvantage of the latissimus flap is patient positioning for harvest. As for scapula flaps, the patient must be placed in the lateral decubitus position, making harvest concurrent with the head and neck procedure difficult or impossible. The donor site morbidity from the loss of latissimus function is well tolerated in most people. The latissimus flap has been most useful in skull base defects and larger scalp defects requiring cranial coverage (Fig. 80-2).43,47,48
Vascularized Bone Flaps
Osteocutaneous Fibula Flap
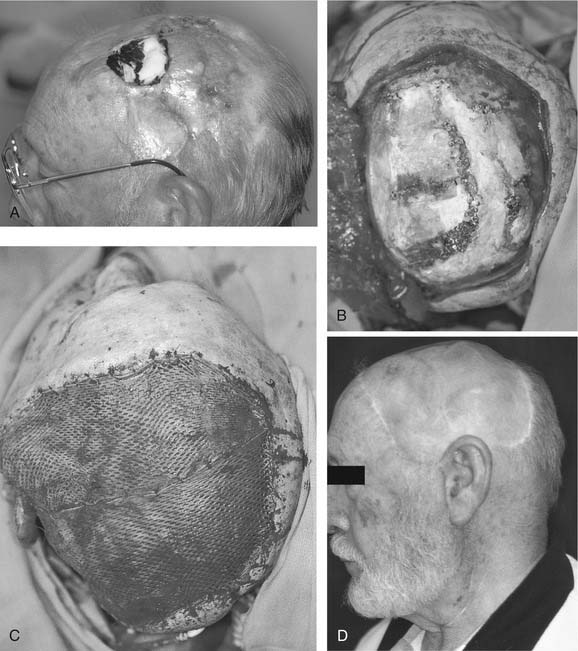
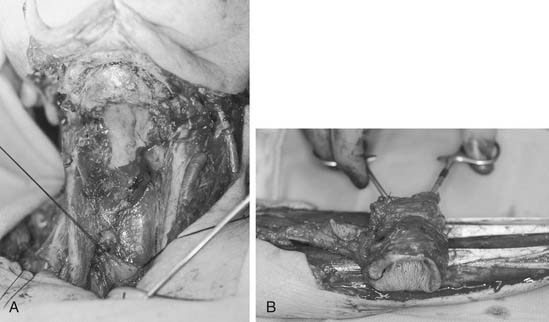
The osteocutaneous fibula free flap has become the mainstay for mandibular reconstruction at most institutions (Fig. 80-3).49–51 The fibula is touted as “the most donateable bone in the body,” with up to 25 cm of bone available for harvest and adequate bone stock to support dental implantation.52,53 With such lengths of bone available, the entire mandible may be reconstructed with vascularized bone if required. Multiple osteotomies may be performed to shape the fibula to reconstruct the anterior arch, body, angle, or ramus of the mandible, as long as the fibular periosteum is not disrupted. The septocutaneous or musculocutaneous perforating vessels of the lower leg can be quite variable in location and quantity, affecting the placement and reliability of the skin paddle.54–56 The skin of the lower lateral leg is thin and pliable, with fairly large amounts of skin available, and may be transferred in a sensate fashion. With smaller skin paddles, the donor defect may be closed primarily. With larger defects, a split-thickness skin graft is utilized. The fibula flap harvest can easily be performed simultaneously with the head and neck procedure.
Confirmation of three-vessel flow to the distal lower extremity should be determined preoperatively to avoid vascular compromise of the foot after harvest of the peroneal artery.57,58 The primary disadvantage of the fibula flap is the limitations of the skin paddle. The skin is inadequate for larger soft tissue defects and most three-layer defects, requiring a second flap for soft tissue repair. The somewhat unreliable nature of the presence and location of the cutaneous perforator vascular supply can usually be minimized by inclusion of a cuff of soleus muscle with the flap, to include musculocutaneous perforators. If dental implants are not planned, use of the fibula bone results in a very broad and rounded neomandible, which is quite difficult to fit for a tissue-borne prosthesis. The donor site morbidity of the fibula flap involves prolonged pain on ambulation for some patients.59–63
Osteocutaneous Radial Forearm Flap
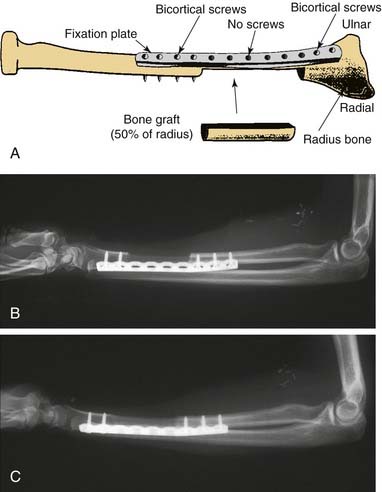
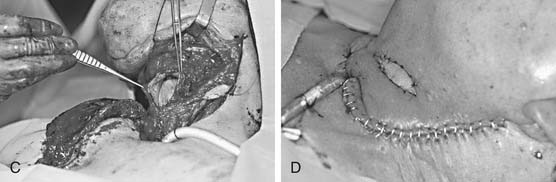
The radial forearm free flap (RFFF) has also been described as an osteocutaneous radial forearm free flap (OCRFFF), with harvest of a portion of the radius bone based on perforators in the intermuscular septum passing to the periosteum.64–66 This addition significantly broadened the applicability of the already widely used forearm flap in reconstructive surgery. Although the OCRFFF is seemingly the best of options, with tremendous soft tissue characteristics and an option for bone harvest, its widespread acceptance has been limited by concerns about the bone quality and possible pathologic fracture of the radius bone postoperatively.
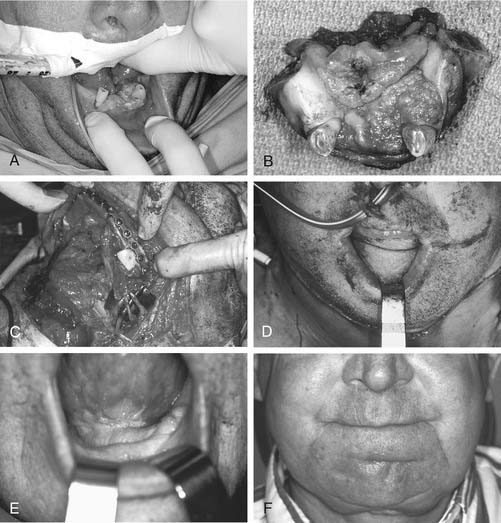
The length of radius bone that can be safely harvested without unacceptable forearm dysfunction is limited to 10 to 12 cm (Fig. 80-4). To avoid pathologic fracture, most surgeons recommend that the thickness of the bone harvested be limited to 40% of the circumference of the radius.67,68 This thickness generally does not provide adequate bone stock to support endosseous dental implants.52,53 When a segment of bone is removed from the radius, the bone is significantly weakened, especially to torsional forces; the weakening previously resulted in a postoperative pathologic radius fracture rate of up to 66%, with an average of 23%.68–71 The prophylactic internal fixation of the radius bone after OCRFFF harvest has been shown to successfully eliminate this risk (Fig. 80-5).72,73
Despite the limitations of bone availability with the OCRFFF, it has been used successfully for oromandibular reconstruction, with fewer complications than those associated with the fasciocutaneous RFFF with plate reconstruction.68,74–76 For limited mandibular defects, the radius bone is quite adequate and can easily bear a tissue-borne prosthesis (denture). This characteristic has proved beneficial, because many patients do not have the financial means for dental implantation, which is frequently not covered by many third-party payers. In this setting, the radius bone provides a better contour for the support of a tissue-borne prosthesis than either the fibula or scapula bone.
Reconstruction of maxillofacial defects is a challenging problem, owing to the complex three-dimensional, esthetic, and functional roles of the midfacial structures. Cordeiro and Santamaria77 presented a classification of maxillofacial defects and suggested an algorithm for their reconstruction. The combination of a large skin paddle to line the palatal mucosal surface, small volume of the soft tissue, and moderate vascularized bone stock are the requirements fulfilled very well by a reconstruction with OCRFFF. The radial forearm free flap is ideal to solve one of the most problematic aspects of choosing the flap for a midfacial reconstruction: a need for a long (10-13 cm) pedicle to span the distance between the midface and cervical vessels. Using an OCRFFF eliminates the need for vein grafts, shortening operative time and improving the success of the reconstruction. Chepeha and colleagues78 expanded the use of the OCRFFF for maxillectomy defects with infraorbital rim involvement in ten patients. They advocate the choice of OCRFFF for total maxillectomy defects, with or without the orbital exenteration, that involve less than 40% of the orbital floor. Rather than using the vascularized bone to reconstruct the malar alveolar ridge, these researchers employed osteotomized radial bone to recontour the infraorbital rim and to provide support for the orbital contents when preserved. Dental rehabilitation was achieved through the use of palatal obturator and dentures.78
Osteocutaneous Scapula Free Flap
The osteocutaneous scapula free flap remains one of the most versatile flaps available for harvest in the human body.30–32,34,79,80 The subscapular arterial system offers tremendous amounts of skin as well as the latissimus dorsi and serratus anterior muscles and scapular bone for harvest, all supplied by one major vascular pedicle, which is favorable in both length and caliber. Each of these different tissue components has a separate vascular branching supply, allowing for almost limitless degrees of orientation in relation to one another and to the recipient bed. The skin of the upper lateral back is usually quite thick, with considerable subcutaneous fat, resulting in significant soft tissue bulk, which can be advantageous in some reconstructive situations. This bulk reliably remains on a long-term basis, without atrophy. The skin can also be separated into scapular and parascapular skin paddles based on the transverse and descending branches of the circumflex scapular artery, respectively.31,32,34 Inclusion of the dorsal cutaneous rami of the T1 or T2 spinal nerves into the flap, which may allow for sensate transfer of scapular cutaneous paddles, has been described.81 A total of 10 to 14 cm of bone is available for harvest from the lateral border of the scapula, supplied by the periosteal branch of the circumflex scapular artery. The separation of the bony and fasciocutaneous components of the flap resulting from unique vascular supplies can be as much as 4 cm, allowing for significant versatility in hard and soft tissue orientation during reconstruction. Osteotomies may be safely performed for mandibular contouring, as long as the periosteum is preserved. The harvested scapular bone has a thick border along its free edge but transitions quickly to thin (1 to 2 cm) bone medial to the edge. The resulting limited bone stock may not be adequate for the support of endosteal dental implants.52,53
The scapular osteocutaneous flap remains an optimal choice for the reconstruction of the oromandibular complex when the surgeon is faced with large complex defects, especially those involving a large surface area, or with three-layer composite defects involving both the oral cavity and external soft tissues (Fig. 80-6).
For extremely large defects, some surgeons have employed the scapular “mega-flap,” which includes not only the scapular bone and extensive skin as described earlier but also the latissimus dorsi muscle or the serratus anterior muscle for additional bulk and coverage.82,83 The muscles are based on the thoracodorsal artery and vein that branch off the subscapular vessels, and therefore can be harvested on the same vascular pedicle, requiring only one arterial anastomosis and one venous anastomosis. The mega-flap offers significant mobilization of the various tissue components relative to one another as a result of the branching vascular supply, providing great reconstructive versatility for the largest of defects.
Iliac Crest Flap
The iliac crest has long been a source of free bone grafts for mandibular reconstruction. A vascularized iliac crest flap harvested off the deep circumflex iliac artery may provide 14 to 16 cm of bone, which has a natural curvature for mandibular reconstruction. Studies have found that, of the available osteocutaneous flaps, the iliac crest provides the highest quality bone for the support of osseointegration for dental implantation.52,53
The cutaneous portion of the flap is both bulky and limited in mobility, restricting its usefulness for large composite defects. Urken and colleagues84–87 described the addition of the internal oblique muscle based on the ascending branch of the deep circumflex iliac artery for mandibular reconstruction, which significantly increased the versatility of the flap and provided excellent results.84–87
Dental Rehabilitation
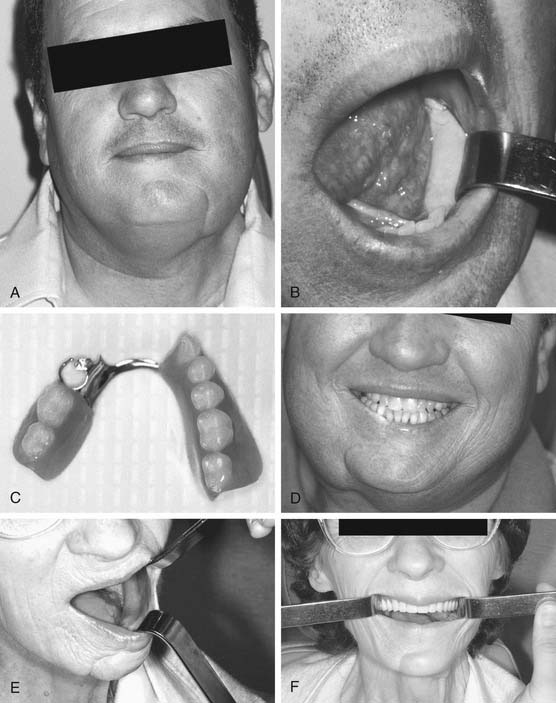
Significant dental rehabilitation of the oral cavity after composite resection for malignancy can be achieved only with microvascular free tissue transfer of bone and soft tissue (Fig. 80-7). The placement of enosseous dental implants into the replaced bone allows for the optimal rehabilitative outcome that approaches function in the patient with normal dentition.84,88 Appropriate contouring of the reconstructed oral cavity may allow the fitting of tissue-borne dentures. Although this reconstruction is generally cosmetically acceptable for most patients, the diet remains significantly affected.
Other Flaps
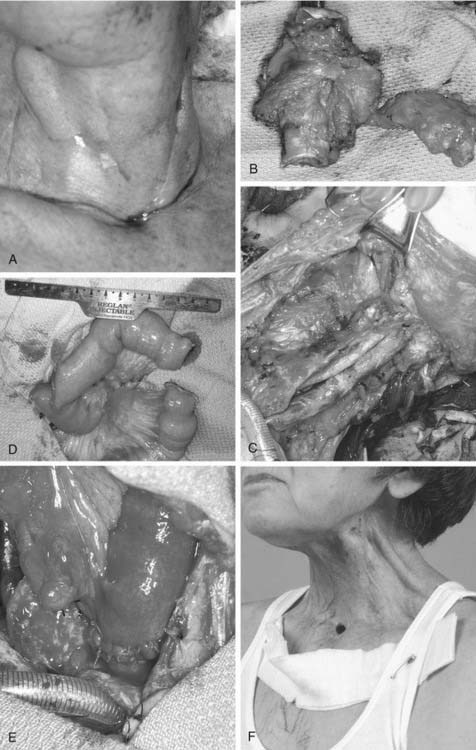
Unique situations that require the employment of less frequently utilized flaps often arise. Pharyngoesophageal defects have long presented one of the more challenging reconstructions in head and neck surgery. This is reflected by the first reported free tissue transfer in the head and neck, described by Seidenberg and colleagues7 in 1959 as a transfer of a free jejunal segment for cervical esophageal reconstruction. The jejunal flap may be harvested off a segmental arcade of the superior mesenteric artery, for free transfer to the head and neck. As a mucus-producing tubular structure of similar size, the jejunum seems a natural replacement for the cervical esophagus. On transfer, the jejunum retains some intrinsic contractile motility, which is beneficial for swallowing (and detrimental if the jejunum is inverted on the inset or placement into the defect) (Fig. 80-8).89–92 Through the years, experience has found the jejunum to be a fairly fragile flap that does not tolerate episodes of ischemia. The donor site morbidity of a bowel resection can be considerable. The reconstructed pharynx can successfully undergo tracheoesophageal puncture89,93; however, this maneuver often results in a very “wet” vocal quality. As a result, most surgeons now routinely use a radial forearm fasciocutaneous free flap, which is “tubed” to recreate the cervical esophagus (Fig. 80-9). This flap results in less donor site morbidity, provides a very functional adynamic “funnel” into the thoracic esophagus, and produces a better tracheoesophageal puncture vocal quality.94
Patient Evaluation and Preparation
History and Current Illness
Unfortunately, smoking and chewing tobacco are prevalent among patients with head and neck cancer. Beyond its ill effects on anesthesia and cardiopulmonary function, nicotine also has adverse vasoconstrictive effects that can compromise flap perfusion and wound healing.95,96 A history of regular alcohol use requires aggressive perioperative withdrawal prophylaxis and nutritional supplementation. Acute withdrawal after surgery is associated with a higher rate of complications and flap loss.97,98
A history of previous radical neck dissections may limit the current reconstructive options at both recipient and donor sites. Careful review of old surgical reports, review of current radiographs, and discussions with previous surgeons is often necessary to supplement the patient’s memory. Similarly, extensive radiotherapy can compromise recipient site vascularity and healing and cause premature vascular wall changes, but may or may not affect free flap viability.99–101 Post-irradiation hypothyroidism should be detected and reversed to help maximize postoperative recovery and healing. Both previous surgery and prior radiotherapy can cause extensive fibrosis in the surgical bed, making safe isolation of needed recipient vessels and nerves very difficult, if not impossible.
Physical Examination
Microvascular free tissue transfer has been used with success in the elderly. Chronologic age itself does not seem to contraindicate surgery, although the incidence of medical complications is higher after treatment.18,102,103 Mental impairment, such as dementia or mental illness, is a very difficult situation to control in the debilitated postoperative patient. Serious consideration should be given to more tolerant alternative reconstructive techniques if surgery is still considered an option in a mentally impaired patient. Morbid obesity not only may influence perioperative recovery and healing but also may require alteration in available useful donor sites because of flap thickness and bulk.104 Morbidly obese patients may also have undetected significant obstructive sleep apnea, which heavily influences perioperative airway management and can also be seriously exacerbated postoperatively by large, bulky, and swollen reconstructions. Potential airway compromise due to the bulky or edematous flap should be anticipated and should be prevented by the intraoperative tracheotomy.
Examination of the potential free flap donor site for scars, asymmetries, or bony deformities can often prompt a patient’s memory of an old injury or operation. Objective evaluation of the mobility, strength, and function of an extremity should confirm the patient’s history. Preoperative disabilities, from previous strokes, spinal injury, or trauma, may either prompt or discourage the use of certain free flap donor sites. Nonhealing sores, cold digits, loss of sensation, as well as significant distal extremity swelling are also indicators of inadequate vascularity and commonly contraindicate the use of the involved extremity as a donor site. Palpation of distal extremity pulses (i.e., radial, ulnar, dorsalis pedis, and posterior tibial arteries) as well as the performance of subjective Allen’s tests and determination of the ankle-arm index can be reassuring, but adequacy determinations should be supplemented by formal vascular assessments in equivocal cases.105–107
Past Medical History
In all patients, special attention should be paid to optimization of any known medical conditions. Certain comorbid conditions can also affect free tissue transfer specifically, although absolutely contraindicating conditions are uncommon.108 Certain extremity vessels are known for a tendency toward advanced atherosclerotic disease in patients with peripheral vascular disease. Radial and peroneal arteries tend to be affected frequently, but the scapular arterial system remains relatively unaffected. Poor digit perfusion can be further exacerbated by donor vessel sacrifice, yielding cold intolerance or even digit necrosis. Patients with severe extremity venous or lymphatic insufficiency should also be considered for other donor sites to prevent postoperative extremity complications. Patients with arterial vascular disease due to atherosclerosis or hypertension must also be evaluated for significant concomitant cerebrovascular and cardiovascular disease. In the patient with significant carotid disease, availability of adequate recipient external carotid artery branches may be reduced; also, intraoperative hypotension or neck extension positioning may cause perioperative strokes. Adequate cardiopulmonary reserve must be present to tolerate the often prolonged anesthesia time and intravascular fluid shifts associated with free tissue transfer operations.109 Such comorbid disease can significantly lower the limits of resectability and reconstructability, because such procedures often require more prolonged general anesthesia and possibly even multiple trips to the operating room.
Patients who have connective tissue disorders with vasculitis are at a relatively high risk for microvascular pedicle compromise, which therefore should be a relative contraindication to microvascular reconstruction. Unfortunately, patients with well-controlled disease who have been aggressively treated with steroids or other immunosuppressive agents can experience poor healing or increased infectious complications.110 Similarly, patients with hematologic disorders (i.e., von Willebrand’s disease, hemophilia, lupus anticoagulant abnormality, thrombocytopenia, polycythemia vera, sickle cell disease, protein C deficiency) should also be strongly considered for nonmicrovascular reconstructions because of the difficulties in maintaining healthy patent anastomoses associated with prothrombotic and coagulopathy states.111,112 Paraneoplastic hypercoagulability or extensive inadvertent preoperative use of anticoagulating medicines (i.e., aspirin, nonsteroidal anti-inflammatory drugs, vitamin E) can also compromise microvascular success. Patients who require perioperative anticoagulation because of concurrent conditions (i.e., prosthetic heart valves, deep venous thrombosis) are predisposed to compressive neck hematomas that can lead to significant blood loss, airway compromise, or flap pedicle compression.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree