Purpose
To explore the visual and anatomic outcomes of patients with refractory or recurrent neovascular age-related macular degeneration (AMD) who were converted from bevacizumab and/or ranibizumab to aflibercept.
Design
Two-center, retrospective chart review.
Methods
Treatment history, visual acuity (VA), and central macular thickness (CMT) on spectral-domain optical coherence tomography were collected. Patients were divided into “refractory” (persistent exudation despite monthly injections) or “recurrent” (exudation suppressed, but requiring frequent injections).
Results
One hundred and two eyes of 94 patients were included; 68 were refractory and 34 were recurrent. Eyes received a mean of 20.4 prior bevacizumab/ranibizumab injections and a mean of 3.8 aflibercept injections. Mean follow-up was 18 weeks. Mean VA was 20/50-1 before conversion, 20/50-2 after 1 aflibercept injection ( P = .723), and 20/50+2 after the final injection ( P = .253). Subgroup analysis of refractory and recurrent cases also showed stable VA. Of the refractory cases, mean CMT had improved after 1 injection ( P < .001) and the final injection ( P < .001). Intraretinal ( P < .001) and subretinal ( P < .001) fluid decreased after 1 injection, and the mean injection interval was extended from 5.2 to 6.2 weeks ( P = .003). Of the recurrent cases, mean CMT improved after 1 injection ( P < .001) and the final injection ( P < .001). Intraretinal ( P = .003) and subretinal ( P = .046) fluid decreased after 1 injection, and the mean injection interval was extended from 7.2 to 9.5 weeks ( P = .001).
Conclusions
Converting patients with chronic neovascular AMD to aflibercept results in stabilized vision and improved anatomic outcomes, while allowing injection intervals to be extended.
The introduction of intravitreal anti–vascular endothelial growth factor (VEGF) agents greatly improved the visual prognosis for patients with neovascular age-related macular degeneration (AMD). The efficacy of monthly ranibizumab (Lucentis; Genentech, South San Francisco, California, USA), an affinity-matured anti-VEGF antibody fragment that binds all VEGF-A isoforms, was demonstrated in the MARINA (Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab In the Treatment of Neovascular AMD) and ANCHOR (Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in AMD) trials. Bevacizumab (Avastin; Genentech) is the parent molecule of ranibizumab and is US Food and Drug Administration (FDA) approved for the treatment of several systemic malignancies. Its intravitreal use has become widely adopted as an off-label, cost-effective alternative to ranibizumab, with recent randomized controlled data demonstrating comparable efficacy in treating neovascular AMD.
However, despite the encouraging results of multiple trials and clinical experience, many patients require continued monthly injections because of recurrent exudation, and others have suboptimal responses or develop tachyphylaxis. The recent introduction of aflibercept (Eylea; Regeneron, Tarrytown, New York, USA) provides an alternative mechanism of VEGF blockade. Aflibercept is a soluble decoy receptor, engineered by fusing VEGF receptor (VEGFR) 1 and VEGFR2 to the Fc portion of human immunoglobulin G-1 (IgG-1), allowing it to bind all isoforms of VEGF-A, VEGF-B, and placental growth factor (PlGF). Aflibercept has been shown to have higher binding affinities compared to ranibizumab and bevacizumab, which allows a less frequent dosing schedule of every 8 weeks after 3 initial monthly injections. Its efficacy in patients with newly diagnosed neovascular AMD was demonstrated in the VIEW trials (VEGF Trap-Eye: Investigation of Efficacy and Safety in Wet AMD [VIEW 1, VIEW 2]), which were 2 parallel phase III trials.
Since the FDA approval of aflibercept for neovascular AMD, many practitioners are converting patients from bevacizumab or ranibizumab to aflibercept with the goal of extending treatment intervals or achieving better responses for refractory cases. In the current study, we examined the short-term visual and anatomic outcomes of patients with neovascular AMD refractory to, or recurrent on, bevacizumab and/or ranibizumab, who were switched to aflibercept injections. We also examined whether aflibercept conversion could extend the injection intervals to decrease injection burden.
Methods
This study is a retrospective, interventional, noncomparative case series of consecutive patients treated at the Massachusetts Eye and Ear Infirmary and Harvard Vanguard Medical Associates. Subjects were identified in our electronic medical records as those with neovascular AMD who were previously treated with ranibizumab and/or bevacizumab and then converted to aflibercept between December 1, 2011, and July 31, 2012. Exclusion criteria were concomitant visually significant ocular pathology (such as vitreous hemorrhage), insufficient clinical records, fewer than 3 previous anti-VEGF injections, and lack of follow-up after conversion to aflibercept. The study protocol was approved by the Institutional Review Boards of the Massachusetts Eye and Ear Infirmary (IRB protocol #12-073H) and Harvard Pilgrim Health Care (IRB protocol #376548). The protocols allowed retrospective data collection of patients with neovascular AMD who were treated with intravitreal aflibercept injections. The study complied with the Health Insurance Portability and Accountability Act of 1996 and conformed to the tenets of the Declaration of Helsinki.
Treatment schedules, re-treatment schedules, and injection methods were at the discretion of individual retina specialists. Local and systemic adverse events were assessed at every clinic visit. Visual acuities were converted from Snellen notation to the logarithm of the minimal angle of resolution (logMAR) scale. Spectral-domain optical coherence tomography (SDOCT) was performed by Cirrus (Carl Zeiss Meditec, Dublin, California, USA), Spectralis (Heidelberg Engineering, Heidelberg, Germany), or RTVue (Optovue, Fremont, California, USA) SDOCT systems. Individual patients were followed using the same SDOCT system. Central macular thickness (CMT) values were obtained using the integrated software. Baseline preconversion OCTs were defined as those performed on the day of aflibercept conversion. The injection intervals for prior anti-VEGF therapies and for aflibercept were calculated from the most recent interval for the respective treatments for each patient. Patients were divided into 2 groups: “refractory” (persistent intraretinal and/or subretinal fluid despite monthly injections) or “recurrent” (patients who responded well, but required frequent repeated injections to maintain a dry macula). Subretinal fluid and intraretinal fluid were categorized into presence of fluid, no fluid, or trace fluid, based on review of entire volume scans. Trace fluid was defined as a few fine intraretinal cysts or noncentral thin slivers of subretinal fluid.
Paired variables before and after aflibercept conversion were analyzed using the Wilcoxon signed rank test. Nonparametric distribution was confirmed using histogram plots. All statistical tests were 2-tailed and significance was defined as P < .05. Missing data were not imputed. Statistical analysis was performed using Stata version 9.0 (StataCorp, LP, College Station, Texas, USA).
Results
Patient Characteristics
One hundred patients (108 eyes) with neovascular AMD who were converted from bevacizumab or ranibizumab to aflibercept were identified. Six patients (6 eyes) were excluded because of vitreous hemorrhage (1), corneal pathology (1), lack of follow-up (1), and use of ranibizumab only once before aflibercept conversion in newly diagnosed patients (3). The remaining cohort was composed of 94 patients (102 eyes). Patient characteristics at the time of aflibercept conversion are summarized in Table 1 .
| Demographics | |
| Patients (eyes) | 94 (102) |
| Mean age (range) | 79.6 (57-93) |
| Women, n (%) | 58 (61.1%) |
| White race, n (%) | 90 (94.7%) |
| Right eye, n (%) | 49 (48.0%) |
| Refractory, n | 68 |
| Recurrent, n | 34 |
| Previous injections, a mean (median; range) | 20.4 (18; 3-65) |
| Aflibercept injections, mean (median; range) | 3.8 (3.5; 1-8) |
Treatment Characteristics
Sixty-eight eyes had refractory neovascular AMD, and 34 were recurrent. Of the refractory cases, 22 were previously treated with ranibizumab alone, 24 with bevacizumab alone, and 22 with both ranibizumab and bevacizumab. Of the recurrent cases, 26 were previously treated with ranibizumab alone, 2 with bevacizumab alone, and 6 with both ranibizumab and bevacizumab. The lengths of previous anti-VEGF treatment were 141.7 weeks overall, 126.2 weeks for refractory patients, and 172.8 weeks for recurrent patients. The most recent injection intervals are displayed in Table 2 , which is a reflection of the clinical activity at the time of conversion.
| All | P | Refractory | P | Recurrent | P | |
|---|---|---|---|---|---|---|
| Mean visual acuity (logMAR) a | ||||||
| Before conversion (SD) | 0.42 (0.30) | 0.44 (0.33) | 0.38 (0.23) | |||
| After 1 injection (SD) | 0.44 (0.36) | .723 | 0.46 (0.41) | .897 | 0.39 (0.25) | .778 |
| Final (SD) | 0.38 (0.27) | .253 b | 0.38 (0.28) | .215 | 0.38 (0.25) | .811 |
| Mean CMT (μm) | ||||||
| Before conversion (SD) | 305.07 (80.65) | 311.57 (77.83) | 288.83 (86.86) | |||
| After 1 injection (SD) | 274.05 (68.98) | <.001 | 283.33 (66.97) | <.001 | 250.83 (69.82) | <.001 |
| Final (SD) | 276.20 (69.82) | <.001 | 283.01 (68.73) | <.001 | 260.97 (70.00) | .001 |
| Injection intervals (weeks) | ||||||
| Previous (SD) | 5.86 (2.55) | 5.18 (1.59) | 7.21 (3.44) | |||
| Aflibercept (SD) | 7.27 (2.94) | <.001 | 6.15 (2.18) | .003 | 9.47 (3.03) | .001 |
a LogMAR values of 0.30, 0.40, and 0.50 are equivalent to 20/40, 20/50, 20/63 in Snellen notation, respectively.
b Second P values are also compared to values before aflibercept conversion.
In addition, 6 eyes also had a history of being treated with photodynamic therapy (PDT), 1 with thermal laser, and 2 with pegaptanib (Macugen; Eyetech, New York, New York, USA). Thirty-eight of 102 eyes (37.3%) were previously treated with enhanced doses of bevacizumab (2.00 mg) or ranibizumab (0.75 mg or 1.00 mg). All patients were treated using a pro re nata (PRN) and/or treat-and-extend protocol. Conversion to aflibercept took place on average 7.0 weeks after the last bevacizumab or ranibizumab injection for both groups combined, 5.7 weeks for the refractory group, and 9.5 weeks for the recurrent group. The mean time between the first aflibercept injection and the subsequent OCT was 5.2 weeks overall, 5.0 weeks in refractory patients, and 5.7 weeks in recurrent patients. Mean follow-up was 18.4 weeks after the first injection of aflibercept (17.6 weeks for refractory, 19.9 weeks for recurrent).
Visual Outcomes
Visual outcomes are summarized in Table 2 . Mean baseline visual acuities before aflibercept conversion were approximately 20/50 in the combined, refractory, and recurrent groups. Mean visual acuity remained stable after 1 injection of aflibercept in all groups. There was a trend towards improved mean visual acuity in the combined and refractory groups on the final visit, but this was not statistically significant.
Anatomic Outcomes
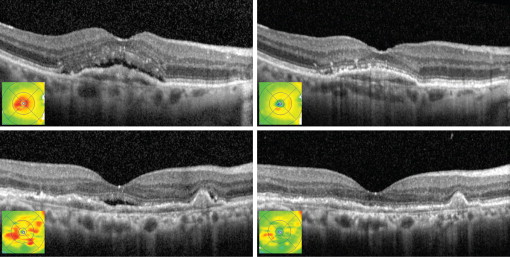
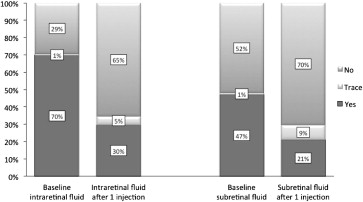
CMT was significantly decreased after 1 injection and at the final visit in all groups ( Figure 1 ; Table 2 ). The proportion of eyes with intraretinal and subretinal fluid decreased after 1 injection ( Figure 2 ). When the intraretinal and subretinal fluid was graded from none to trace to present, there were statistically significant improvements in the combined and refractory groups ( Table 3 ). In recurrent cases, the proportion of patients with intraretinal fluid decreased after 1 injection and at the final visit, whereas the proportion with subretinal fluid decreased after 1 injection and trended lower at the final visit, but lost statistical significance.
| All (Eyes) | Refractory (Eyes) | Recurrent (Eyes) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No | Tr | Yes | P | No | Tr | Yes | P | No | Tr | Yes | P | |
| Intraretinal fluid | ||||||||||||
| Before conversion | 30 | 1 | 71 | 16 | 1 | 51 | 14 | 0 | 20 | |||
| After 1 injection b | 53 | 4 | 24 | <.001 | 33 | 4 | 20 | <.001 | 20 | 0 | 4 | .003 |
| Final b | 54 | 4 | 40 | <.001 a | 34 | 3 | 30 | <.001 | 20 | 1 | 10 | .005 |
| Subretinal fluid | ||||||||||||
| Before conversion | 53 | 1 | 48 | 30 | 1 | 37 | 23 | 0 | 11 | |||
| After 1 injection b | 57 | 7 | 17 | <.001 | 35 | 7 | 15 | <.001 | 22 | 0 | 2 | .046 |
| Final b | 65 | 8 | 25 | <.001 | 41 | 8 | 18 | <.001 | 24 | 0 | 7 | .180 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


