CHAPTER 159 Cochlear Implantation
Medical and Surgical Considerations
The physical characteristics of receiver stimulators and electrode arrays vary among manufacturers, thus necessitating surgical techniques that are specific to the device to minimize complications.1–4 The facial nerve monitor is recommended in all cases as an adjunct for preventing overheating or other damage to the facial nerve while working within the facial recess and the middle ear.
Medical and Surgical Evaluation
This evaluation includes all aspects of cochlear implant candidacy, including critical analysis of hearing tests, motivation of the patient and family, and status of language development (Box 159-1). It should also focus on the patient’s general health, especially with regard to his or her ability to undergo general anesthesia. A complete history and physical examination is followed by appropriate laboratory tests. Imaging studies play a particularly important role in this evaluation. When necessary, consultation with appropriate specialists is indicated.
Hearing tests with ear-specific auditory information in both aided and unaided conditions must be obtained. It is critically important to be certain that the patient’s hearing aids are appropriate for the hearing loss. In young, prelinguistically deafened children, it may not be possible to accurately assess hearing during a single session or even a series of sessions within a sound booth. In very young children, a combination of diagnostic audiometric strategies may be necessary, including auditory brainstem response (ABR), auditory steady-state response (ASSR), and behavioral observation. The ABR is limited in its ability to obtain tone-specific information (limited frequency specificity). ASSR uses a continuous tone with a carrier frequency that is amplitude and frequency modulated. Similar to an ABR, the ASSR is recorded from scalp electrodes, but the recordings show primary energy at the carrier frequency and two sidebands separated from the carrier by the modulation frequency. Recent studies have demonstrated correlation between the degree of hearing loss measured by standard PTA and ASSR at 500, 1000, 2000, and 4000 Hz.5
Genetic testing continues to evolve as an important adjunct in the diagnosis of hearing loss. Approximately half of the estimated 1 in 1000 infants born with bilateral severe to profound deafness are thought to have genetic factors. There are more than 46 genes currently identified that contribute to hearing loss, but one gene, GJB2, which encodes the protein connexin 26 and maps to the autosomal recessive deafness gene (DFNB1) locus, is responsible for a significant proportion of these cases. When mutations in GJB2 and GJB6 (connexin 30) are screened together, the cause of 50% of bilateral severe to profound hearing loss is identified in certain populations. Four main benefits of genetic testing have been identified.6 First, the identification of a specific genetic cause precludes further costly testing. It also defines the chance of transmission by understanding the specific genetic cause of the hearing loss and its method of inheritance. Testing also provides parental reassurance that it was nothing done during the pregnancy that contributed to the hearing loss, thus helping to prevent unnecessary guilt. It also provides prognostic information with respect to the possible development of other medical problems and can help in predicting outcomes with hearing rehabilitation such as cochlear implantation.7
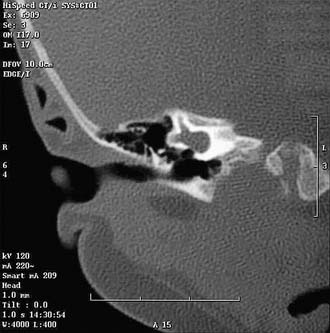
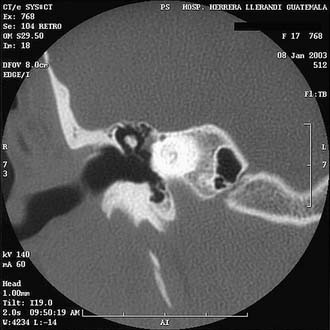
Both high-resolution computed tomography (HRCT) and magnetic resonance imaging (MRI) may be used in the preoperative evaluation of patients with profound deafness who are candidates for cochlear implantation. HRCT may assist in the evaluation of inner ear morphology (Fig. 159-1), patency of the cochlea (Fig. 159-2), position of the facial nerve, location of large mastoid emissary veins, size of the facial recess, thickness of the parietal bone, and height of the jugular bulb (Box 159-2). MRI has been used more frequently for preimplantation evaluation in recent years. Although MRI is not as effective in visualizing the fallopian canal or the bony structure of the otic capsule, it is more effective in identifying fibrosis of the cochlea by assessing its patency, in identifying the presence and caliber of the cochlear nerve (especially on sagittal T2 imaging of the internal auditory canal), and in providing information about brainstem and cortical lesions. A study compared the ability of HRCT and MRI for identifying abnormalities of the cochlea and modiolus, and MRI was found to be superior.8 For these reasons, it has subsequently become the preferred means for preoperative evaluation of children undergoing cochlear implantation at many institutions. It is recommended, however, that HRCT still be performed in cases in which there are malformations of the external canal, semicircular canals, or vestibule due to the higher incidence of an anomalous facial nerve.8 Both tests may be indicated in the presence of labyrinthine anomaly.
Ear Selection
Residual Hearing Level
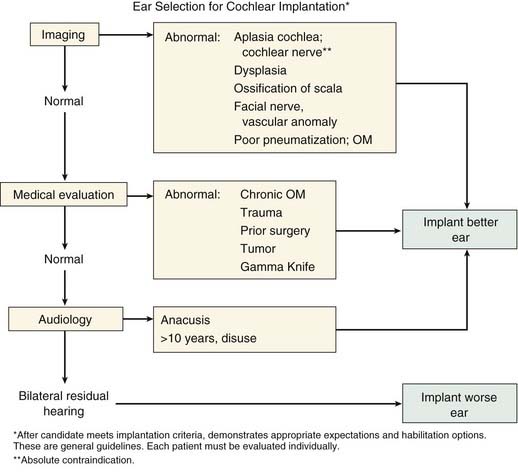
At this time, the risk of losing residual hearing during electrode insertion is estimated at 50% to 70%, although recent techniques may reduce the risk to 20%.9 Thus, if there is significant aided benefit from the better ear, the worse ear is often favored. Conversely, it is widely held that patients with more residual hearing have better outcomes from cochlear implantation. For this reason, if a patient is anacusic, the ear that has more residual hearing may be favored. Fig. 159-3 presents an algorithm that may be helpful when considering these issues.
Vaccination for Meningitis
In 2003, a New England Journal of Medicine article highlighted an increased incidence of pneumococcal meningitis in cochlear implant recipients in comparison to an age-matched general population.10 A total of 4264 children were evaluated and 26 children were identified with streptococcal meningitis, representing an incidence of 0.6% in cochlear implant recipients. This was a 30-fold increase over their age-matched convenience sample cohort. Two major limitations of the study were that, first, 11.5% of children receiving an implant have a previous history of meningitis, and these children have an increased risk of recurrence. Second, 8.5% of children receiving implants have labyrinthine dysplasia, which also places them at an increased risk for meningitis. The degree to which an increase in meningitis is due to a cochlear implant, versus high risk factors such as prior meningitis or dysplasia, requires an adequate control group of deaf children rather than normal children as used in this study.
In order to test whether this risk was theoretical or actual in nature, animal experiments were performed in rats to determine whether placement of a cochlear implant increases the risk of meningitis.11 In this study, rats receiving a cochlear implant were at a significantly increased risk of developing bacterial meningitis when inoculated with S. pneumoniae by any route (intraperitoneal, middle ear, inner ear) in comparison to surgical controls. In a later companion study, vaccination with 23-valent pneumococcal capsular polysaccharide vaccine (PPV23, Pneumovax) prevented the development of meningitis in rats inoculated by an intraperitoneal route and significantly reduced the frequency of meningitis in rats inoculated by a middle ear route. There was a nonsignificant reduction in frequency of meningitis in rats inoculated via the inner ear. This suggested PPV23 can protect healthy rats from meningitis caused by a vaccine-covered S. pneumoniae serotype.12
There are currently two vaccines available that both generate antibodies to the polysaccharide capsule of S. pneumoniae. These include the PPV23 vaccine, which is a 23-valent vaccine composed of pure capsular antigen, and the heptavalent pneumococcal conjugated vaccine (PCV7, Prevnar), which contains seven pneumococcal serotypes conjugated to a nontoxic variant of diphtheria toxin. Both of these generate an antibody response, although PPV23 is not recommended for children younger than 2 years of age because of immaturity of their immune systems that may lead to a potentially poor response to pure capsular antigen.13 It is the current recommendation of the Centers for Disease Control and Prevention (CDC) to ensure that children receiving a cochlear implant are immunized according to age-appropriate pneumococcal vaccination with PCV7, PPV23, or both according to the Advisory Committee on Immunization Practices (ACIP) schedules for persons at high risk (Box 159-3).14
Box 159-3 ACIP Recommendations for Persons Receiving a Cochlear Implant
Surgical Technique
Incisions and Flap Design
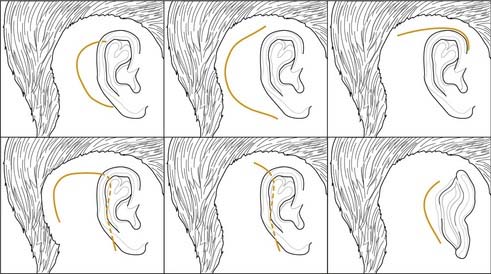
Cochlear implant incisions have evolved over time (Fig. 159-4), but the principles of incision design remain constant. The resulting flap should cover the implant with margins of at least 1 cm in all directions, and a good blood supply should be maintained. Access to the linea temporalis, mastoid tip, and spine of Henle, without undue retraction, is necessary.
The original postauricular C-shaped incision used with single-channel implants was effective and had a low rate of complications. However, as multichannel devices came into use, the larger receiver stimulators required a larger flap. These larger postauricular C-shaped incisions were associated with a higher rate of complications—mainly device extrusion—and were subsequently replaced by an inferiorly based “inverted U” flap. This has undergone further modification (see Fig. 159-4), to an extended postauricular incision that has gradually become shortened and remains the most commonly used incision at this time. An extended endaural incision has also been used, but early experience with this incision demonstrated an unacceptably high incidence of skin breakdown at the external auditory meatus.
Before incision, the intracutaneous infiltration of 1 to 3 mL of 1 : 100,000 epinephrine may be used to obtain better hemostasis. Similarly, a monopolar cautery knife may be used for both incision and flap elevation. It should be noted, however, that in cases of simultaneous or sequential cochlear implantation, monopolar cautery is contraindicated due to potential current interaction with the circuitry of the receiver-stimulator. In these cases, it is advisable to perform the approach for both sides before placing the implant (simultaneous implants)15 to use a nonconducting means of dissection to provide hemostasis, including the bipolar, Shaw scalpel, and most recently the Plasma Knife in cases of sequential implantation.16