CHAPTER 78 Chemotherapy and Targeted Biologic Agents for Head and Neck Cancer
Squamous Cell Carcinoma
The serendipitous identification of the metal compound cisdiamine-dichloroplatinum (II) (cisplatin) as a potential anticancer agent by Rosenberg1 in 1968 spurred clinical research efforts to test new agents and to develop combination chemotherapy regimens for the palliative benefit of patients with locally recurrent and metastatic cancers. After highly active chemotherapy regimens were identified, drug therapy was incorporated into combined-modality approaches for treating the newly diagnosed patient. The objectives then changed, with more focus on increasing tumor eradication in order to improve survival time. It became clear that if administered as the initial treatment, chemotherapy could result in rapid regression of tumor in the majority of patients without substantially increasing the morbidity of subsequent surgery or irradiation. Moreover, in a proportion of patients with responding tumors, there is no histologic evidence of tumor in resected specimens. This observation led to consideration of altering standard surgical approaches to some primary tumors with the objective of organ preservation. In addition to investigational trials using chemotherapy before definitive local therapy, adjuvant radiation and chemotherapy administered after surgical resection and chemotherapy used as a radiosensitizer concomitant with radiotherapy have been under active study.
A new class of drugs (i.e., targeted biologic agents or molecular targeted agents) has become available for the treatment of cancer. These compounds have a distinct mechanism of action, blocking specific molecular pathways key to the carcinogenic process. As a result, molecularly targeted drugs exhibit antiproliferative, proapoptotic, antiangiogenic and chemotherapy/radiotherapy-sensitizing properties that contribute to antineoplastic effects. Adverse events associated with these drugs are very distinct from the classic side effects observed with cytotoxic agents and are largely determined by the target pathway inhibited. Bonner and colleagues2 demonstrated that cetuximab (a biologic agent targeted at EGFR), given concomitantly with radiotherapy, improves survival of patients with head and neck cancer, with no increased incidence of severe radiotherapy-induced side effects. This study can be accepted as proof of principle that biologic agents may become substantial therapeutic agents for this disease. Moreover, the report inaugurates an exciting era for both research and clinical management of SCCHN.
A role for human papillomavirus (especially subtype 16) has now been recognized in the pathogenesis of a substantial percentage of oropharyngeal squamous cell carcinomas.3 Transmission is sexual. The E6 and E7 viral oncogenes are overexpressed and associated with degradation of wild type p53. Indeed, molecular studies4 suggest that human papillomavirus–positive tumors have relatively minimal genomic alterations and that, stage for stage, prognosis is better than that for human papillomavirus-negative tumors, which tend to have multiple cytogenetic changes, p53 mutations, loss of p16, cyclin D1 overexpression, and increased surface expression of EGFR.
Combined-Modality Therapy
Concomitant Chemoradiation
For early-stage cancers of the larynx, definitive radiotherapy or conservation laryngeal surgery produces excellent tumor control with preservation of speech and swallowing functions. These approaches are discussed in Chapter 112. A majority of patients with SCCHN present with stage III to IV M0 disease and may require extensive or radical surgery, with potential deleterious effects on speech or swallow function, depending upon the primary tumor site. Of course, general appearance may also be altered. In the 1990s, the dual objectives of improving survival and preserving organs represented a major challenge and became a focus for clinical trial development. This led to the Department of Veterans Affairs (VA) Laryngeal Cancer Study,5 which tested a nonsurgical alternative for locally advanced squamous cancer of the larynx, matching induction chemotherapy followed by definitive radiation against laryngectomy with postoperative radiotherapy. Thus, initial drug treatment as a component of primary treatment in sequence before definitive radiotherapy was tested in a prospective, phase III randomized trial.
Induction chemotherapy with cisplatin-based regimens is highly active, with clinical partial and complete responses observed in 80% to 90% of previously untreated patients. It was postulated that a substantial response to initial treatment with chemotherapy would lead to an improvement of therapeutic efficacy for surgery or radiotherapy. If so, radiotherapy might then become an acceptable alternative to laryngectomy with preservation of prospects for tumor control and organ function. In the VA Study, 332 patients with stage III or IV squamous cell carcinoma of the larynx were randomly assigned to receive either induction chemotherapy consisting of cisplatin and 5-FU followed by radiotherapy or surgery with postoperative radiotherapy.5 Patients who experienced no tumor response to chemotherapy or those who had locally persistent or recurrent cancer underwent salvage laryngectomy. Two-year survival for both treatment groups was 68%, and 41% of patients randomly assigned to the experimental arm were alive with a functional larynx at 2 years. Thus, the efficacy of chemotherapy followed by radiotherapy (with surgical salvage) was similar to that of surgery followed by radiotherapy, establishing organ preservation as a realistic goal of nonsurgical treatment administered with curative intent. Lefebvre and colleagues6 later reported data from a European trial involving patients with cancers of the hypopharynx. In this randomized study, comparing induction chemotherapy followed by irradiation in subjects who had complete response with surgical resection and postoperative radiotherapy, overall survival rates in the two arms were considered equivalent, again demonstrating that larynx preservation could be achieved in some patients without jeopardizing their survival. Calais and associates7 presented data from GORTEC (Groupe d’Oncologie Radiotherapie Tête et Cou) 2000-01, which compared induction chemotherapy using cisplatin and 5-FU with 5-5-FU (TPF) or without docetaxel for organ preservation in hypopharynx and larynx cancer. In 220 patients, preliminary results show a superior overall response rate of 83% with TPF, versus 61% for cisplatin and 5-FU. The rate of larynx preservation was also better, offered in 80% of patients in the TPF arm, versus 58% in the cisplatin and 5-FU arm. Neutropenia was greater in the experimental arm (57% vs. 35%). On balance, TPF demonstrated a significantly superior tumor response rate, was tolerated with acceptable toxicity, and appeared to be associated with better larynx preservation.
In the VA study,5 there were observed trends in patterns of tumor relapse, with 20% of patients in the chemotherapy arm having locoregional recurrence, compared with 7% in the surgery arm. Distant disease recurrence was more likely in the surgical arm, affecting 17% of patients in that group, but only 11% in the chemotherapy/radiotherapy arm. Salvage laryngectomy was required more often in patients with glottic cancers than in those with supraglottic primary sites (43% vs. 31%, respectively), in patients with fixed vocal cords than in those with mobile vocal cords (45% vs. 29%), and in patients with gross invasion of thyroid cartilage than in patients without (41% vs. 35%). Notably, salvage laryngectomy was required in 56% of patients with T4 cancers but in only 29% of patients with smaller primary tumors (P = .001).
The VA study has prompted further investigations of chemotherapy and radiotherapy in the treatment of intermediate-stage laryngeal cancer using the sequential administration of induction chemotherapy, consisting of cisplatin and 5-FU, followed by radiotherapy as the control arm.8 In a study reported in 2003 by Forastiere and colleagues,8 called the Intergroup Radiation Therapy Oncology Group (RTOG) 91-11 trial, this modality was compared with concomitant cisplatin and radiotherapy and with radiotherapy administered as a single treatment modality. For all groups, totaling 547 patients, surgical salvage was reserved for patients with persistent or locally recurrent disease. Eligible patients had stage III or IV disease, but all of those with T1 and most of those with T4 disease were excluded. The rate of preservation of the larynx was greatest in patients receiving concomitant chemoradiotherapy, and the difference between it and the preservation rate in patients receiving radiotherapy as a single modality reached statistical significance. Survival was not affected by treatment assignment. Acute mucocutaneous toxic effects of treatment were greatest in the patients who received radiotherapy with concomitant cisplatin.
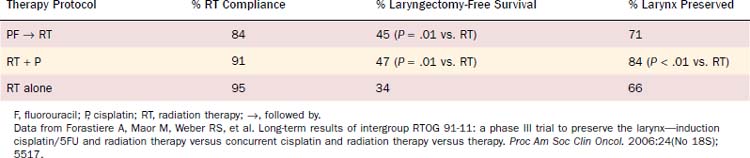
In 2006, Forastiere and colleagues9 provided a 5-year analysis of Intergroup RTOG 91-11 data (Table 78-1). Most notably, laryngeal preservation was greatest in those patients randomly assigned to concomitant radiotherapy with cisplatin (84% vs. 66% with radiotherapy alone; P = .0002). Data for patients living with preservation of the larynx was similar when contrasted with the cisplatin and 5-FU induction arm with concomitant chemoradiation. Moreover, overall survival was 59% in the induction arm compared with 55% in the concomitant chemoradiation arm, not a statistically significant difference. Disease-free survival was similar for the sequential and concomitant chemoradiation strategies.
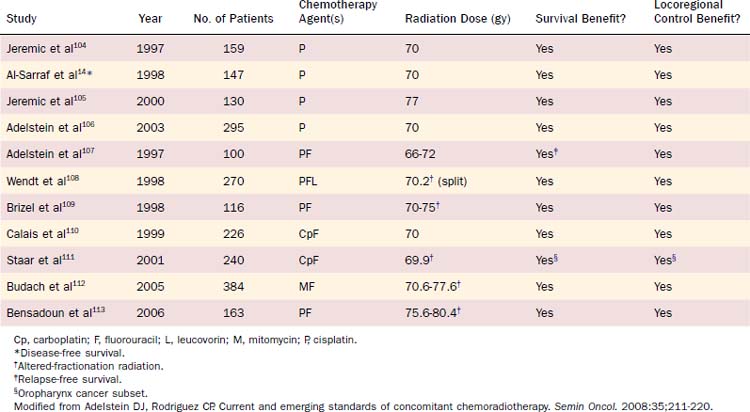
Within the past decade, concomitant chemoradiotherapy has become the standard-of-care treatment option for patients with locally advanced squamous cell carcinomas of the larynx, oropharynx, and hypopharynx (Table 78-2). The theoretical rationale of and mechanism for the interaction between cytotoxic drugs and irradiation that results in additive or synergistic enhancement have been reviewed in detail.10 This biologic phenomenon rests on several mechanisms, as follows: (1) inhibition of DNA repair, (2) redistribution of cells in sensitive phases of the cell cycle, and (3) promotion of oxygenation of anoxic tissues. The net effect is to improve cellular cytotoxicity.11 The benefits of adding chemotherapy delivered during irradiation are notably demonstrated in two meta-analyses.12,13 Concomitant administration of chemotherapy and irradiation was reported to achieve better local control and an 8% survival advantage at 5 years over radiotherapy administered as a single modality. Notably, however, this approach to improved tumor control appears to be associated with increased acute and chronic toxicities, inanition, esophageal stenosis with gastrostomy tube dependence, and recurrent infection related to aspiration.
The choice of the best chemotherapy regimen to be used in conjunction with irradiation has not been well addressed in randomized trials, but cisplatin 100 mg/m2 administered on days 1, 22, and 43 of a daily radiation treatment plan is widely accepted as a standard. The major drugs with efficacy for SCCHN and in vitro evidence of radiation enhancement capability have been tested as single agents since the 1960s. Most of the single agents used to treat patients with head and neck cancer have been combined with irradiation. Multiagent chemotherapy given concomitantly with radiotherapy has also been evaluated in several studies. Data from selected randomized trials of combined therapy are shown in Table 78-2. In general, to date, there is no convincing evidence to suggest that multiagent chemotherapy is superior to single-agent chemotherapy when given concomitantly with radiotherapy. Additionally, nearly all reported trials of concomitant chemotherapy (single-agent or multi-agent) and radiotherapy have noted enhanced acute radiation-induced toxicity, primarily mucosal, which may result in dose reductions and interruptions in the radiation fractionation schedule. This result violates a guiding principle of radiation therapeutics. Thus, in combining these two treatment modalities, especially when multi-agent chemotherapy regimens are used, it is essential that sensitizing drug-related toxicity not compromise an optimal radiation dose and volume plan. As a result, single-agent cisplatin therapy has evolved as a predictable and effective regimen and is commonly accepted as a standard in the United States in chemoradiation programs for head and neck cancers of any site. In part, this development has been due to favorable results observed in the Intergroup 0099 trial for nasopharyngeal carcinomas.14 In this study, patients were to receive either radiotherapy alone or cisplatin (100 mg/m2 on days 1, 22, and 43) during radiotherapy, followed by adjuvant chemotherapy with cisplatin and 5-FU (three cycles). An analysis of 147 randomly assigned patients revealed significant differences in 3-year survival time (78% vs. 47%) and progression-free survival time (69% vs. 24%) favoring the chemotherapy group.
An alternative approach to combined drug therapy and radiotherapy has been developed by Robbins and Homma,15 with rapid infusion of high-dose intra-arterial cisplatin followed by the systemic neutralizing agent sodium thiosulfate and administered with concomitant irradiation, a technique known as RADPLAT. Drug is infused selectively into the tumor bed after technically demanding placement of microcatheters with the intent of concentrating the cytotoxic compound within the tumor and minimizing systemic exposure, in order to gain a marked therapeutic effect with modest toxicity. In a 2008 report on a series of 240 patients with T3 or T4 squamous cell cancers of the oral cavity, oropharynx, and hypopharynx, in whom RADPLAT was used, local disease control ranged from 69% to 89% and was most favorable for oropharyngeal lesions.16 These control data approximate what may be achieved with concomitant intravenous chemotherapy. Severe mucositis was observed in 31% of patients. The precise risk of long-term complications, such as osteonecrosis and neurologic deficits, is under scrutiny. Continued research is anticipated.
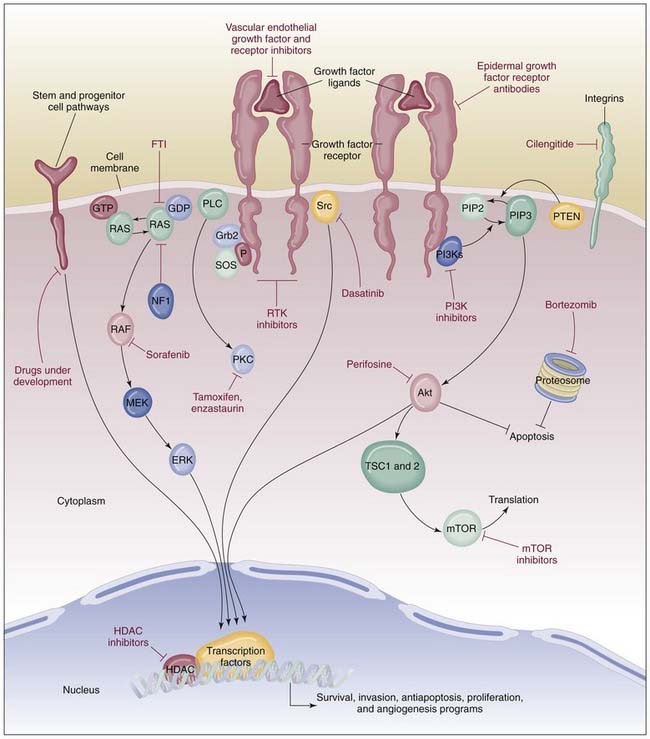
Biologic agents have been studied in combination with radiotherapy as well, with EGFR-targeted agents representing the class of drugs in the most advanced stages of clinical development. See Figure 78-1 for a depiction of signaling pathways that are considered potential targets for new drugs in development. In an important study, Bonner and associates2 conducted a prospectively randomized phase III trial in 424 treatment-naïve patients with stage III and IV SCC of the oropharynx, hypopharynx, and larynx. Patients were randomly assigned to radiotherapy as a single modality or radiotherapy with cetuximab administered in a weekly schedule at 250 mg/m2 after an initial loading dose of 400 mg/m2. The experimental regimen was generally well tolerated. Three per cent of patients receiving the targeted agent demonstrated severe hypersensitivity reactions, but notably, severe “in-field” mucocutaneous toxicity was not increased in the combined-modality treatment arm. Moreover, locoregional tumor control (median 24.4 months vs. 14.9 months for radiotherapy only) and overall survival (hazard ratio for death 0.74; P = .03) were higher in the combined-modality arm. In a retrospective subset analysis, the survival advantage was most favorable for patients with oropharyngeal primary cancers, although the study was not sufficiently powered for a survival analysis according to primary site. This is a landmark study, the first to demonstrate a survival advantage for targeted therapy in the primary treatment of SCCHN.
Concomitant Chemoradiation as Adjuvant Therapy
Adjuvant chemotherapy after definitive surgery is a treatment strategy used in many types of cancer with the goal of eradicating residual micrometastatic disease and thus ultimately improving long-term survival. In head and neck cancer, adjuvant chemotherapy as a single-modality treatment is still not widely accepted because previous trials have shown difficulty with completing the treatment plan and efficacy has not been demonstrated. Through the Head and Neck Intergroup organization, a large multi-institutional trial was conducted to test whether the addition of chemotherapy to surgery and radiotherapy prolonged survival time or altered the pattern of recurrence.17 Patients with stage III or IV squamous cell carcinoma of the oral cavity, oropharynx, or larynx and patients with stage II, III, or IV of the hypopharynx in whom resection margins were pathologically negative were eligible. Randomization was to either immediate postoperative radiotherapy or three cycles of cisplatin plus 5-FU chemotherapy followed by radiotherapy. An analysis of the 503 patients in the study showed no significant difference in disease-free survival time, overall survival time, or locoregional control between the two treatment groups. However, there was a significantly lower rate of distant metastases as a site of failure (P = .016) at any time for patients treated with adjuvant chemotherapy. Perhaps more important was the finding that a high-risk subset of patients (those with extracapsular extension, carcinoma in situ, or narrow surgical margins) appears to benefit from adjuvant chemotherapy, with increases in survival time and local control that approached statistical confidence in comparison with those receiving radiotherapy alone.
Two trials testing induction chemotherapy added maintenance chemotherapy to one treatment group and observed differences in outcome. The Head and Neck Contracts Program18 trial of one course of cisplatin and bleomycin induction chemotherapy before surgery and irradiation also planned for 6 months of maintenance chemotherapy in one of the three treatment arms. A significant decrease in the distant metastatic rate was observed for patients in that arm. Ervin and associates19 randomly assigned patients showing a response to cisplatin, bleomycin, and methotrexate induction chemotherapy to receive either three additional cycles or observation after definitive surgery and radiotherapy. The 3-year disease-free survival time for patients receiving maintenance chemotherapy was 88%, compared with 57% for the control subjects (P = .03). In a phase II pilot study, Johnson and colleagues20 treated 42 patients who had extracapsular spread of tumor in cervical lymph node metastases with 6 months of methotrexate and 5-FU after resection and radiotherapy. The 2-year disease-free survival rate was 66%, which was better than the control rate of 38% that was expected on the basis of historical experience.
More recently, prospective phase III studies conducted in the United States21 and Europe22 testing the value of concomitant cisplatin and radiotherapy in patients considered at high risk for recurrence after surgery because of tumor margin involvement of the resected specimen, nodal extracapsular spread, or both have demonstrated better local tumor control and overall survival.23 Although the entry criteria were slightly different in the two studies, patients were randomly assigned to either radiotherapy alone or combined treatment. The rate of acute mucocutaneous side effects was higher in the combined-modality arm, but the chronic toxicity was not reported to be elevated. There was an overall 42% reduced risk of locoregional treatment failure. Thus, for patients who have undergone surgery and have the selection factors previously indicated, postoperative drug therapy with concomitant radiotherapy is now considered the standard of care.
Induction Chemotherapy
Theoretically, treatment with chemotherapy before surgery or irradiation—known as induction chemotherapy—has several advantages. Initial treatment with chemotherapy is feasible. Patients are not debilitated because of surgery or radiotherapy. Clearly the drug activity is optimal in the previously untreated patient, probably because there had been no disruption of normal vascularity. Effective systemic therapy induces primary and nodal disease response in a high percentage of patients, and further, there may be a systemic effect with a reduction in risk of distant disease recurrence.24
With the introduction of cisplatin into clinical trials in the mid-1970s, Hong and coworkers25 combined cisplatin with a 5- to 7-day continuous infusion of bleomycin and observed an overall response rate of 75%, with a 20% complete response rate. Other investigators added vinblastine, vincristine, or methotrexate to the two-drug combination and had similar results. An alternative and probably more effective regimen tested in the 1980s at Wayne State University was cisplatin (100 mg/m2) followed by a 5-day infusion of 5-FU (1 g/m2 per day by continuous infusion). In phase II trials, this regimen produced an overall response rate as high as 93% and a 54% complete response rate when three cycles were administered.26 Although the toxicity from cisplatin is the same, 5-FU appeared to be better tolerated than bleomycin, without the associated allergic phenomena or lung toxicity.
Ensley and colleagues27 from Wayne State University have reported a high complete response rate with the use of five or six courses of cisplatin and 5-FU alternating with methotrexate, leucovorin, and 5-FU. In one study, the complete response rate was 65% in 31 patients completing the protocol, although toxicity was formidable and approximately one third of patients withdrew from the study early. Despite the potential for improvement in response rate, the applicability of alternating regimens has yet to be demonstrated. This group of investigators and others showed that a response to induction chemotherapy correlated with a response to subsequent radiotherapy.28–30 Thus, patients whose tumors are resistant to cisplatin-based induction chemotherapy had a high likelihood of not having a response to radiotherapy.
Most authorities agree that randomized trials incorporating appropriate stratification factors and prognostic variables, and with adequate follow-up periods, are necessary to draw conclusions about the effects of induction chemotherapy on disease-free survival and overall survival.24,31 The results of numerous randomized controlled trials of induction chemotherapy before surgery, radiotherapy, or both, have been published.
The previously mentioned Head and Neck Contracts program,18 a Southwest Oncology Group trial,32 and the VA Laryngeal Cancer Study Group trial5 were large multi-institutional randomized studies. The patients in all studies had advanced resectable head and neck cancer, and the treatment arms were well balanced as to TN stage and primary site. The Head and Neck Contracts program randomly assigned patients to receive one of the following three treatments: (1) surgery followed by irradiation, (2) induction chemotherapy with one cycle of cisplatin plus bleomycin followed by surgery and irradiation, and (3) induction chemotherapy, surgery, irradiation, and maintenance chemotherapy with cisplatin for 6 months. The 5-year survival rates for the three regimens were 35%, 37%, and 45%, respectively; the differences were not statistically significant. However, the time to development of distant metastases and the frequency of distant metastases as a site of first recurrence were significantly lower in patients in the maintenance chemotherapy arm than in the other two groups.18 Subgroup analysis also found a significant difference in disease-free survival time for patients receiving maintenance chemotherapy for oral cavity primary tumors and for N1 or N2 disease.33 In retrospect, it is not surprising that this trial did not show improvement in overall survival time because only one cycle of cisplatin and bleomycin was administered before surgery, resulting in a low response rate of 37%.
In the Southwest Oncology Group trial, patients were randomly assigned to receive either three cycles of cisplatin, bleomycin, methotrexate, and vincristine before surgery and radiotherapy or standard treatment with surgery and radiotherapy. The median survival time was 30 months for patients in the standard treatment arm and 18 months for those in the induction chemotherapy arm. The distant metastatic rate was 49% with standard treatment and 28% with induction chemotherapy. Although differences in survival time and pattern of recurrence are striking, they did not reach statistical significance. This trial fell short of its accrual goals and had a high rate of noncompliance, with only 56% of patients assigned to induction chemotherapy completing the treatment per protocol.32
Domenge and associates34 have reported a phase III trial in which 318 patients with locally advanced oropharyngeal squamous cancers were randomly assigned to receive either induction chemotherapy with cisplatin and 5-FU followed by locoregional treatment or locoregional therapy alone. Overall survival was better in the chemotherapy group (median, 5.1 vs. 3.3 years; P = .03).
Encouraging data emerge from induction chemotherapy trials and in the area of organ preservation concurrent chemoradiotherapy. The VA Laryngeal Cancer Study Group5 completed a randomized trial in patients with resectable stage III and IV squamous cell cancer of the larynx. Patients were randomly assigned to receive either standard therapy with total laryngectomy and postoperative radiotherapy or a maximum of three cycles of cisplatin and 5-FU chemotherapy followed by radiotherapy. Surgery was reserved for salvage in patients with persistent or recurrent disease. If patients did not have at least a partial response at the primary site after two cycles of chemotherapy, they underwent immediate surgery. The complete and partial response rate after two cycles of chemotherapy was 85%, and after three cycles it was 98%. The pathologically confirmed complete response rate at the primary site was 64%. At a median follow-up period of 33 months, there was no significant difference in survival time. However, the patterns of relapse differed: recurrence at the primary site was 2% for the surgery group compared with 12% for the chemotherapy group (P = .0005); regional node recurrence rates were similar (P = .305); rates of distant metastases were 17% and 11%, respectively (P = .016); and the rates of second primary malignancies were 6% and 2%, respectively (P
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree