(1)
Department of Ophthalmology, St. Thomas’ Hospital, London, UK
11.1.1 Clinical Features
11.1.2 Vitreous Haemorrhage and CNV
11.1.5 Specific Complications
11.1.6 Success Rates
11.2.1 Introduction
11.2.2 Surgery
11.3 Summary
Abstract
Choroidal neovascular membranes (CNVs) most commonly occur with age-related macular degeneration (ARMD). Features of ‘dry’ AMD include hard drusen, soft drusen, retinal pigment epithelial disruption and geographic atrophy. Patients with extensive small drusen, non-extensive intermediate size drusen or pigment abnormalities have only a 1.3 % 5-year probability of progression to advanced AMD according to the ARED Study (Age-Related Eye Disease Study Research Group 2001). Those with extensive intermediate size drusen, at least one large druse, noncentral geographic atrophy in one or both eyes, or advanced AMD or vision loss due to AMD in one eye, are at risk of vision loss from advanced AMD in up to 50 % (large drusen with pigmentary changes) after 5 years (Ferris et al. 2005).
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-642-31872-6_11) contains supplementary material, which is available to authorized users.
11.1 Age-Related Macular Degeneration
11.1.1 Clinical Features
Choroidal neovascular membranes (CNVs) most commonly occur with age-related macular degeneration (ARMD). Features of ‘dry’ AMD include hard drusen, soft drusen, retinal pigment epithelial disruption and geographic atrophy. Patients with extensive small drusen, non-extensive intermediate size drusen or pigment abnormalities have only a 1.3 % 5-year probability of progression to advanced AMD according to the ARED Study (Age-Related Eye Disease Study Research Group 2001). Those with extensive intermediate size drusen, at least one large druse, noncentral geographic atrophy in one or both eyes, or advanced AMD or vision loss due to AMD in one eye, are at risk of vision loss from advanced AMD in up to 50 % (large drusen with pigmentary changes) after 5 years (Ferris et al. 2005).
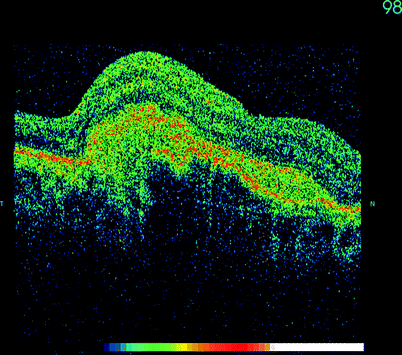


Fig. 11.1
OCT can demonstrate CNV membranes and be used to detect fluid around the membrane indicating a need for repeat anti-VEGF therapy. This membrane is currently dry

Fig. 11.2
Type 1 CNV is associated with age-related macular degeneration, presenting in all layers of the retina: sub-RPE in occult membrane, sub-neuroretina in classic membrane and possibly in the retina in retinal angiomatous proliferation (RAP)
Simplified AREDS scoring system:
1 or more large drusen (≥125 μm, width of a large vein at disc margin) in an eye = 1 risk factor
Any pigment abnormality in an eye = 1 risk factor
Risk factors summed across both eyes
The 5-year risk of developing advanced AMD in at least one eye:
0 factors
0.5 %
1 factor
3 %
2 factors
12 %
3 factors
25 %
4 factors
50 %
If no large drusen but intermediate drusen present in both eyes = 1 risk factor
This risk can be reduced by taking a cocktail of high-dose vitamins (commercially available in combination preparations) such as 500 mg vitamin C; 400 IU vitamin E; 15 mg beta-carotene (to be avoided in smokers or ex-smokers of less than 10 years because of an increased risk of lung carcinoma); zinc, 80 mg, as zinc oxide and copper, 2 mg, as cupric oxide (Age-Related Eye Disease Study Research Group 2001). This cocktail has been shown to reduce the chance of advancement in patients with high-risk characteristics by approximately 30 %.
Patient’s CNVs already in one eye are at particular risk of progressing to ‘wet’ ARMD with CNV production. The CNVs cause distortion and loss of vision with serous elevation of the retina, subretinal haemorrhage and finally disciform scar formation.
The CNVs are usually classified on fluorescein angiography into:
Classic: appear early and thought to be beneath the neuroretina
Nonclassic, indistinct and slower appearance: thought to be under the RPE
Mixed: can be either predominantly classic or nonclassic
The frequent bilaterality of the condition results in a high proportion of patients who are technically blind, with severe loss of central vision. For this reason, surgical approaches have been tried. However, these are much less commonly used since the effectivity of anti-VEGF treatments bevacizumab (Avery et al. 2006), pegaptanib (Chakravarthy et al. 2006) and ranibizumab (Rosenfeld et al. 2006a, b; Brown et al. 2006) has been proven. The last is now established as the therapy of choice for CNVs from AMD.
Vitreomacular traction is more common in eyes with exudative AMD (38 %) compared with nonexudative AMD (10 %) and PVD that are less common (21 and 68 %, respectively), suggesting to some investigators a role for the vitreous in exudative AMD (Robison et al. 2009).
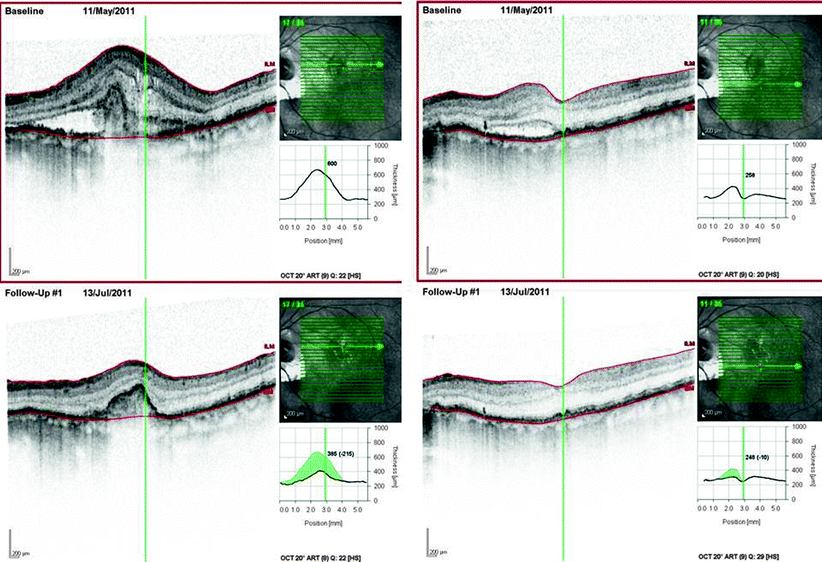

Fig. 11.3
A CNV shrunken with resolution of fluid leakage after ranibizumab injection, resulting in 20/20 vision
Table 11.1
∎
Drug | Structure | Dosage | Route of administration |
|---|---|---|---|
Antibody fragment | 0.5 mg/0.05 ml | Intravitreal | |
Pegaptanib (Macugen) | Aptamer (oligonucleotide) | 0.3–1.0 mg/0.1 ml | Intravitreal |
Bevacizumab (Avastin) | Complete immunoglobulin | 1.25 mg/0.05 ml | Intravitreal |
There may be situations where the surgery can be of use:
Vitreous haemorrhage with CNV and subretinal haemorrhage
Pneumatic displacement of subretinal haemorrhage
Failure of anti-VEGF regimes
11.1.2 Vitreous Haemorrhage and CNV
A patient with sudden onset vitreous haemorrhage with evidence of a large subretinal craggy mass on ultrasound is very likely to have suffered a subretinal bleed from a CNV from AMD (Orth and Flood 1982). The subretinal haemorrhage is usually in the macular area but occasionally is due to a peripheral CNV, and the macula is clear of blood. Removing the vitreous haemorrhage is useful to restore peripheral vision. The haemorrhage is often very thick and may be altered to an ochre colour seen in severe bleeds. The vitreous may or may not be detached. If attached, separate it from the retina as in macular hole surgery. If there are large bullae of subretinal haemorrhage, there is a temptation to remove some of the subretinal blood via retinectomy; however, this is often not very effective in removing the blood as fibrinous mass has a ‘cottage cheese’ consistency and cannot be washed out. The retinectomies may allow fluid blood to egress in the postoperative period causing severe hyphaema and the potential for raised IOP and corneal staining. A larger 180° retinectomy will allow access to the blood for removal but runs the risk of postoperative PVR, and visual recovery is unlikely to be much improved after prolonged surgery. If the subretinal blood is localised, leave it in situ; in most circumstances, it will remain localised, allowing the patient to achieve vision from unaffected parts of the retina.
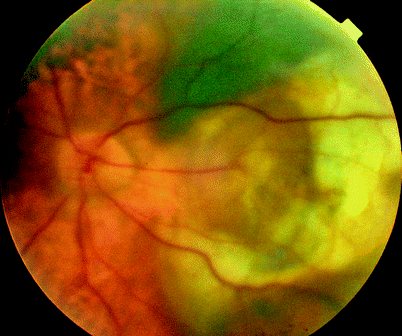
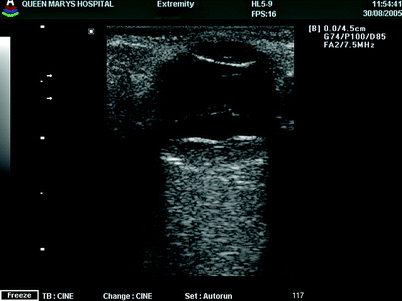
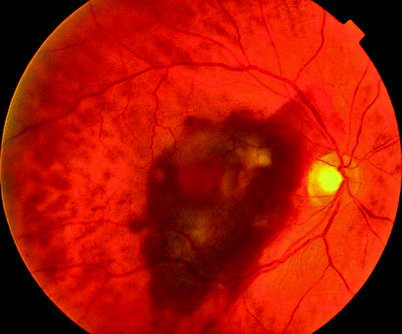

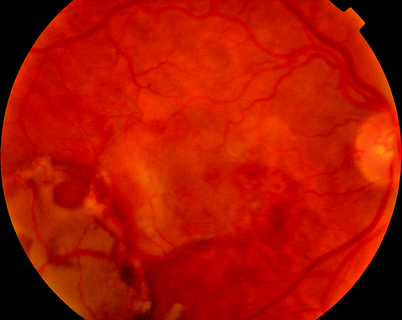
Fig. 11.4
Very large subretinal bleeds from CNV can cause severe vitreous haemorrhage. These are usually obvious on ultrasound as a crenated posterior mass

Fig. 11.5
An ultrasound of a patient with vitreous haemorrhage from subretinal bleed and age-related CNV, note the PVD and large craggy mass at the posterior pole

Fig. 11.6
Subretinal blood can occur from a number of reasons including type I choroidal neovascular membranes and macular aneurysms. Often, the blood will break through the retina, as seen in this case, into the posterior vitreous gel
Often these patients are on antiplatelet or anticoagulation therapy (Kuhli-Hattenbach et al. 2010). Examine the other eye for evidence of AMD.
Preoperative injection of intravitreal tissue plasminogen activator can be used to try to liquefy the clot (Oshima et al. 2007).
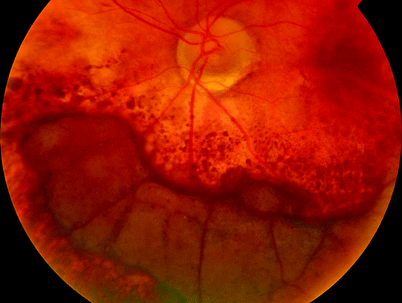

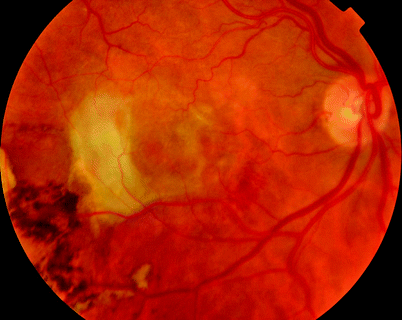
Fig. 11.7
An eccentric CNV caused a vitreous haemorrhage after a subretinal haemorrhage which did not affect the macula
11.1.3 Pneumatic Displacement of Subretinal Haemorrhage
A bleed from a CNV may spread under the macula, giving a rise to a large central scotoma. It is possible to facilitate resorption of the haemorrhage and perhaps to displace the bleed away from the fovea by performing a PPV and gas. The patient is required to posture upright to allow the gas bubble to act on the haemorrhage displacing it inferiorly. There is a debate whether either intravitreal tissue plasminogen activator (tPA, 0.05 ml, 50 μg) or subretinal tPA should be injected to facilitate the breakup of the clot (Gopalakrishan et al. 2007; Ohji et al. 1998; Hesse et al. 1996; Singh et al. 2006). The molecular size of tPA is similar to bevacizumab which is able to cross the retina in AMD. Any agent injected into the vitreous cavity in a vitrectomised eye however tends to have a shorter half-life because of more rapid clearance of the drug in the fluid-filled vitreous cavity in comparison to the gel-filled cavity. If injecting tPA into the subretinal space, the drug can be inserted using a 40-gauge needle to raise a bleb of fluid under the retina. As with any injection under the macula, take care that the pressure rise in the subretinal space does not ‘blow’ a hole in the fovea, the weakest point in the macula.
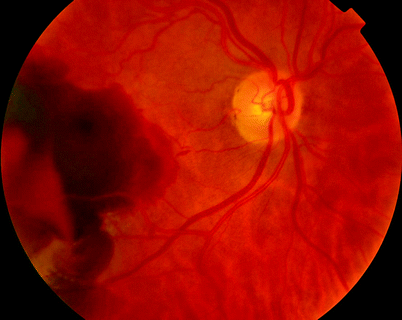
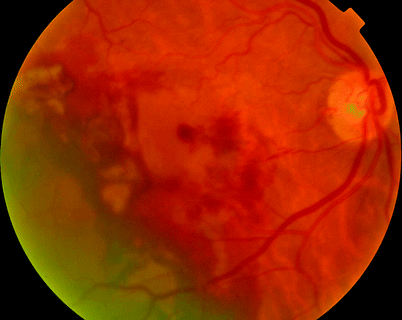

Fig. 11.8
Some CNV in ARMD will bleed into the subretinal space; a PPV and gas insertion (with or without subretinal tissue plasminogen activator, TPa) can displace the blood away from the fovea, thereby increasing vision despite a persistent CNV. This sequence of images shows the preoperative status and sequential improvement in the retinal appearance over months (see Figs. 11.9–11.11)

Fig. 11.9
See previous figure
11.1.4 Surgery for Failed Anti-VEGF Therapy
Introduction
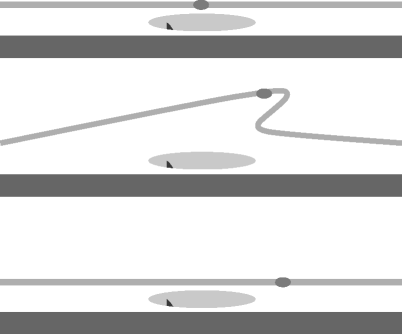
Fig. 11.12
The principle of retinal translocation is to move the fovea onto an undamaged area of RPE
Surgical approaches have been applied to subfoveal CNV. The most established strategy has been 360° macular translocation (Machemer and Steinhorst 1993a, b; Eckardt et al. 1999). Success rates are reportedly 33 % improvement in vision, but the usual benefit is for reading speed (bdel-Meguid et al. 2003; Wong et al. 2004) with 10 % recurrence. Translocation for geographic atrophy has been complicated by the rapid return of the atrophy in the translocated macula (Khurana et al. 2005).
Various other methods such as limited macular translocation and transplant of a RPE and choroidal patch are of unproven benefit (van Meurs et al. 2004; Stanga et al. 2002; Lappas et al. 2004; Angunawela et al. 2005; Thomas et al. 1992; Phillips et al. 2003; Fujii et al. 2003; Pieramici et al. 2000; Joussen et al. 2006). Removal of peripapillary CNV on its own is of doubtful benefit (Bains et al. 2003). Clinicopathologic studies of eyes with surgically removed CNV reveal breaks in Bruch’s membrane and persistent sub-RPE CNV (Grossniklaus et al. 2006).
In most circumstances, the monthly injection of ranibizumab is the current treatment of choice for CNV from AMD. However, the long-term dosage regimes for these agents are not well understood, and combination therapy with intravitreal steroid and PDT are being investigated to try to reduce the need for years of repeated injections. In those patients with loss of vision in their second affected eye who are not responsive to these agents, there may still be a role for 360° macular translocation surgery.
360° Macular Translocation
Table 11.2
Difficulty rating for 360° macular translocation
Difficulty rating | Very difficult |
Success rates | Low |
Complication rates | High |
When to use in training | Late |
Additional surgical steps
Phacoemulsification of the lens with IOL
Artificial inducement of retinal detachment via infusion of fluid through a retinotomy
Removal of the CNV
Drainage of subretinal fluid and insertion of heavy liquids
Translocation of the retina
Reattachment of the retina
360° Laser retinopexy
Silicone oil tamponade
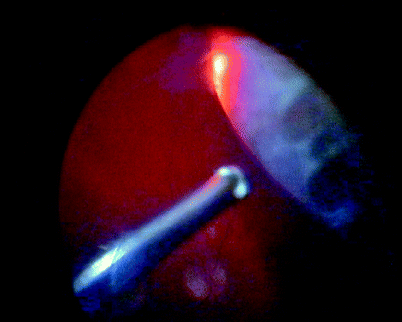
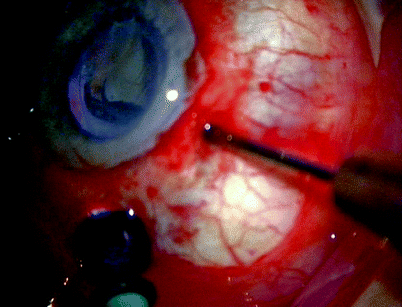
Fig. 11.13
For 360° macular translocation after cataract extraction and lens implantation, trim the vitreous base with a high-speed cutter. With a 40-gauge cannula, create a retinal detachment and then spread the SRF with fluid–air exchange. Create the 360° retinectomy, dissect off the CNV, rotate the retina, laser the retinectomy and insert silicone oil (see Figs. 11.14–11.19)
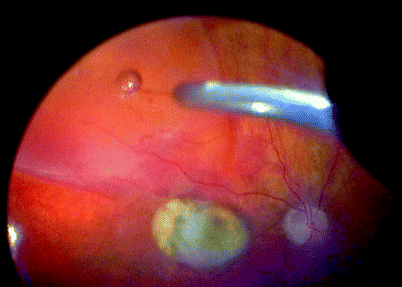
Fig. 11.14
See previous figure