Chapter 15 External eye disease and the oculocutaneous disorders
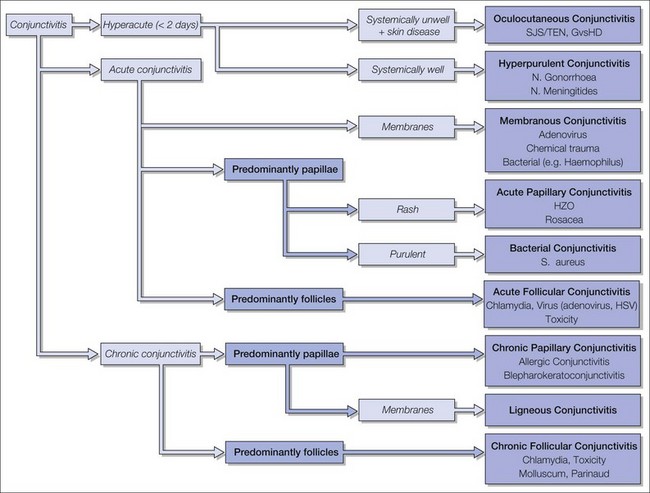
This chapter focuses on the most common external eye conditions in children, especially conjunctivitis, blepharokeratoconjunctivitis, and allergic eye disease (Fig. 15.1).
Blepharokeratoconjunctivitis
Blepharitis is common in all age groups: it is a disorder of the lid margins with or without obvious inflammation. Lid disease can involve the anterior lid margin (lash follicles) or posterior lid margin (meibomian glands).1 Corneal disease occurs with both. The clinical features include:
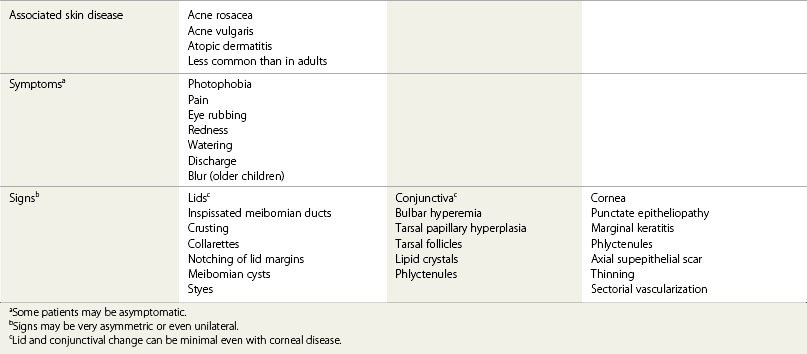
In children, dermatologic associations (rosacea and acne vulgaris) are less common,2 but corneal disease is more likely to progress to significant vision loss without obvious surface inflammation, termed blepharokeratoconjunctivitis (BKC)3 (Table 15.1).
Pathogenesis of blepharokeratoconjunctivitis
BKC is a delayed hypersensitivity response to bacterial antigens released into the tear film. The release of breakdown products of meibomian gland lipid into the tear film may cause inflammation. An unstable tear film amplifies the effect through surface drying. An immunologic mechanism is supported by the observation that rabbits immunized with Staphylococcus aureus or with the bacterial cell wall component ribitol teichoic acid develop ulcerative keratitis, phlyctenules, and marginal corneal ulcers.4,5 Posterior lid margin disease is not the result of infection. Clinically, there is keratinization of the ductules of the meibomian glands and meibomian gland “drop out.”6
The symptoms of early cases of BKC are chronically uncomfortable red eyes (Fig. 15.2). The disease can be very asymmetric or unilateral, with photophobia if there is keratitis (Table 15.1). Photophobia can limit activity. There may be eye rubbing and crusting in the morning, but discharge is not a major feature. The disease can remain asymptomatic until reduced vision or a corneal opacity alerts the patient or parents.
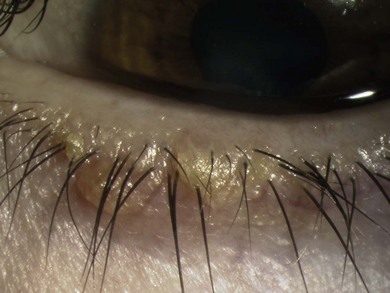
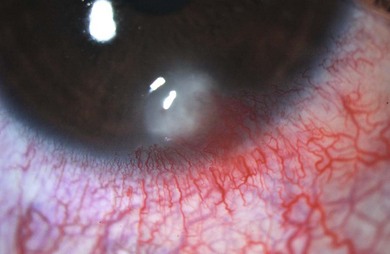
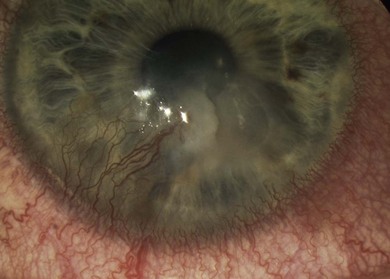
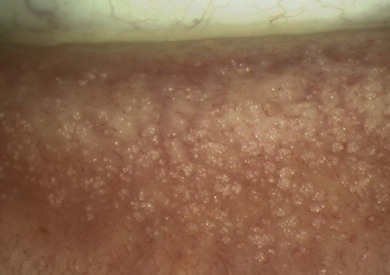
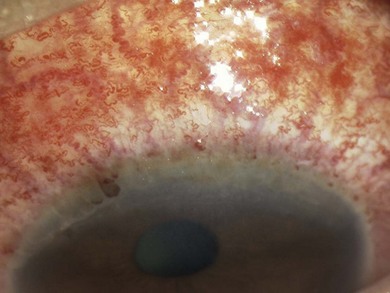
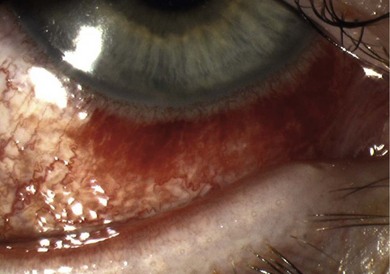
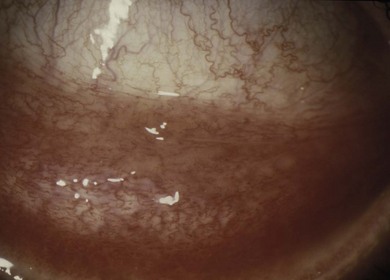
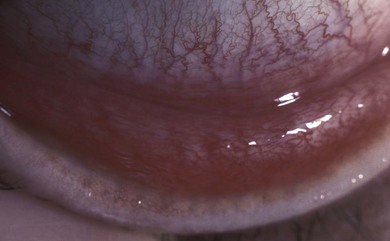
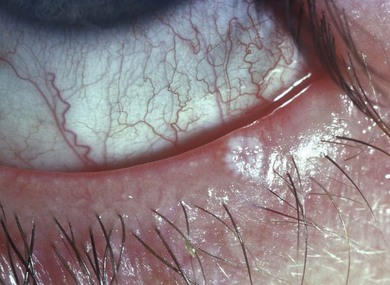
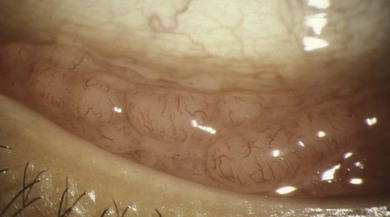
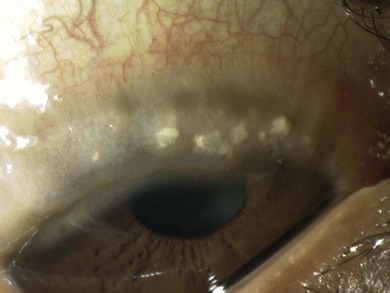
The signs of anterior blepharitis include scales and collarettes at the bases of the lashes (Fig. 15.3). The appearance of the posterior lid margin can be surprisingly normal, or there may be inspissation of the meibomian gland openings with expression of white sebum following gentle lid pressure. In more active disease there is a mixed papillary and follicular change especially in the upper and lower tarsal conjunctiva (Fig. 15.4) with limbitis and conjunctival or corneal phlyctenules (Figs 15.5 and 15.6). A phlyctenule (or phlycten) is a small white collection of polymorphs and leukocytes with an overlying epithelial defect. Conjunctival crystals are not always present but are a specific sign for this condition (Fig. 15.7).7 The corneal signs range from a mild inferior punctate keratopathy to diffuse corneal stain with fluorescein (Fig. 15.8). More severe corneal changes include peripheral corneal thinning and vascularization at the site of previous phlyctenules (Fig. 15.9). Vision loss may be severe and insidious. It is reversible if it is the result of epitheliopathy, but permanent if it is from central extension of peripheral corneal disease or diffuse central stromal scar. Some patients have corneal changes compatible with BKC but with minimal or absent lid and conjunctival change.
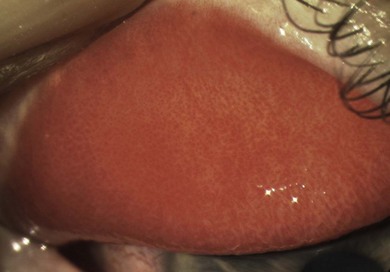
Fig. 15.4 A predominantly papillary response in a patient with very active blepharokeratoconjunctivitis.
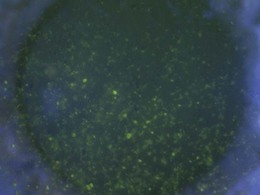
Fig. 15.8 Punctate epithelial erosion and microcysts secondary to blepharitis. Stained with fluorescein.
Treatment of blepharokeratoconjunctivitis
• Treat acute infection or styes with a warm compress and a short course of topical antibiotic (Table 15.2).
• Meibomian cysts tend to resolve spontaneously but they should be incised if they do not resolve after 3 months, or if they affect vision by altering lid position. A course of oral antibiotic (e.g. doxycycline) should be considered for recurrent cysts.
• For chronic disease, a daily warm compress followed by lid cleaning with a cotton bud moistened in boiled water followed by topical antibiotic ointment. This can take 4–6 weeks to work.
• Topical corticosteroid is the basis of treatment for most cases of BKC with conjunctival phlyctenules or significant corneal disease. For example, fluoromethalone 0.1% four times a day, reducing to once daily after 4 weeks. Long-term treatment (i.e. for years) with low-dose steroid may be required.
• Treat visually significant keratitis with long-term (8–12 weeks) low-dose oral antibiotic (Table 15.2).8 Corneal phlyctenules can rarely lead to perforation of the cornea and loss of vision due to scarring. They should be treated with intensive topical corticosteroids and topical antibiotics if there is an epithelial defect. Systemic antibiotics reduce the frequency and severity of relapses of phlyctenular disease.
• The effect of manual expression of the meibomian gland secretions is debated and it is difficult to perform without sedation in young children.
• Dietary supplement with oral flaxseed oil has been recommended.9
• Rarely, for inexorable corneal opacification, systemic immunosuppression (e.g. with mycophenolate) is required.
• Secondary microbial keratitis can occur; it should be treated immediately.
• An axial corneal opacity or irregular astigmatism may cause amblyopia. Visual correction may be possible in older children using a rigid contact lens.
Table 15.2 Antibiotic treatment for blepharokeratoconjunctivitis
| Indication + common pathogens | First line treatment | Alternatives |
|---|---|---|
| Oc Chloramphenicol 1% Applied after eye lid hygiene and at night | ||
| po Erythromycin 125 mg bd Until clinical improvement noted (usually 2 to 4 weeks) | ||
| Oc Chloramphenicol 1% after eye lid hygiene at night |
Doxycycline should not be used in children under 12 years and patients should be warned about skin sensitivity to sunlight and gastrointestinal side-effects.
The great majority of cases of phlyctenular disease in developed countries are associated with staphylococcal lid margin disease, but phlyctenules are associated with tuberculosis, and very rarely with helminthiasis, leishmaniasis, and candidiasis.10 In areas where tuberculosis is common, a child with a phlyctenule should be screened for tuberculosis.
Other uncommon causes of chronic blepharokeratoconjunctivitis
Lesions of discoid lupus erythematosus at the lash margin may mimic chronic blepharitis. These may respond to topical treatment with steroid but systemic therapy (e.g. hydroxychloroquine) is usually required.11 Lash infestation by crab lice (Phthirus pubis) can cause low-grade irritation and conjunctivitis (Fig. 15.10). Removal of the eggs (nits) and lice at the slit-lamp followed by application of white soft paraffin eye ointment to the lid margin twice daily for 10 days to smother the lice is effective. An alternative is pilocarpine 4% gel, which is toxic to adult lice. Insecticides (e.g. permethrin 1%, malathion 1%) are not licensed for ocular use; they are used in shampoos but they cause irritation. Decontamination of bed wear is essential. A pediatrician should be involved and screening for sexually transmitted infection such as chlamydia may be indicated.
Conjunctivitis
The causes of conjunctivitis are outlined in Fig. 15.1. Cultures are only necessary when there is:
Ophthalmia neonatorum (infection before 28 days) is discussed in Chapter 12.
Acute conjunctivitis
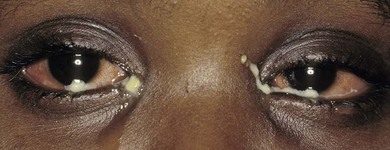
Acute bacterial conjunctivitis is common in young children. It is bilateral in 70% of cases with a mucopurulent discharge, diffuse bulbar redness, and papillary hypertrophy of the upper tarsal plate conjunctiva. Haemphilus influenzae, Streptococcus pneumoniae, Moraxella catarrhalis, and Staphylococcus aureus are the most common pathogens.12–14 One-quarter of children with conjunctivitis have a symptomatic otitis media,15,16 which usually spontaneously resolves.17 Viruses (adenovirus, picornovirus, herpes virus) are isolated in 6% of cases,14 and preauricular adenopathy is one of the few characteristic features of viral infection. Chemical irritation, BKC, and allergic conjunctivitis should be considered as alternative causes. Herpes simplex virus (HSV) rarely causes follicular conjunctivitis, just 5% of cases in one Japanese study.18
Acute infective conjunctivitis is typically self-limiting and resolves spontaneously over 2 weeks (65% in 2–5 days) (Fig. 15.11). Sight threatening complications are uncommon. Management for most cases is irrigation with boiled water and the removal of secretions on the lid with a moist cotton pad. Antibiotics are unnecessary for the majority of cases although, if used within 5 days of onset of symptoms, they accelerate the resolution of symptoms and bacterial clearance.19,20 Complications of untreated disease are unusual. There is no evidence to support the superiority of any particular antibiotic. It is not necessary to exclude children from school unless there is an epidemic.
Hyperpurulent conjunctivitis
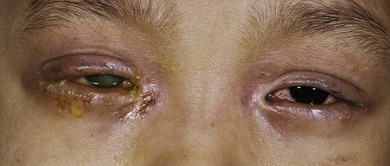
Hyperpurulent conjunctivitis requires urgent attention: it may be associated with severe corneal and systemic disease. Patients are typically febrile with a rapid onset of periocular swelling, mucopurulent discharge, pain, hemorrhagic conjunctivitis and chemosis with possible preauricular lymphadenopathy. Swelling and photophobia may make examination difficult. It is important to exclude infection with Neisseria gonorrhoeae and N. meningitidis, although Staphylococcus, Streptococcus, Haemophilus, and Pseudomonas spp. may rarely cause a similar picture (Figs 15.12 and 15.13). Neisseria gonorrhoeae conjunctivitis is not always sexually acquired, although this possibility should be excluded. Infection has occurred with the use of traditional medicines containing infected urine.21,22 Neisseria gonorrhoeae causes a rapidly progressive ulcerative keratitis with a characteristic superior corneal gutter that can rapidly perforate. Neisseria meningitidis conjunctivitis is usually acquired by airborne spread within schools. Although less severe, in 15% it causes epithelial breakdown and ulcerative keratitis.23 Metastatic spread of N. menigitidis to the eye can occur as a terminal event following septicemia.
Diagnosis and investigation of conjunctivitis
Conjunctivitis is a clinical diagnosis. Investigation is only indicated for persistent conjunctivitis or hyperpurulent conjunctivitis. Samples should include an urgent Gram stain and culture on blood agar. Polymerase chain reaction (PCR) should exclude Neisseria spp., chlamydia, adenovirus, and HSV. Treatment guidelines are presented in Table 15.3.
Table 15.3 Antibiotic treatment for conjunctivitis
| Indication + common pathogens | First line treatment | Alternative |
|---|---|---|
| Acute conjunctivitisa | Observation, or | A fluoroquinolone, e.g. G Levofloxacin 0.5% qds for 7 days (prescribe if Gram-negative infection suspected) |
| G Chloramphenicol 0.5% qds for 7–10 days | ||
| Hyperacute bacterial conjunctivitis | IM Ceftriaxone once daily for 3 days | IM Spectinomycin once daily for 3 days |
| Note Seek opinion from genitourinary and infectious diseases specialist | G Cefuroxime (PF) 5% hourly for 24 hours, then six times a day until resolved | G Ceftazidime (PF) 5% or a fluoroquinolone hourly for 24 hours, then six times a day until resolved |
| Chlamydia conjunctivitis | Oc Erythromycin 0.5% tds until review by genitourinary specialist | PO Doxycycline 100 mg bd or 200 mg daily for 7 days for children over 12 years of age |
| Note Seek genitourinary opinion | PO Azithromycin 1 g STAT (for children who weigh ≥ 45 kg) |
a Acute conjunctivitis has a 65% chance of resolving without treatment within 2–5 days.
Systemic treatment for microbial conjunctivitis
Systemic treatment is required for patients with hyperpurulent conjunctivitis, if there is evidence of generalized infection, and immunosuppressed patients. These patients should be admitted until the diagnosis and management have been confirmed. Consultation with an infectious diseases specialist should be made. Patients with otitis media and hyperpurulent conjunctivitis may harbor identical strains of Haemophilus influenzae in both sites; systemic therapy is indicated.24
Acute follicular conjunctivitis
Adenoviral keratoconjunctivitis
In many parts of the world adenoviral keratoconjunctivitis is the most common viral infection of the ocular surface, causing community or clinic-based epidemics. The cause depends on the route of referral. A British study of children seen in primary care with acute infective conjunctivitis found that 8% of cases were due to adenovirus infection.14 An American study of adults and children seen in an ophthalmic emergency room found that 62% of cases of acute infective conjunctivitis were due to adenovirus.25 The clinical signs of the ocular pathogenic strains (serotypes 8, 19, and 37) are indistinguishable, but viral serotyping is routine in some countries (e.g. Japan). This permits epidemiologic tracking of outbreaks. Classification of disease subtypes (pharyngoconjunctival fever, epidemic keratoconjunctivitis) related to specific serotypes is of limited value.
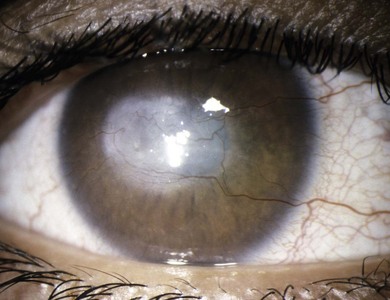
Adenovirus infection causes acute lid swelling, follicular conjunctivitis, petechial hemorrhages, conjunctival membranes, and preauricular lymphadenopathy (Fig. 15.14). There may be an upper respiratory tract infection, vomiting and abdominal pain, urethritis, and cervicitis. Corneal epithelial sloughing and anterior uveitis can occur. Usually, a focal epithelial keratitis develops in 3 to 5 days, followed after 2 weeks by the development of immune mediated focal subepithelial infiltrates (Fig. 15.15). After 3 weeks, the epithelial changes subside, leaving subepithelial scarring and irregular astigmatism that often resolves over 6 months but which may be permanent. Subconjunctival scarring is common but not progressive or clinically significant. Diagnosis can be confirmed by PCR.26 Because there may be 4–10 days of virus shedding before clinical disease is apparent, and because adenovirus can survive on dry surfaces, spread of infection between patients and clinicians is common.
There are no controlled trials showing a benefit of topical steroid or antiviral therapy for adenoviral keratoconjunctivitis. Treatment is based on symptomatic relief such as cold compress.27,28 Topical corticosteroid should be restricted to cases with visual reduction secondary to keratitis29 or if there is a membranous conjunctivitis or uveitis, but this may lengthen the period of virus shedding. Topical steroid is safe and unlikely to precipitate herpetic epithelial disease even if there is HSV conjunctivitis.18 Topical non-steroidal agents, interferon, and antivirals (acyclovir, trifluorothymidine) have no effect and should not be used (Table 15.4). The lack of effective treatment has led to strict protocols to limit nosocomial virus spread.30 Patients with an acute conjunctivitis should be seen promptly in a separate area of the clinic where they cannot mix with other patients; equipment should be decontaminated after use.
Table 15.4 Treatment options for viral conjunctivitis
| Indication + common pathogens | First line treatment | Alternative |
|---|---|---|
| Viral conjunctivitis Usually subsides without treatment within 4–7 days unless complications occur | Acyclovir eye ointment 3% 5× daily for 7 days (or continue for at least 3 days after complete healing) | Trifluorothymidine eye drops 1% (Pres/PF) 5× daily for 7 days |
| or | ||
| Oral acyclovir 200 mg 5× daily for 5 days | ||
| Viral conjunctivitis Notes: Exclude HIV/immunosuppression | Oral acyclovir 800 mg 5× daily for 7 days | |
| Viral conjunctivitis Notes: Highly contagious. Self-limited, with improvement of symptoms and signs within 5–14 days | Nil |
• For adenovirus infection instruct patient to avoid sharing personal items (towels, sheets, pillows, etc.), use meticulous hand washing, and avoid close personal contact for approximately 2 weeks.
• There is no effective treatment for adenovirus infection; however, artificial tears, topical antihistamines, or cold compresses may be used to mitigate symptoms.
• Dose reduction required when using oral antivirals in renally impaired patients.
Herpes simplex blepharoconjunctivitis
Herpes simplex conjunctivitis or blepharoconjunctivitis (Fig. 15.16) is less common than bacterial conjunctivitis in children. It is not known if it is a primary infection or recurrent disease following asymptomatic primary infection. It is often unilateral, follicular, and associated with preauricular lymphadenopathy. Blepharoconjunctivitis can be severe if there is coexisting atopic dermatitis (eczema herpeticum). There may be a diffuse punctate keratitis or a dendritic ulcer, with multiple dendrites or geographic lesions in the presence of atopic dermatitis or vernal keratoconjunctivitis. It is often self-limiting but healing is accelerated by treatment with an antiviral such as acyclovir ointment or trifluorothymidine drops five times a day for 1 week. Oral acyclovir is effective, especially for eczema herpeticum.
Acute hemorrhagic conjunctivitis
Large outbreaks of acute hemorrhagic conjunctivitis caused by enterovirus 70 or coxsackievirus A24 occurred in central Africa and Asia in the 1980s.31 It has a rapid onset and resolution with characteristic petechial subconjunctival hemorrhages, without permanent corneal change. Direct inoculation and the use of traditional eye medicines, rather than the fecal–oral route, spreads the infection. Confirmation of infection is by PCR. There is no effective treatment; management relies on infection control. The outcome is usually benign, although a polio-like paralysis (radiculomyelitis) develops in one in 10 000 enterovirus 70 patients.32
Trachoma
This is an important cause of external eye disease and the leading infectious cause of blindness. Repeated infection with serotypes A–C of Chlamydia trachomatis can cause conjunctivitis in children that progresses to scarring and blindness as adults. Without reinfection, it is a self-limiting disease. In 2002, at least 1.3 million people were blind from trachoma. Currently, 40 million people have active disease in the 50 countries where it is endemic, principally in poor rural communities in sub-Saharan Africa.33 It has disappeared from most developed countries. Transmission is by direct contact with eye or nasal secretions, eye-seeking flies, or by aerosol.
In some endemic areas, 80% of children have active disease and scarring disease can be seen in late childhood. Active disease in children is usually characterized by a mixed follicular and papillary response best seen over the everted upper tarsal plate, often accompanied by a severe inflammatory response that obscures the underlying vessels over the tarsal plate, and a superior pannus.34 Resolved follicles at the superior limbus leave depressions (Herbert’s pits) that are pathognomonic of previous infection. Corneal blindness is the result of trichiasis, dry eye, secondary infection, and vascularization.
Control of the disease is by implementation of the SAFE strategy:
Treatment of acute conjunctivitis is with a single dose of oral azithromycin (20 mg kg body weight), or 6 weeks of 1% tetracycline ointment.35 Topical azithromycin is an alternative. Mass treatment is recommended when the prevalence of active infection is > 5%.33 Clinical signs may persist for months after active infection has been eliminated. If there is trichiasis, lash epilation and lid taping are short-term options prior to lid eversion surgery.36
Molluscum contagiosum
Molluscum contagiosum is a double stranded DNA poxvirus. Transmission is by direct contact or autoinoculation to the eye. Molluscum lesions are umbilicated; when on the lid or close to the lash line they can easily be missed (Fig. 15.17). In this location they can cause a chronic follicular conjunctivitis, usually unilateral with a peak incidence at ages 2 to 4 years. Multiple lesions can develop in patients with atopy and in the immunosuppressed. Treatment is by expression or curettage of the core of the lesion, facilitated by making a small incision in the inner margin of the lesion with the tip of a needle – cautery or cryotherapy may cause depigmentation of the lid margin and loss of lashes; resolution is then rapid. Chronic cases can develop a punctate keratopathy with secondary peripheral vascularization.
Parinaud’s oculoglandular syndrome
This rare condition causes a unilateral granulomatous conjunctivitis with surrounding follicles, often associated with fever and ipsilateral regional lymphadenopathy.37 It is a variant of cat-scratch disease, which is usually caused by a Gram negative bacterium, Bartonella henselae, following a scratch from a cat or inoculation of contaminated cat-flea feces into the conjunctiva. The diagnosis is confirmed by a rising IgG serology, indirect fluorescent antibody for Bartonella spp., or PCR from affected tissue. There is a tendency to resolution but treatment is with oral azithromycin, doxycycline, or ciprofloxacin. Other rarer causative agents are tularemia (Francisella tularensis), sporotrichosis, tuberculosis, and chlamydia.
Ophthalmia nodosa
This is a granulomatous reaction of the conjunctiva or cornea to implanted plant or insect hairs. There is rapid onset of irritation, photophobia, and chemosis after exposure. Migration of barbed hairs into the tissue is aggravated by eye rubbing. Intraocular penetration of the hairs can occur, with symptoms of chronic keratoconjunctivitis, uveitis, vitritis or chorioretinitis.38 Caterpillar hairs (setae) were the first reported causative agent but hairs from pet tarantulas (e.g. Chilean rose tarantula – Grammostola rosea) are now more common. Tarantulas release a cloud of hairs as a defensive ploy when threatened. Protruding hairs can be removed, but physical removal of buried hairs is usually impossible. Mild topical steroid is effective to control inflammation. Conjunctival granulomas can be excised (Fig. 15.18).39
Conjunctival folliculosis
Folliculosis is a marked follicular response without other signs of ocular inflammation mostly seen in adolescents and young adults. There may be only mild discomfort; the follicles may have been noted coincidentally. The follicles may be present on the tarsal, forniceal, and bulbar conjunctiva (Fig. 15.19). Treatment or investigation is not usually required. A trial of topical steroid or oral doxycycline is appropriate but spontaneous resolution occurs: it may take years.
Chronic papillary conjunctivitis
Vernal keratoconjunctivitis
Clinical features
VKC usually develops in the first decade of life (82% by age 10 years with a mean age of 7 years). In 95% of cases there is remission by the late teens.40,41 In Africa, India, and the Middle East it is a substantial public health problem, accounting for 3% of eye clinic patients, and 10% of outpatient attendances.42 It affects 3–10% of children in Africa and the Middle East.43,44 The prevalence in Western Europe is less than 0.03%.43 VKC is more common in males although the gender difference is less marked in the tropics.45 In temperate regions, 45% to 75% of patients have a history of asthma or eczema; in tropical regions this is lower (0–40%). There is a family history of atopy in 50% of patients, although the expression (eczema, asthma, or allergic rhinitis) may vary in different family members. Limbal VKC is more frequent in patients of African or Asian descent; this racial predisposition persists after migration to temperate regions.46
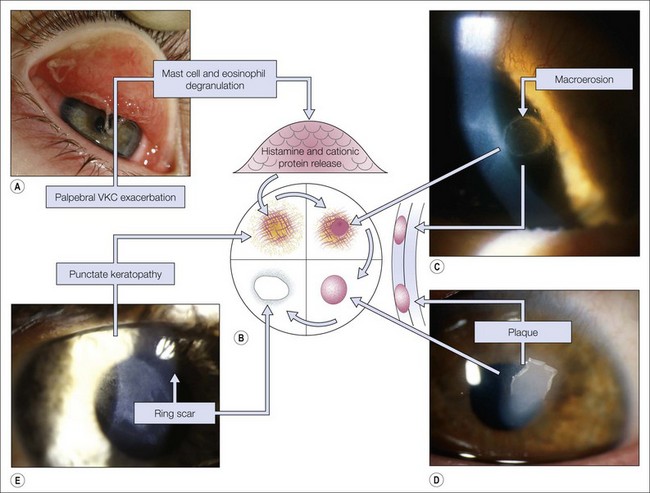
Symptoms of VKC consist of itch, photophobia, discomfort, blepharospasm, blurred vision, and mucous discharge. The disease can be markedly asymmetric. The skin of the lids may be eczematous with excoriation at the canthi and a reactive ptosis (Fig. 15.20). Papillary hypertrophy and cellular infiltration over the upper tarsal plates obscures the pattern of underlying vessels. Giant papillae (>1 mm diameter) give a cobblestone appearance and, in active disease, mucus accumulates between the papillae (Fig. 15.21). Papillae can form at the limbus appearing as gelatinous or vascular mounds with white Horner-Trantas dots (aggregates of degenerated eosinophils and epithelial cells) on the apices (Fig. 15.22). Reticular scarring can develop over the upper tarsal plate, rarely of clinical significance.47 VKC is classified as palpebral, limbal, or combined disease according to the distribution of the giant papillae. Palpebral or combined limbal and palpebral diseases behave similarly, whereas purely limbal disease, which is the more common form in tropical regions, is a more benign variant in temperate regions.48
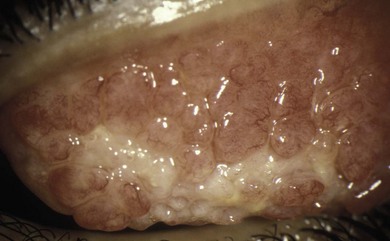
Fig. 15.21 Active palpebral vernal keratoconjunctivitis. There are giant papillae with adherent mucus.
Corneal changes
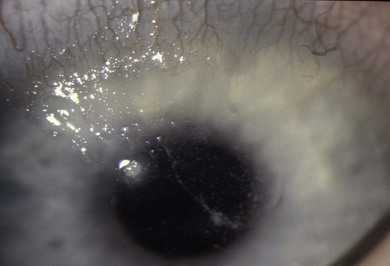
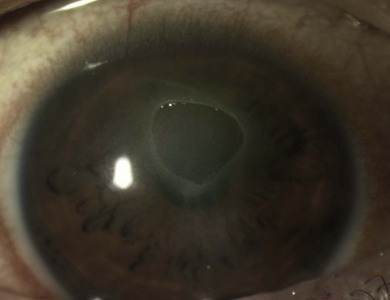
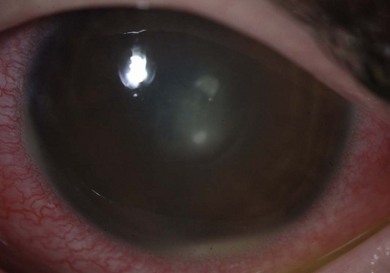
In mild disease there may be punctate epithelial erosions on the superior and central cornea. If there is active palpebral disease, mucus may be deposited on the superior corneal epithelium (Fig. 15.23), which can stimulate the formation of superficial corneal neovascularization. If there is severe palpebral disease, this may progress to corneal epithelial necrosis (macroerosion) caused by the toxic agents (e.g. eosinophilic major basic protein) released from the epithelium of the upper tarsal conjunctiva (Fig. 15.24).49 An epithelial erosion may heal completely with early intensive treatment, but in neglected cases mucus and calcium deposition on Bowman’s layer can prevent re-epithelialization and a vernal plaque (shield ulcer) develops (Fig. 15.25). These plaques rarely vascularize, but they cause intense discomfort. Because there is a risk of secondary infection, including crystalline keratopathy, a prophylactic antibiotic should be prescribed (Fig. 15.26). Although visual loss from limbal disease is uncommon an arcuate infiltrate can develop adjacent to limbal papillae (pseudogerontoxon), and there may be cystic degeneration of the conjunctiva in previously affected areas. In tropical regions, untreated limbal VKC is an important cause of visual loss.
Associated disease
Patients with VKC may have other conditions that affect their vision:
2. Keratoconus in up to 26% of patients.
3. A characteristic anterior capsular cataract in 8%.50
4. Complications of unsupervised steroid treatment in up to 20%.51
The risk of visual loss is greatest in tropical regions, varying between 0% and 10%.
The higher rate of visual loss in tropical areas is related to poor access to treatment and coexistent disease such as trachoma and bacterial conjunctivitis. In developed countries, minor visual loss from corneal scar occurs in about 6% of patients;52 25% of patients with VKC in Western Europe develop corneal complications.43
Disease mechanisms
In atopy a subpopulation of T lymphocytes (Th2) is abnormally expanded; these cells drive the disease process via the type I (IgE-mediated) immediate hypersensitivity response. Th2 cells generate cytokines and interleukins (IL-3, IL-4, and IL-13) that promote the synthesis of IgE by B cells.53,54 When an allergen comes into contact with conjunctival mast cells coated with IgE antibodies specific to that allergen, the mast cell degranulates and releases histamine and other cytokines that recruit other inflammatory cells such as eosinophils, which in turn attract more inflammatory cells.55 Additional inflammatory mediators are released into the tissue and tears.56 Tarsal and limbal papillae consist of a central vascular core of mononuclear cells surrounded by edematous connective tissue infiltrated with plasma cells, mast cells, activated eosinophils, and lymphocytes.57 Squamous metaplasia of the overlying epithelium may also contain mast cells but a reduced number of goblet cells. Scar tissue (collagen type III) forms in the core of the papillae.
Mechanical irritation can precipitate a clinical picture similar to VKC: “contact lens associated giant papillary conjunctivitis (GPC).” The role of secondary irritation (diesel particles, infection, or smoke) amplifying the symptoms of VKC has not been fully explored. The genetic basis for VKC is not fully determined. An altered epithelial and mucosal barrier function is important, allowing environmental allergens access to the immune system. Mutations in the filaggrin gene, a protein that controls keratin aggregation, may be significant.58
• Total serum IgE and tear IgE are usually elevated, but these measurements are non-specifically elevated if there is atopic dermatitis.
• Measurement of local IgE production by radio-allergosorbent test confirms allergic conjunctivitis but is only available in some specialist departments.41,59
• A cytology specimen, taken by swabbing the conjunctiva with a nylon brush, will contain eosinophils and MCT mast cells (tryptase positive, chymase negative) if there is severe allergic eye disease.60
• “Allergy testing” is not indicated in the majority of patients. In temperate regions epidermal or conjunctival challenge testing shows that at least 50% of patients are sensitive to house dust mite allergen, pollens, and animal dander.41,59 Testing for local environmental allergens (pollens, house dust mite, etc.) can support an atopic basis for the disease, but patients may react to several allergens with no indication as to which is causing the allergic conjunctivitis. Conversely, allergen-specific conjunctival provocation may reveal sensitivity to allergens that do not provoke a response by skin testing. In the same individual the allergens provoking asthma and allergic conjunctivitis may be different. Advice should be sought from a clinical allergist if the disease is refractory.
Management
There is the potential to retain good vision in the majority of cases of VKC, and iatrogenic disease must be avoided. The presence of papillae is not a good indicator of activity. This is best reflected by the presence of mucus between the papillae, Trantas dots, mucus adherent to the corneal epithelium, as well as corneal epithelial breakdown, vascularization, and ulceration. A grading system for severity of disease based on the size of superior tarsal papillae and associated scarring, the presence of limbal papillae, the extent of encroachment of the papillae onto the peripheral cornea, and secondary corneal changes has been proposed.61 Medical management is proportional to symptoms and signs; intensive topical corticosteroid is reserved for crises (Fig. 15.27).61 The following should be considered:
• Allergen avoidance by eliminating feather pillows, carpets, pets. Allergens are often locally distributed, but geographic treatment by relocation may not be practical. The patient’s school may need to be informed that VKC is not an infection. School staff may need to administer treatment during the school day.
• An oral antihistamine can help sleep and reduce nocturnal eye rubbing.
• Topical medications are effective, but no one agent is superior.62 For mild disease, topical histamine (H1) antagonists (levocobastine 0.05%, emedastine 0.05%) produce rapid symptomatic relief. Topical cromones (sodium cromoglycate 2–4%, nedocromil sodium 2%) and other mast cell stabilizers (lodoxamide 0.1%) prevent mast cell degranulation. Dual action agents active against H1 receptors and mast cell degranulation (e.g. olopatadine 0.1%) have been introduced. All are safe for long-term maintenance therapy, reducing the number and severity of exacerbations and the need for supplementary topical corticosteroid.
• Topical acetylcysteine 5–10% reduces mucus adherence to the cornea during exacerbations.
• The role of topical non-steroidal agents (diclofenac 0.1%, ketorolac 0.5%), a potentially safe option, needs better evaluation.
• Topical corticosteroid is very effective, but patients should be carefully monitored for side-effects (glaucoma, cataract, and ocular herpetic infection). Synthetic steroids (fluoromethalone, loteprednol, rimexolone) may reduce the risk of glaucoma and cataract. Steroid ointment, such as betamethasone, may be useful at night to reduce treatment frequency.
• Steroid injected into the supratarsal space after lid eversion (0.5–1.0 ml of either dexamethasone (4 mg/ml) or triamcinolone (40 mg/ml)) is reserved for severe disease not responding to topical treatment, or given following surgery for a vernal plaque.
• Cyclosporin A (0.05% to 2%) is safe in children.63 It is an alternative to topical corticosteroid, but probably less well tolerated, less effective, and more expensive.64 The safety of topical antimetabolites such as mitomycin 0.01% needs to be confirmed.65
• Systemic immunosuppression with corticosteroids, cyclosporin A, tacrolimus, or azathioprine is reserved for severe unremitting disease with corneal complications. Leukotriene receptor antagonists (e.g. montelukast) appear to only be effective if there is associated atopic asthma.66 Molecular antagonists of IgE (e.g. omalizumab) and immunoglobulin are very expensive and have not yet been fully evaluated.
• Surgical excision or cryotherapy of papillae produces only temporary remission. Application of mitomycin 0.02% after excision may reduce the rate of recurrence.67 Cryotherapy at the limbus risks causing limbal stem cell failure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree