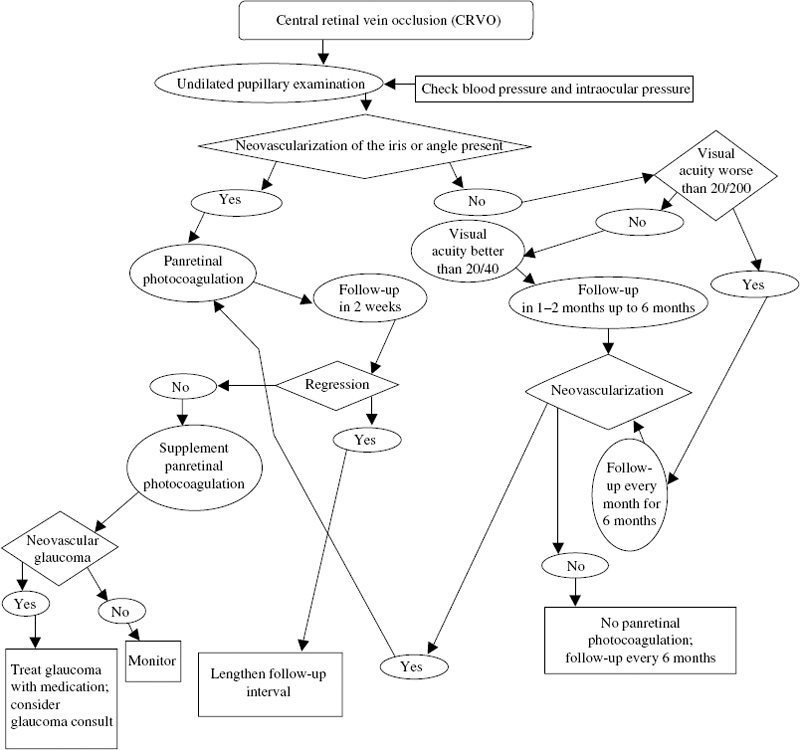
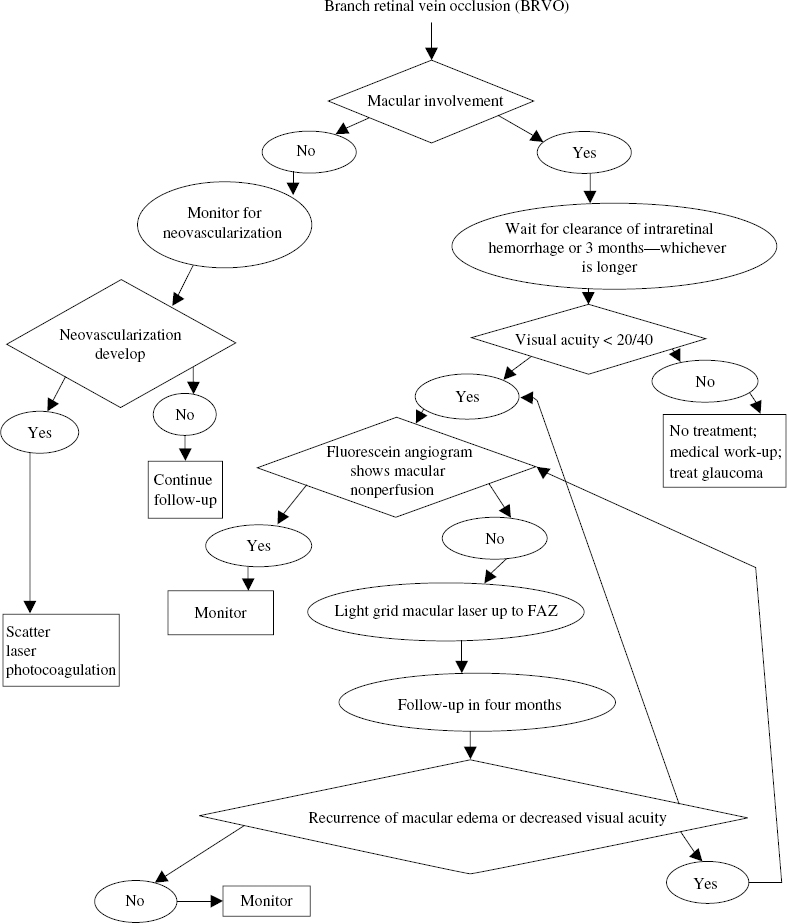
23 Choroidal neovascularization (CNV) is a significat cause of severe visual loss in patients with age-related macular degeneration (AMD) and ocular histoplasmosis syndrome (OHS). The Macular Photocoagulation Study (MPS) was a randomized, controlled clinical trial designed to determine whether laser photocoagulation of CNV prevents severe visual loss (SVL). Several clinical trials were conducted. These included (1) extrafoveal CNV in patients with AMD, OHS and idiopathic CNV (ICNV); (2) juxtafoveal CNV in patients with AMD, OHS, and ICNV; and (3) new and recurrent subfoveal CNV in patients with AMD only. Three trials investigated the role of laser treatment to extrafoveal CNV in patients with AMD, OHS, and ICNV. To be eligible for these trials, patients were required to have angiographic evidence of a choroidal neovascular membrane (CNV) with the foveal edge 200 to 2500 from the center of the foveal avascular zone (FAZ). The visual acuity was 20/100 or better in the study eye. Eyes with peripapillary membrane were included, if the laser treatment spared at least 1.5 clock hours of peripapillary nerve fiber layer adjacent to the temporal half of the disc. Patients enrolled in the AMD trial had evidence of drusen. Patients in the OHS trial had at least one atrophic scar (histo spot). Patients in the ICNV trial had no drusen, histo spots, or other clinical findings that could account for the CNV. Eligible eyes were randomly assigned to argonlaser photocoagulation or no treatment. Argon blue-green laser was used to obliterate the entire neovascular complex, including any areas of blood or blocked hyperfluorescence. Treatment resulted in uniform whitening of the overlying retina. Treated patients were seen at 3 to 9 weeks to assess the adequacy of treatment. Treated patients were encouraged to monitor their vision with an Amsler grid and report any new distortion or enlargement of scotoma. Additional treatment was promptly applied when persistence or recurrence of a treated membrane was found. The primary outcome measure for the MPS was occurrence of SVL, defined as a loss of six or more lines of visual acuity (equivalent to a quadrupling of the visual angle). The AMD trial enrolled 236 eyes. Compared with observation, laser photocoagulation significantly reduced the risk of SVL during the entire follow-up period: 47% versus 62% at 3 years and 46% versus. 64% at 5 years.1–3 After 5 years, the mean visual acuity was 20/125 in the laser-treated eyes and 20/200 in untreated eyes. The recurrence rates for the treated eyes were high: 24% at 6 months and 52% at 2 years.4 The OHS trial enrolled 232 eyes. Laser photocoagulation significantly reduced the risk of SVL during the entire follow-up period: 10% versus 45% at 3 years and 12% versus 42% at 5 years.2,3,5 After 5 years, the mean visual acuity was 20/40 in the laser-treated eyes and 20/80 in untreated eyes. The recurrence rates for treated eyes were 22% at 6 months and 28% at 2 years.4 Sixty-seven eyes were enrolled in the ICNV trial. There was a trend for laser photocoagulation to reduce the risk of SVL: 20% versus 36% at 3 years and 23% versus 48% at 5 years.2,3,6 These results were not statistically significant because of the small sample size. After 5 years, the mean visual acuity was 20/64 in the laser-treated eyes and 20/80 in untreated eyes. The recurrence rates for treated eyes were 15% at 6 months and 28% at 2 years.4 Three trials investigated the role of krypton laser treatment to juxtafoveal CNV in patients with AMD, OHS, and ICNV. Krypton-red laser was used for treatment within the FAZ because the red wavelength is not absorbed by the xanthophyll in the inner retina. To be eligible for these trials, patients were required to have angiographic evidence of neovascularization with foveal edges between 1 and 199 μ from the center of the FAZ or between 200 and 2500 μ from the center if associated blood or blocked fluorescence extended within 200 μ of the FAZ center. These lesions could have blood or blocked fluorescence due to pigment that extended through the entire FAZ. The visual acuity was 20/400 or better in the study eye. Eyes with peripapillary membranes were included if the laser treatment spared at least 1.5 clock hours of peripapillary nerve-fiber layer adjacent to the temporal half of the disc. Patients enrolled in the AMD trial had evidence of drusen. Patients in the OHS trial had at least one atrophic scar (histo spot). Patients in the ICNV trial had no drusen, histo spots, or other clinical findings that could account for the CNVM. Eligible eyes were randomly assigned to krypton-laser photocoagulation or no treatment. Treatment required a fluorescein angiogram taken within 72 hours and retrobulbar anesthesia. Krypton-red laser was used to obliterate the area of hyperfluorescence and obtain uniform whitening of the overlying retina. Unlike the extrafoveal trials, treatment of blood or blocked fluorescence associated with neovascularization was not required. Treatment extended 100 μ beyond the edge of neovascularization except when closer than 100 μ to the fovea. Treated patients were seen at 2, 4, and 6 weeks following treatment. Fluorescein angiography was obtained to assess the adequacy of treatment. Treated patients were encouraged to monitor their vision with an Amsler grid and report any new distortion or enlargement of scotoma. If persistence or recurrence of a treated membrane was found and met the initial treatment criteria, additional treatment was promptly applied. The AMD trial comprised 496 eyes. There was a trend for laser photocoagulation to reduce the risk of SVL: 49% versus 58% at 3 years and 52% versus 61% at 5 years.7,8 After 5 years, the median visual acuity was 20/200 in laser treated eyes and 20/250 in untreated eyes. The benefit of laser was greater in patients who were not hypertensive.7 For these patients, 38% of the treated eyes lost six or more lines of visual acuity at 5 years compared with 70% of untreated eyes. For the hypertensive patients, there was no apparent benefit for treatment. The estimated rate of persistence or recurrence for treated eyes was 78% at 5 years.8 The treatment benefit for these eyes might have been greater if subfoveal recurrences had been treated. Eyes with only classic CNV benefitted more than the other subgroups in the study.9 At 3 years, this subgroup showed severe visual loss in 48% of the treated eyes and 71% of the untreated eyes. When occult CNV was present, treatment of only the classic CNV was not beneficial. The study recommended treatment of juxtafoveal lesions with classic CNV but no occult CNV with laser photocoagulation.9 The OHS trial comprised 288 eyes. Laser photocoagulation significantly reduced the risk of SVL during the follow-up period: 5% versus 25% at 3 years and 12% versus 28% at 5 years.8,10 After 5 years, the median visual acuity was 20/40 in the laser treated eyes and 20/64 in untreated eyes. The rate of persistence or recurrence for treated eyes was 33% at 5 years.8 The ICNV trial comprised 49 eyes. Laser photocoagulation appeared to reduce the risk of SVL: 10% versus 37% at 3 years and 21% versus 34% at 5 years.8,11 These results were not statistically significant because of the small sample size. After 5 years, the mean visual acuity was 20/50 in the laser-treated eyes and 20/80 in untreated eyes. The rate of persistence or recurrence for treated eyes was 22% at 5 years.8 Treated patients were seen at 3 and 6 weeks following treatment and fluorescein angiography was obtained. Additional treatment was promptly applied when persistence or recurrence of a treated membrane was found and met the initial treatment criteria. The study comprised 206 eyes. Immediately following treatment, treated eyes lost more vision than untreated eyes.12,13 At 3 months, the rates of SVL were 14% for the treated eyes and 9% for the untreated eyes. After the 3-month follow-up visit, treated eyes had relatively stable visual acuity, and the untreated eyes continued to lose visual acuity. The rates of SVL were 9% at 2 years and 12% at 3 years for the treated eyes, compared with 28% at 2 years and 34% at 3 years for the untreated eyes. The rate of persistence or recurrence for treated eyes was about 50% at 3 years.14 There was no difference between the argon-green and krypton-red treated eyes.15 This trial investigated the role of laser treatment for new neovascularization directly under the fovea. Patients with well-demarcated subfoveal CNV smaller than 3.5 standard MPS disc areas were randomized to laser photocoagulation or no treatment. Eyes in the treatment group were randomized to receive kryptonred or argon-green laser. Laser treatment covered the entire neovascular lesion and extended 100 μ beyond the edge of the lesion. Treated patients were seen at 3 and 6 weeks following treatment and fluorescein angiography was obtained. If persistence or recurrence of a treated membrane was found and met the initial treatment criteria, additional treatment was promptly applied. The study enrolled 373 eyes. Initially, treated eyes lost more visual acuity than the untreated eyes.16,17 At 3 months, the rates of SVL were 20% for the treated eyes and 11% for the untreated eyes. By 1 year after study entry, however, treated eyes had lost less visual acuity than untreated eyes. This benefit persisted for at least 3 additional years.13 The rates of SVL were 20% at 2 years and 22% at 4 years for the treated eyes compared with 37%% at 2 years and 47% at 4 years for the untreated eyes. The rate of persistence or recurrence for treated eyes was about 50% at 3 years.14 No difference was found between the argon-green and krypton-red treated eyes.15 When subgroups of patients with different combinations of initial visual acuity and lesion size were analyzed, four distinct patterns of visual acuity loss were identified and are rated by group18 (Fig. 23–1). Group A eyes were characterized by less loss of visual acuity in treated eyes than in untreated eyes at every follow-up examination. These eyes were excellent candidates for treatment. Group B was characterized by more loss of visual acuity in treated eyes than untreated eyes at 3 months but less loss of visual acuity in treated eyes than untreated eyes at 12 months and thereafter. It was found that eyes treated 1 year after the initial treatment maintain a visual acuity benefit. Alternatively, these eyes may be followed up and treated when the vision starts to decrease or the lesion size starts to increase. Group C was characterized by relatively low proportions of large decreases in visual acuity in both treated and untreated eyes and by slightly less loss of visual acuity in treated eyes during follow-up. These lesions are good candidates for treatment because the risk of further loss with treatment is low and there is a small treatment benefit. Group D was characterized by substantially more visual acuity loss in treated eyes than in untreated eyes at 3 months through 18 months and little difference between the two groups thereafter. These lesions are poor candidates for treatment. The Treatment of AMD with Photodynamic Therapy (TAP) Study Group reported the 1- and 2-year results of two multicenter randomized clinical trials investigating the role of photodynamic therapy (PDT) in treatment of subfoveal CNV secondary to AMD. Eligible patients had subfoveal CNV caused by AMD measuring 5400 μ or less in greatest linear diameter with evidence of some classic CNV and best-corrected visual acuity of about 20/40 to 20/200. Six hundred nine patients were randomly assigned to PDT with Verteporfin or placebo. Eyes treated with PDT were significantly more likely to lose fewer than 15 letters of visual acuity than untreated eyes: 61% versus 46% at 1 year and 53% versus 38% at 2 years. In subgroup analysis, the eyes with 50% or more classic CNV had a significant treatment benefit, but those with greater than 0% and less than 50% had no benefit from treatment.19,20 Eyes with extrafoveal and juxtafoveal CNV secondary to OHS or idiopathic causes are treated with laser photocoagulation according to the MPS guidelines. Subfoveal CNV secondary to OHS or idiopathic causes is not treated with laser photocoagulation. The Submacular Surgery Trial is investigating the role of surgical removal of CNV for these lesions. Some physicians may consider treating these lesions with PDT. Eyes with extrafoveal or juxtafoveal CNV secondary to AMD that are 100% classic are treated with laser photocoagulation according to MPS guidelines. Generally, lesions containing nearly 100% occult CNV are not considered for PDT based on the verteporfin in PDT (VIP) Study.20a For subfoveal CNV secondary to AMD, there may be more than one treatment option. Laser photocoagulation may be used to treat well-defined subfoveal CNV with smaller size and poorer visual acuity according to MPS guidelines, but many consider PDT may be used to treat subfoveal CNV with more than 50% classic component according to the TAP study guidelines. The Branch Retinal Vein Occlusion Study (BRVOS) was a multicenter, randomized clinical trial designed to investigate efficacy of (1) scatter-laser photocoagulation in the management of neovascularization and vitreous hemorrhage and (2) focal laser for the treatment of macular edema in patients with branch retinal vein occlusions (BRVO). To be eligible, patients must have had a BRVO 3 to 18 months earlier, macular edema reducing visual acuity to 20/40 or worse, sufficient clearing of intraretinal hemorrhage to permit evaluation of fluorescein angiography and safe laser photocoagulation, and absence of hemorrhage directly in the fovea. Eligible patients were randomized to focal laser photocoagulation or observation. Treatment consisted of argon-laser photocoagulation applied in a grid pattern over the area of capillary leakage in the macular region identified by fluorescein angiography. Photocoagulation was not extended closer to the fovea than the edge of the foveal avascular zone (FAZ) and not peripheral to the major vascular arcades. Patients were evaluated at the time of entry into the study and every 4 months thereafter. Additional photocoagulation was applied at each 4-month visit if untreated leaking areas and foveal edema persisted with continued loss of visual acuity. The primary outcome measure for this part of the study was a gain of two or more lines of visual acuity at two or more consecutive 4-month follow-up visits. Between July 1977 and February 1984, 139 eligible eyes from 139 patients were recruited: 71 randomized to laser and 68 to observation. The average duration of follow-up was 3.1 years. Treated eyes with macular edema were more likely to gain two or more lines of visual acuity than control eyes.21 At 3 years, 65% of treated eyes gained two or more lines of visual acuity compared with 37% of control eyes. Treated eyes were more likely to have visual acuity of 20/40 or better (60% versus 34%), and less likely to have visual acuity of 20/200 or worse (12% versus 23%). The average visual acuity was 20/40 to 20/50 in the treated group and 20/70 in the control group. Treatment was beneficial for all eyes regardless of the duration of occlusion.21 Eyes at risk for development of neovascularization were enrolled in group I and those at risk for vitreous hemorrhage were enrolled in group II. To be eligible, patients must have had a BRVO 3 to 18 months earlier with at least five disc areas of retinal involvement and sufficient clearing of intraretinal hemorrhage to permit safe laser photocoagulation. In addition, group I eyes were required to have no neovascularization, and group II eyes were required to have disc or peripheral neovascularization. Eligible eyes in each group were randomized to scatter laser photocoagulation or to observation. Treatment consisted of argon laser to the involved segment to achieve “medium” white burns (200 to 500 μ in diameter) spaced one burn width apart, extending no closer than two disc diameters from the center of the fovea. Patients were evaluated at the time of entry into the study and every 4 months thereafter. The outcome measure for group I was neovascularization, defined as any amount of surface or preretinal new vessel formation. The outcome measure for group II was vitreous hemorrhage, defined as any amount of preretinal or vitreous hemorrhage. From July 1977 to February 1985, 319 eligible eyes from 316 patients were recruited to group I; 160 eyes were randomized to laser and 159 eyes to observation. The average duration of follow-up was 3.7 years. Treated eyes were less likely to develop neovascularization than the control eyes.22 Retinal neovascularization developed in 19 (12%) of the treated eyes compared with 35 (22%) control eyes. The probability of neovascularization was greater in eyes with nonperfusion on fluorescein angiography. All eyes that developed neovascularization had at least five disc diameters of capillary nonperfusion on initial or follow-up fluorescein angiography. Eighty-two eligible eyes from 82 patients were recruited to group II from July 1977 to February 1985. Forty-one eyes were randomized to laser and 41 eyes to observation. The average duration of follow-up was 2.8 years. Treated eyes were significantly less likely to develop vitreous hemorrhage.22 Vitreous hemorrhage developed in 29% of the treated eyes opposed to 61% of the control eyes. These results suggested similar rates of vitreous hemorrhage whether laser treatment is applied before or after development of neovascularization. Application of laser after development of neovascularization would obviate unnecessary treatment to many eyes that never would develop these complications. For patients who have a decreased visual acuity of 20/40 or worse following a BRVO, the study recommended waiting for sufficient clearing of retinal hemorrhages to obtain high-quality fluorescein angiography. If macular nonperfusion is the cause of visual loss, no treatment is offered. If macular edema is the cause of visual loss, and vision continues to be 20/40 or worse without spontaneous improvement, grid macular photocoagulation is recommended. For patients who have a BRVO with retinal involvement larger than five disc diameters, the study recommended waiting for sufficient clearing of retinal hemorrhage for high-quality fluorescein angiography. If more than five disc diameters of nonperfusion are present, patients should be followed at 4-month intervals for development of neovascularization. If neovascularization develops and is confirmed, scatter-laser photocoagulation treatment is recommended in the involved quadrant. General guidelines for management of patients with BRVO are outlined in Figure 23–2. The Central Retinal Vein Occlusion Study (CRVOS) was a multicenter, randomized clinical trial designed to evaluate (1) the timing of panretinal photocoagulation (PRP) for ischemic central vein occlusion; (2) the role of macular grid-pattern photocoagulation for macular edema with reduced visual acuity resulting from CRVO; (3) the natural history of eyes with CRVO. This study was not designed to evaluate the efficacy of PRP in the treatment of anterior segment neovascularization, that is, neovascularization of the iris or angle (NVI/NVA), in patients with CRVO. To be eligible, patients must have had CRVO of less than 1 year in duration with at least 10 disc areas of retinal nonperfusion. Visual acuity of light perception or better and intraocular pressure lower than 30 mm Hg were required. Eligible eyes were randomized to either immediate PRP or laser only if NVI/NVA developed. Prophylactically treated eyes received additional PRP if NVI/NVA developed. Treatment consisted of 1000 to 2000 evenly spaced laser spots applied to achieve moderately intense white burns (500 to 1000 μ in diameter) spaced 0.5 to 1 burn apart, avoiding retinal hemorrhage or retinal vessels. Treatment was applied no closer than two disc diameters from the center of the fovea and no closer than 500 μ nasal to the disc. Patients were evaluated at the time of entry in the study and monthly for the first 6 months after entry. After the first 6 months, patients were seen at 2 months and every 4 months thereafter. At each visit, undilated slit-lamp examination and gonioscopy were performed to examine for anterior segment neovascularization. The primary outcome of the study was anterior segment neovascularization defined as two clock hours of NVI or any NVA. From August 1988 to August 1992, 181 eligible eyes from 180 patients were recruited. Ninety eyes were randomized to prophylactic PRP and 91 eyes to PRP when anterior neovascularization developed. The follow-up period was 3 years, except for patients recruited after February 28, 1991, who were followed up until February 28, 1994. After adjusting for baseline variables, there was a trend for decreased risk of anterior segment neovascularization in the early treated group that was not statistically significant.22 Anterior segment neovascularization developed in 18 (20%) of 90 eyes treated prophylactically and in 32 (35%) of 91 untreated eyes. Regression of anterior segment neovascularization within 1 month was more than four times as likely in response to treatment in previously untreated eyes than in response to retreatment in prophylactically treated eyes.22 Four (22%) of 18 eyes in the early treatment group showed prompt regression within 1 month, compared with 18 (56%) of 32 eyes in the group that did not receive early treatment. Long-term regression of neovascularization occurred in about 90% of eyes in both groups. Only four eyes in each group had neovascular glaucoma with persistent anterior segment neovascularization. The most important risk factor for both occurrence and persistence of anterior segment neovascularization was the amount of nonperfused retina measured at study entry.22 The risk of anterior segment neovascularization ranged from 16% in eyes with 10 to 29 disc areas of nonperfusion to 52% for eyes with 75 or more areas of nonperfusion at baseline. In all but two patients, anterior segment neovascularization occurred in the first year of follow-up, usually within the first 3 months. In most eyes, NVI was present at the time anterior segment neovascularization was discovered. NVA without NVI was present in three patients. To be eligible, patients must have had CRVO of at least 3 months in duration with visual acuity between 20/50 and 5/200 as a result of macular edema without capillary nonperfusion involving the fovea and documented by fluorescein angiography. Eligible eyes were randomized to either grid-pattern laser treatment or to observation. From August 1988 to August 1992, 155 eyes of 155 eligible patients were recruited; 75 were randomized to grid-pattern laser treatment and 78 to observation. The follow-up period was 3 years, except for patients recruited after February 28, 1991, who were followed up until February 28, 1994. Although treatment significantly reduced the amount of macular edema present on fluorescein angiography, there was no significant difference between treated and untreated patients in level or change in visual acuity.23 The mean visual acuity score at baseline was 42 letters for the treated eyes and 44 letters for untreated eyes. The mean visual acuity score at 36 months was 39 letters for treated eyes and 43 letters for the untreated eyes. The mean change in visual acuity score, from baseline to the 36-month visit, was a loss of four letters in treated eyes and a loss of three letters in untreated eyes. The visual acuity improved by two or more lines in 17 (23%) of the treated eyes. Eleven of these 17 eyes came from patients younger than 60 years. There was a trend for treatment to be more beneficial in patients younger than 60 years of age, but this finding was not statistically significant when adjusted for potential interactions. Treatment significantly reduced the amount of macular edema measured on fluorescein angiography.23 At the 12-month annual visit, no measurable macular edema was present in 21 (31%) of 68 treated eyes, whereas all of the 72 untreated eyes showed some macular edema (p < 0.0001). To determine their natural history, 547 perfused eyes (defined as fewer than 10 disc areas of nonperfusion on fluorescein angiography) were monitored to determine their natural history. About one third of these eyes (185 of 547, or 34%) developed ischemia (defined as greater than 10 disc areas of nonperfusion) at 3 years.24 Conversion to ischemia was most rapid in the first 4 months after enrollment25; 81 (15%) progressed to nonperfusion by the 4-month visit. Most eyes with sufficient intraretinal hemorrhage to prevent determination of perfusion status were determined to be ischemic.25 At the 4-month visit, 38 (83%) of 46 such eyes demonstrated at least 10 disc areas of nonperfusion (28 eyes) or developed NVI/NVA before the retinal status could be determined (10 eyes). In the CRVOS population of 714 eyes, NVI/NVA developed in 117 eyes (16%) during the course of the follow-up. In 10 eyes, new vessels failed to regress in response to photocoagulation. For eyes initially categorized as nonperfused or indeterminate, NVI/NVA developed in 35% (61/176), whereas 10% (56/538) of eyes initially categorized as perfused developed NVI/NVA. The risk of NVI/NVA increased in relation to the number of disc areas of nonperfusion and initial visual acuity. In patients with recent (less than 1 month) onset of vein occlusion, visual acuity of less than 20/200 alone contained virtually all the predictive information. The eyes with better initial visual acuity had a greater chance of having better final visual acuity. Visual acuity worse than 20/200 was highly correlated with the presence and development of ischemia. The annual risk of any vascular occlusion in an unaffected fellow eye was about 0.9% per year. At the initial examination, it is important to perform undilated slit-lamp examination and gonioscopy to evaluate for NVI/NVA. If NVI/NVA is present at the initial examination, PRP should be considered. Intraocular pressure should be measured because of the reported association of CRVO and glaucoma. Patients are recommended to be under general medical care, with yearly measurement of blood pressure. Color photography and fluorescein angiography are not essential but might be useful in providing a good baseline and identifying the perfusion status. In patients with bilateral simultaneous CRVOs, the possibility of hyperviscosity should be considered. Patients with unilateral CRVO are notified that the risk of a vascular occlusion in the fellow eye is about 1% per year. Visual acuity is the most important factor in determining a prognosis and follow-up plan. If the patient’s initial visual acuity is 20/40 or better, there is a good likelihood of retaining good acuity. If the visual acuity is 20/50 to 20/200, the visual acuity is variable; the visual acuity may improve (19%), remain unchanged (44%), or deteriorate (37%). If the initial visual acuity is worse than 20/200, prognosis for vision is poor (80% remain at that level or worse). Visual acuity is more important than the initial fluorescein angiogram in determining prognosis and clinical management of the patient, because more than one third of initially perfused eyes develop nonperfusion and NVI/NVA. If the visual acuity is 20/40 or better, the patient is seen every 1 to 2 months for 6 months, tapering the interval to annual follow-ups as the patient’s condition stabilizes. Patients with visual acuity of less than 20/200 are seen every month for the initial 6 months. Patients with visual acuity of 20/50 to 20/200 are seen either monthly or bimonthly, depending on the level of visual acuity and the progression or improvement of the CRVO. If the visual acuity drops to less than 20/200, it is likely that nonperfusion has developed and the patient should be followed monthly for 6 months. Treatment with PRP is instituted only at the first sign of neovascularization. After PRP, the patient is monitored closely every 2 to 4 weeks to ensure that the NVI is not advancing. If NVI recurs, photocoagulation should be added in untreated areas of the retina. Focal laser photocoagulation is not recommended for patients with decreased visual acuity secondary to macular edema. Some investigators consider this treatment in patients younger than 65 years. General guidelines for management of patients with CRVOs are outlined in Figure 23–3.
Clinical Trials in Retina
Treatment of Choroidal Neovascularization (Macular Photocoagulation Study and Others)
Why Was the Macular Photocoagulation Study Conducted?
How Was the Extrafoveal Choroidal Neovascularization Trial Conducted?
Does Laser Photocoagulation Preserve Vision in Patients with Extrafoveal Choroidal Neovascularization?
AGE-RELATED MACULAR DEGENERATION TRIAL
OCULAR HISTOPLASMOSIS TRIAL
IDIOPATHIC CHOROIDAL NEOVASCULARIZATION TRIAL
How Was the Juxtafoveal Choroidal Neovascularization Trial Conducted?
Does Krypton Laser Photocoagulation Preserve Vision in Patients with Juxtafoveal Choroidal Neovascularization?
AGE-RELATED MACULAR DEGENERATION TRIAL
OCULAR HISTOPLASMOSIS TRIAL
IDIOPATHIC CHOROIDAL NEOVASCULARIZATION TRIAL
How Was the Recurrent Subfoveal Choroidal NeovascularizationTrial for Age-Related Macular Degeneration Conducted?
Does Laser Photocoagulation Preserve Vision in Patients with Recurrent Subfoveal Choroidal Neovascularization
How Was the New Subfoveal Choroidal Neovascularization Trial for Age-Related Macular Degeneration Conducted?
Does Laser Photocoagulation Preserve Vision in Patients with New Subfoveal Choroidal Neovascularization?
Patterns of Visual Acuity
GROUP A
GROUP B
GROUP C
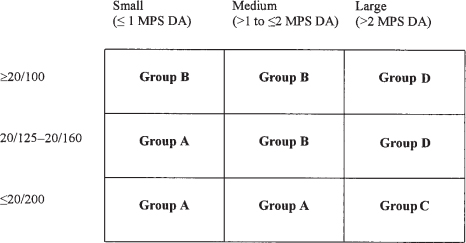
GROUP D
Is Photodynamic Therapy with Verteporfin Effective in Treatment of Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration?
What Are the Recommendations for Treatment of Choroidal Neovascularization?
Branch Retinal Vein Occlusion Study
Why Was the Branch Retinal Vein Occlusion Study Conducted?
How Was the Trial of Focal Laser for Treatment of Macular Edema Conducted?
Can Photocoagulation Improve Visual Acuity in Eyes with Macular Edema Reducing Vision to 20/40 or Worse?
How Were the Trials of Laser Photocoagulation for Prevention of Neovascularization and Vitreous Hemorrhage Conducted?
Can Photocoagulation Prevent the Development of Neovascularization and Vitreous Hemorrhage?
What Were the Recommendations of the Branch Retinal Vein Occlusion Study for Management?
Central Retinal Vein Occlusion Study
Why Was the Central Retinal Vein Occlusion Study Conducted?
How Was the Trial of Panretinal Photocoagulation for Ischemic Central Retinal Vein Occlusion Study Conducted?
Should Panretinal Photocoagulation for Ischemic Central Retinal Vein Occlusion Be Performed at Diagnosis or Only When Anterior Segment Neovascularization Develops?
How Was the Trial of Grid-Pattern Laser Photocoagulation for the Treatment of Macular Edema Secondary to Central Retinal Vein Occlusion Conducted?
What Is the Role of Grid-Pattern Laser Photocoagulation in the Treatment of Macular Edema Secondary to Central Retinal Vein Occlusion?
What Is the Natural History of Eyes with Central Retinal Vein Occlusion?
What Were the Recommendations for the Clinical Care of Patients with Central Retinal Vein Occlusion?
Ento Key
Fastest Otolaryngology & Ophthalmology Insight Engine