Fig. 1.1
Auditory neurons connect mechanosensory hair cells in the organ of Corti to cochlear nuclei neurons in the brain stem. a Cross section through the cochlea and brain stem of an adult tau-GFP neuron reporter mouse (2 months old). SGN spiral ganglion neurons; OC organ of Corti; CN cochlear nucleus. a′ High-magnification view of the spiral ganglion neurons showing the dendrites extending in the periphery to the organ of Corti (OC) and the axons traveling to the central nervous system. a″ High-magnification view demonstrating the spiral ganglion neuron endings (red Vglut1, vesicular glutamate transporter 1) on the neurons of the cochlear nucleus (green tau-GFP, with blue nuclei). Work performed by Dr. Koji Nishimura
1.2 Historical Perspective
The development of the auditory neurons and their connectivity was studied by early anatomists such as Gustaf Retzius, Fredrich-Christian Rosenthal, Alfonso Corti, Rudolph Albert von Kölliker, and Rafael Lorente de Nó. Indeed, the synapse between the hair cell and the sensory neurons was defined already by Retzius (1884) and made him one of the first adopters of the newly formulated neuron theory proposed by S. Ramon y Cajal. Retzius’ silver impregnations showed a sharp stop at hair cells and thus led him to believe that the proposed discontinuity of sensory neurons and hair cells was in line with the neuron theory, which contrasted with the prevailing reticular theory of a syncytial nerve net (Retzius, 1884; Grant, 2011). Retzius’ studies also presented the first detailed account of the development and cellular organization of the organ of Corti, the hearing organ of mammals. In addition, they clarified much of the evolution of the human organ of Corti from the basilar papilla found in other tetrapods (Retzius, 1884; Fritzsch et al., 2013). At the end of the 19th century, Retzius’ ear studies provided the most detailed and comprehensive analysis of any vertebrate organ system development and evolution. Based on his standing in science at the time, Retzius was nominated for the Nobel Prize 11 times, without ever receiving it.
It took about 50 years before another great neuroanatomist, R. Lorente de Nó, could surpass the achievements of Retzius. As summarized in his 1981 volume (Lorente de Nó, 1981), Lorente de Nó in the 1930s showed, in great detail, how spiral ganglion neurons project to the organ of Corti (Fig. 1.2) and how the central projections into the cochlear nuclei are cochleotopically organized (Fig. 1.3). This beautiful work, unfortunately, followed the perception of Lorente de Nó’s teacher, Ramon y Cajal, in assuming that there is no efferent system to the ear. Because of this bias, Lorente de Nó showed both afferent and efferent fibers in the organ of Corti in a bewildering complexity of a rich variety of spiral ganglion neurons (Fig. 1.2). In contrast to the baffling differences in peripheral projections, the central projections drawn by Lorente de Nó (Fig. 1.3) showed a much greater uniformity. The discovery that efferents to the ear exist (Rasmussen, 1953; Simmons et al., 2011; Sienknecht et al., 2014) and that spiral ganglion neurons comprise only two distinct types (Spoendlin & Schrott, 1989) overturned much of the early interpretations of Lorente de Nó. However, these different interpretations do not invalidate the information presented in the exquisite drawings that followed in great detail those of his teacher, S. Ramon y Cajal. Indeed, it took 100 years before modern techniques were developed that matched or exceeded the resolution of the Golgi technique to study spiral ganglion neurons (Figs. 1.1, 1.2, 1.3) and their peripheral and central projections. The present SHAR volume provides insights that go well beyond those early stages of the neurobiology of the spiral ganglion by adding molecular and physiological dimensions that will inform future clinical applications how to combat hearing impairment.
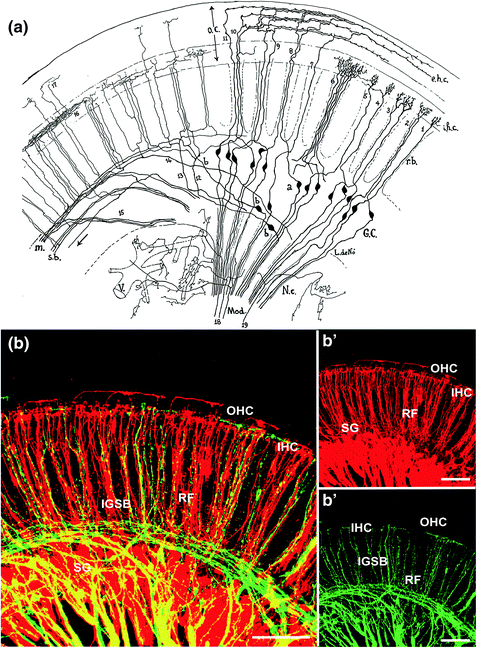
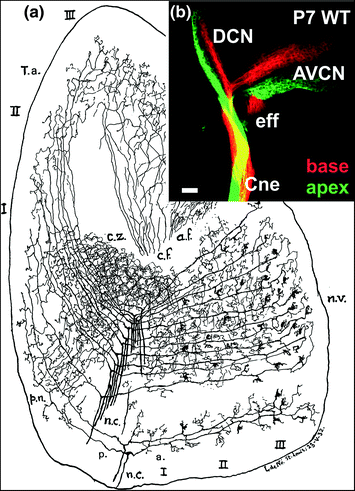

Fig. 1.2
Lorente de Nó (1981) showed in exquisite detail the organization of spiral ganglion neurons and their projections to the organ of Corti in postnatal mice (a). Note that only the right half of the image shows indeed the two types of spiral ganglion cells; on the far right the type I bipolar ganglion cells reaching to inner hair cells; and in the center the type II, partially pseudounipolar ganglion cells reaching to outer hair cells. The left side of this drawing shows what is now known as efferents but was interpreted by Lorente de Nó as a different set of spiral ganglion neurons. Only modern tracing techniques using injections of different colored tracers into the efferents (green in b) and cochlear nucleus (red in b) of mice around birth showed that efferents and afferents take partially overlapping but distinct trajectories. Although they overlap in radial fiber bundles (RF), only efferents contribute to the intraganglionic spiral bundle (IGSB). Both afferents and efferents form distinct terminals on outer (OHC) and inner hair cells (IHC) of the organ of Corti. Images are from Lorente de Nó (1981) and Simmons et al. (2011)

Fig. 1.3
Lorente de Nó used his Golgi impregnations (a) to demonstrate the topological organization of spiral ganglion afferents in the cochlear nuclei of postnatal rodents and the vastly different terminals in the different parts of the cochlear nucleus complex. Note that each fiber provides multiple contacts with different second-order neurons that have radically different morphologies. Modern techniques using differently colored tracers show that indeed the cochlear nuclei receive fibers from distinct nonoverlapping areas of the organ of Corti in this mouse embryo (b). Injections into the base (red) label fibers terminating dorsally, whereas injections into the apex (green) label fibers terminating ventrally in the cochlear nucleus complex. In addition, efferent fibers (eff) segregate from the cochlear nerve (Cne) to enter the brain via the vestibular root. AVCN, anteroventral cochlear nucleus; DCN, dorsal cochlear nucleus. Images taken from Lorente de Nó (1981) and Fritzsch et al. (2015)
1.3 Objectives of the Book
Beyond these early neuroanatomical insights, research over the last 30 years has shown that loss or damage of hair cells and/or spiral ganglion neurons is the leading cause of congenital and acquired hearing loss affecting millions of humans (Yamasoba et al., 2013). The most common therapeutic strategies for hearing loss are based on either using hearing aids to increase hair cell stimulation in moderate hearing loss or utilizing cochlear implants (CIs) as an electronic substitute for hair cells in cases of severe hearing loss resulting from severe hair cell loss (Zeng et al., 2011). However, both scenarios depend on the presence of functional spiral ganglion neurons to achieve a positive outcome. Indeed clinical data suggest that the function of CIs depends critically on the remaining number of spiral ganglion neurons (Nadol et al., 1989; Reiss et al., 2012). Thus, the development of methodologies that could be used in the maintenance, repair, and regeneration of spiral ganglion neurons in a damaged ear have significant implications for future advances in cochlear implant technology and the treatment of neurosensory hearing loss.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


