Eye Movement Characteristics and Recording Techniques
Louis F. Dell’Osso
Robert B. Daroff
The day of the last hypothesis would also be the day of the last observation.…An hypothesis which becomes dispossessed by new facts dies an honorable death; and if it has called up for examination those truths by which it is annihilated, it deserves a moment of gratitude.
Jacob Henle (1809–1885)
In foveate animals, the purpose of eye movements is to bring visual stimuli into the peripheral field of vision (peripheral retina) to the central point of best visual acuity (fovea) and to maintain foveal fixation of a moving object. The acquisition (gaze shifting) and securing (gaze holding) of stationary object images on the fovea and the stabilization of images on the fovea during head movement (gaze holding) or target movement (gaze shifting) constitute the basic functions of human eye movements. Although many specific types of eye-movement abnormalities require sophisticated recording and analysis techniques, there are clinical tests that, when properly applied, can provide valuable information about diagnosis, pathophysiologic mechanism, or response to therapy.1
PHYSIOLOGIC ORGANIZATION
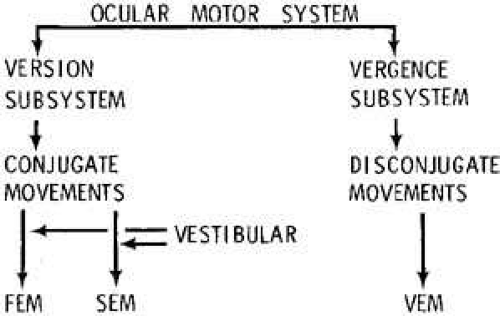
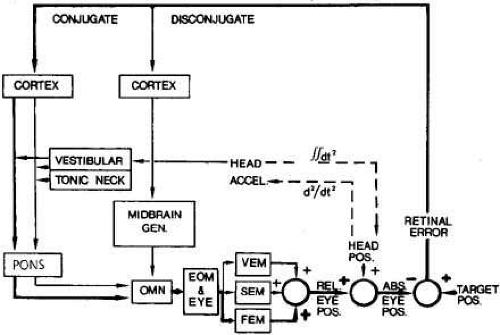
The ocular motor system can be conceptualized as two independent major subsystems, version and vergence, acting synergistically (Fig. 1).2 The version subsystem mediates all conjugate eye movements, whereas the vergence subsystem mediates all disjugate eye movements. Fixation and vestibuloocular inputs influence the version subsystem. At the most peripheral level, regardless of input, there are only three major categories of eye movement output: fast eye movements (FEM or saccades) and slow eye movements (SEM) from the version subsystem and vergence eye movements (VEM) from their own subsystem. All three outputs share a common neural pathway from the ocular motor neurons to the muscles (Fig. 2). In addition, the version subsystems share a common neural network that integrates (mathematically) velocity information into position signals. The fast mode of the version subsystem mediates all conjugate saccades (FEM), and the slow mode mediates all SEM. The latter includes, but is not limited to, the pursuit function. Without knowledge of the conditions that were used to elicit a particular response, one could not differentiate (1) the eye movement record of a voluntary saccade from a nystagmus fast phase or (2) the record made by pursuit of a slowly moving target from that of slow rotation of the subject while fixating a stationary target. The many terms used to describe eye movements generally specify the eliciting input, the functional subsystem, or the circumstance of occurrence, but the eye movements themselves consist of one or more of the three main outputs (FEM, SEM, VEM) of the ocular motor system (Table 1).
TABLE 1. Eye-Movement Classifications | ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||
There is ample physiologic, anatomic, and clinical justification for regarding the subsystems as autonomous. However, the neurons within the oculomotor, trochlear, and abducens nuclei are not specific for types of eye movement. Rather, different firing patterns of homogeneous neuronal pools determine the type of eye movement.3
The simplified schema described previously, which uses the three unique ocular motor outputs as a basis for conceptualization of the ocular motor system, is used in this introductory chapter for purely pedagogic reasons. Our approach to the ocular motor system has undergone continuous refinement since its inception in 19742 and has served as the introduction to ocular motility for many medical students, ophthalmology and neurology residents and fellows, biomedical engineering undergraduate and graduate students, and postdoctoral fellows. We have attempted to present an overview of the functional mechanisms thought to comprise the ocular motor system with minimum inclusion of putative anatomical sites or physiological mechanisms. This chapter should provide clinicians with an basic understanding of this complex motor system and students of ocular motility with a solid foundation on which to expand their knowledge base. For the latter, we recommend these textbooks containing the anatomy, physiology, and functional mechanisms of ocular motility in greater detail.4,5,6
If one used inputs rather than outputs as a basis for conceptualizing the ocular motor system, the latter could be divided into additional subsystems separated by phylogenetic origins and physiologic modes of action. From an evolutionary point of view, the vestibular subsystem probably developed first, closely followed by the optokinetic and saccadic subsystems; the latter are required to generate reflex fast (quick) phases associated with passive head movement and “afoveate” saccades for active head movement. With the development of a fovea came subsystems for fixation, pursuit, and voluntary saccades and finally the vergence subsystem for binocular single vision and stereopsis. Because the neurophysiologic substrates and varied purposes of these subsystems result in specific properties and limitations, their origins and individual modes of action are key to a complete understanding of the ocular motor system and are especially important if one wishes to study them in situ or with the use of computer models. Observations indicate that a distinct subsystem may mediate the SEM of fixation in synergy with the saccadic and pursuit subsystems. Studies of the latter7 and of human congenital nystagmus8,9,10 have provided evidence in support of a separate fixation subsystem. Some of the quantitative characteristics of a fixation or “stabilization” subsystem have begun to be elucidated.11,12,13 The different inputs, outputs, and components of these subsystems are discussed in the sections of this chapter dealing with the major output subsystem to which they belong (i.e., FEM, SEM, or VEM).
FAST EYE MOVEMENTS (SACCADES)
Fast eye movements are rapid version (conjugate) eye movements that are under both voluntary and reflex control. Examples of voluntary saccades are willed refixations and those in response to command (e.g., “Look to the right…Look up.”). The sudden appearance of a peripheral visual object or an eccentric sound may evoke a reflex saccade in the direction of the stimulus. In the natural state, a head movement in the same direction usually accompanies these saccades. However, in clinical examinations and in most physiologic experiments, the head is stabilized.
The visual stimulus for FEM is target (object) displacement in space. After an instantaneous change in target position, the ocular motor system will respond with a FEM after a latency (delay) of 200 to 250 milliseconds. Both the peak velocity and the duration of FEM are dependent on the size (amplitude) of eye movement, which varies from 30°/second to 800°/second and 20 to 140 milliseconds, respectively, for movements from 0.5° to 40° in amplitude. FEM are conjugate and ballistic. The control system responsible for their generation is discrete. At discrete instants in time, control decisions are made based on the continuous inflow of visual information from the retina. In normal persons, these decisions are essentially irrevocable; once the eyes are in motion, their trajectory cannot be altered. The control signal is retinal error (disparity of image position from the fovea), which is automatically reduced to zero by the nature of negative feedback.
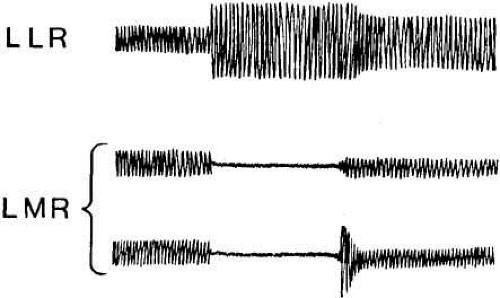
After the appropriate latency, the FEM response to target displacement (Fig. 3) consists of a period of acceleration to a peak velocity and then deceleration of the eyes as they approach the new target position. The muscular activity in the agonist–antagonist pair of each globe is characterized by a burst of maximal facilitation in the agonist and total inhibition in the antagonist during the movement (Fig. 4). Electromyographic (EMG) recordings reveal that FEM deceleration is usually not consequent to active braking by the antagonist muscle. Rather, the two muscles merely assume the relative tensions necessary to hold the new target position. This is sufficient to accomplish the rapid deceleration because of the braking effect (damping) of the “ocular motor plant” (i.e., globe, muscles, check ligaments, and fatty supporting tissue of the orbit). EMG recordings have identified active dynamic braking in the antagonist muscles for some saccades. The active braking seems to be associated more often with small saccades than with large saccades. Occasionally a saccade is of such magnitude that it overshoots the target, and a saccade in the opposite direction follows it without latency; this is called a dynamic overshoot. Dynamic overshoots are common in both voluntary and involuntary (e.g., the fast phases of nystagmus) saccades. There is also evidence that with an unrestricted head, intersaccadic latencies may be reduced.14
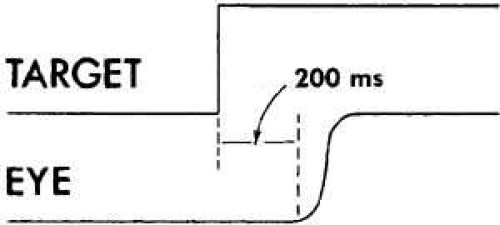 Fig. 3 FEM response to a rightward target displacement illustrating the latency (200 msec) and trajectory of the FEM (saccade). |
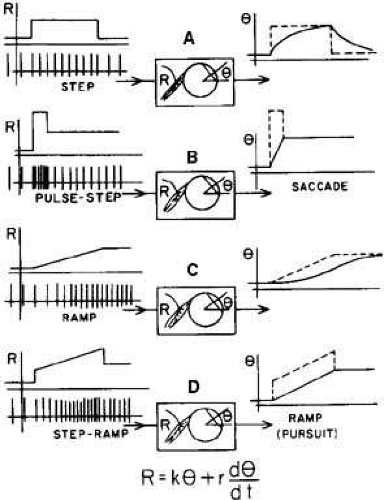
The overdamped plant (mechanical resistance of orbital structures) requires that the neural signal necessary to achieve the rapid FEM acceleration must be a high-frequency burst of spikes, followed by the tonic spike frequency required to stop and then hold the eyes at the new position. This combination of phasic and tonic firing patterns is designated the “pulse-step” of neural innervation (Fig. 5). The eye movement in Figure 5A results from a step change in neural firing frequency and, reflecting the overdamped plant dynamics, is considerably slower than a normal FEM. A normal FEM trajectory occurs only when a pulse precedes the step (see Fig. 5B). A neural “pulse generator” and “integrator” combine to form the required pulse-step of innervation (Fig. 6). The pulse generator consists of burst cells, whose activity is normally inhibited by pause cells (see Fig. 6). When the pause cells cease firing, the burst is turned on, and the duration of its high-frequency pulse of innervation is determined by a feedback circuit that contains a second, resettable neural integrator.15 This resettable neural integrator, within the pulse generator, feeds back a signal that simultaneously turns off the burst cells and reactivates the pause cells. The resettable neural integrator of the pulse generator is not the same as the common neural integrator used to generate the tonic innervation levels sent to the ocular motor nuclei. Because there are pathologic conditions (e.g., gaze-evoked nystagmus) that affect the ability of the eyes to maintain gaze but do not alter the trajectory of saccades, two separate neural integrators seem to be required: a resettable integrator within the pulse generator that functions to set pulse width, and a second, common integrator that is responsible for generating the constant level of tonic innervation required to maintain gaze.15,16 This hypothesis of normal saccade generation was supported by ocular-motility studies of common human clinical conditions. Almost a decade passed before neurophysiologic studies in animals provided additional supportive evidence.17 The pulse generator for horizontal eye movements is located within the pontine paramedian reticular formation (PPRF) at the level of the abducens nuclei, specifically, in the nucleus pontis caudalis centralis (see Chapter 10, Fig. 4).18 Vertical burst neurons are located in the rostral interstitial nucleus of the medial longitudinal fasciculus (MLF). The superior colliculus is intimately involved in the generation of saccades, especially short-latency “express” saccades.19 Projections to the superior colliculus come from the frontal, parietal, and occipital cortex; projections from the superior colliculus go to areas in the brainstem reticular formation containing the saccadic burst and omnipause neurons. The superior colliculus appears to contain signals equivalent to motor error and may include the resettable integrator.20,21,22,23,24,25 The existence of the resettable neural integrator has also been questioned.26 Thus, despite the concerted efforts of several groups, the location of the resettable neural integrator or its equivalent remains a topic of speculation and competing hypotheses 30 years after it was first hypothesized. The horizontal common integrator may be located in the nucleus prepositus hypoglossi, the medial vestibular nucleus, and possibly other (cerebellum) locations. The vertical integrator is probably in the interstitial nucleus of Cajal.27 The location of the summing junction for the pulse and step is uncertain but must be prenuclear with respect to the third cranial nerve because MLF axons carry neural information that is already summated (pulse plus step).28 Both burst neurons (pulse) and tonic neurons (step) project to an area of the nucleus of the abducens nerve, where intranuclear interneurons project to the nucleus of the oculomotor nerve via the MLF. Thus, the summing junction is probably in the area of the nucleus of the abducens nerve.
Because saccades are not always accurate and their trajectories are not always normal, a scheme has been devised to describe both their metrics and trajectories. The pulse-step of innervation necessary to make a saccade is used to define what is meant by orthometric, hypometric, or hypermetric eye movements. The final gaze position that the eye assumes (after the effects of both the pulse and step) is used to measure saccadic accuracy. The step determines metrics, and the relationship between the pulse and step determines the trajectory (i.e., the way in which the eye arrives at its final position). Saccades may be either orthometric, hypometric, or hypermetric and may have numerous trajectory variations. The latter have been identified as normal, slow, overshoot, undershoot, dynamic overshoot, discrete decelerations, and multiple closely spaced saccades. A complete description of a particular saccade must include both metrics and trajectory; a refixation may include several saccades of varying metrics and trajectories. A thorough discussion of saccadic metrics along with a recursive shorthand notation for metrics and trajectories may be found in Schmidt et al.29 All the possible departures from the norm of the saccadic system were derived from and illustrated in Schmidt’s article on myasthenia gravis.
Other factors may influence the speed of saccades; both attention and state of convergence can play a role. Saccades made under conditions of increased demand for accuracy are slower than normal.30 This has been found to be associated with increased co-contraction of the extraocular muscles, presumably increasing the stiffness of the plant, both statically and dynamically.31 The discovery of fibromuscular “pulleys,” through which the extraocular muscles pass, provides a mechanism by which this can be accomplished.32 These compliant pulleys are under active control and can change the effective moment arm of the muscles, thereby altering the dynamics of the resulting eye movement.
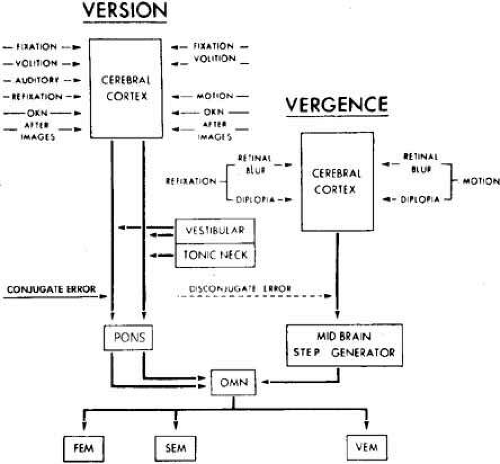
The closed-loop nature of the FEM mode of the version subsystem can be depicted in a simplified block diagram (Fig. 7) containing only primary signal paths and lacking the paths containing the control signals that appear to be necessary to simulate known ocular motor responses of both normals and those with dysfunction due to either congenital or acquired conditions. The conjugate retinal error signal, representing a discrepancy between target and eye position, is sensed in the cerebral cortex. Signals derived from this information are used in the brain stem to generate the neural command to the ocular motor neurons necessary for the FEM, which moves the eye to its new position, thereby reducing the retinal error to zero (foveal fixation).
The FEM subsystem can be modeled as a discontinuous or, more specifically, a sampled-data control system in which visual information is used during sample intervals (intermittent sampling). Between samples, new visual information, although perceived, cannot be used to modify any eye movement decisions. The study of patients with pathologically slow saccades has revealed that under these conditions it is possible to modify a saccade in flight based on new visual information.33 A detailed presentation of the control-system analysis of the various types of eye movements is beyond the scope of this chapter.
Rapid eye movements (REM) of paradoxical sleep and the fast phases of evoked (vestibular, optokinetic) or pathologic nystagmus are also examples of saccadic eye movements. These saccades and those of refixation share the same physiologic characteristics.
During a saccade, the visual threshold is elevated about 0.5 log units (saccadic suppression). This phenomenon is controversial; some investigators postulate an active central inhibitory process,34 whereas others35 favor a retinal-image “smear” mechanism. In either case, the relatively small visual threshold elevation cannot account entirely for the subjective sense of environmental stability during saccades. A mechanism, designated “corollary discharge” or “efference copy,” in which the visual system is “altered” centrally (by way of fronto-occipital connections) for forthcoming retinal image movement probably serves to cancel conscious perception of environmental motion during a saccade.36
Plasticity
The saccadic system, as well as other ocular motor systems, is plastic (i.e., its gain is under adaptive control based on feedback signals that monitor its performance). Although saccades are programmed in the brain stem, their size is controlled by means of cerebellar circuits, and it is these circuits that change saccadic gain in response to neurologic deficits. By alternately patching one eye in a patient with a third nerve palsy and studying the gain of the saccadic system as it varied with time, Abel et al.37 documented the plastic gain changes in the saccadic system and measured the time constants of this adaptation. The time constants were found to be on the order of 1 to 1.5 days; both the duration of the innervational pulse and the magnitude of the step were adjusted independently.
One of the ways in which the cerebellum is thought to make parametric adjustments in the saccadic system is by varying the amount of position information fed back to the input of the common neural integrator (Fig. 8). Because this neural integrator is an imperfect one (i.e., it cannot hold its output without a decay in the signal, referred to as a “leak”), the gain (Kc) of the position feedback is adjusted to overcome its inherent leakiness. By using eye-position feedback, the cerebellum evaluates the performance of the common neural integrator, and adjustments in Kc are made. Problems either in the neural integrator itself or in this parametric adjustment circuitry can cause various types of nystagmus. If Kc is too small, the inherent leakiness of the neural integrator will cause the eyes to gradually drift back toward primary position from any eccentric gaze position. If Kc is too high, the eyes will accelerate centrifugally away from the desired gaze position with an ever-increasing velocity until reset by a centripetal saccade.
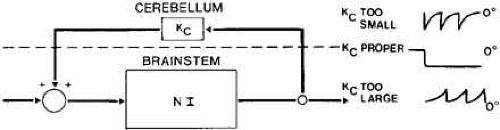 Fig. 8 Block diagram of the cerebellar positive feedback path with gain Kc around the leaky neural integrator (NI). |
During evaluation of the ocular motility of a patient, the eye movements seen are a result of both the initial insult and the plastic adaptation that has resulted; if the insult is to the structures involved in system plasticity, either hypometric or hypermetric activity is possible.
SLOW EYE MOVEMENTS
Pursuit
The major stimulus for pursuit in foveate animals is a fixated target that moves; this evokes a pursuit SEM after a latency of 125 milliseconds. The maximum sustained pursuit velocities are about 90°/second,38 although higher values can be obtained for large-amplitude, full-field, or self-moved target motions.39 The SEM of the vestibuloocular reflex (VOR) and of optokinetic nystagmus (OKN) or congenital nystagmus (CN) can be considerably faster. SEM are conjugate, smooth, and under a control system capable of continuous modification of motor output in response to visual input (in contrast to discrete FEM control). The input signal is retinal error (“slip”) velocity, which is reduced to zero when eye velocity matches target velocity. The work of Yasui and Young40 suggests that retinal slip velocity is used along with corollary discharge to recreate a target velocity signal, and it is this “perceived target velocity” that drives the SEM system. This would provide an explanation for many of the “pursuit” responses to nonmoving targets (e.g., afterimages). True pursuit is an SEM in response to a moving target. There are many other ways to elicit SEM (see Table 1), and further study is required to uncover other mechanisms. Under normal conditions, a moving target is usually required for a pursuit SEM; attempts to move the eyes smoothly without actual target motion result in a series of small saccades.41
When a foveated target suddenly moves at a constant velocity, the pursuit response begins after a 125-millisecond latency (Fig. 9). The initial movement is the same velocity as the target, but because of the latency, the eyes are behind the target and require a catch-up saccade for refoveation while continuing the tracking with a pursuit SEM. The catch-up saccade follows the initiation of the pursuit movement because of the longer latency of the FEM subsystem. Plant dynamics do not permit a simple linear increase (ramp) in neural firing frequency to rapidly accelerate the eyes to the velocity of the moving target (see Fig. 5C); a “step-ramp” of innervation is needed (see Fig. 5D). Thus, an instantaneous jump in firing frequency (the step) is followed by a linear increase in frequency (the ramp). It is commonly accepted that the same neural integrator used to generate the tonic firing level necessary for FEM is used for the step-ramp of SEM. Like FEM, the SEM subsystem is a closed loop with negative feedback (Fig. 10). The conjugate retinal error signal (slip velocity) is sensed at the visual cortex, and this information is used in the brain stem to generate the required pursuit SEM to reduce the retinal error velocity to zero. Target position, target velocity, and retinal slip velocity have all been related to the generation of smooth pursuit movements, but none of these alone adequately accounts for all the observed characteristics of pursuit SEM. Efferent eye position, velocity information, or both are probably used in addition to the aforementioned stimuli. The role of target acceleration in smooth pursuit is in dispute.42,43
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree