Chapter 72 Central Serous Chorioretinopathy
Introduction
Central serous chorioretinopathy (CSC) was first described by Albrecht von Graefe as central recurrent retinitis in 1866.1 It is a chorioretinal disorder, incompletely understood, with systemic associations, a multifactorial etiology, as well as a complex pathogenesis. It typically affects young to middle-aged men and is characterized by serous detachment of the neurosensory retina, which is usually located at the posterior pole. It is usually idiopathic but might also be secondary to high levels of endogenous or exogenous corticosteroids. Advances in imaging, particularly in indocyanine green angiography (ICGA) and optical coherence tomography (OCT), have led to a greater understanding of the pathophysiology of CSC. Most cases of CSC are self-limiting, with spontaneous resolution and good visual prognosis. However, some patients may suffer from persistent or recurrent serous macular detachment with subsequent progressive visual loss. A greater understanding of CSC has led many to believe that CSC is not a completely benign condition. Treatments for CSC, in particular the photodynamic therapy (PDT) using lower doses and reduced fluence, and the antivascular endothelial growth factor (anti-VEGF) therapy, are evolving. Although treatment results appear to be promising, more randomized large-scale controlled studies are needed before their treatment roles can be fully delineated.
Pathogenesis, predisposition, and risk factors
The pathophysiology of CSC has yet to be fully elucidated. It has, however, been thought to involve multiple etiologies and mechanisms that ultimately lead to widespread choroidal circulation abnormalities.2 Hyperdynamic choroidal circulation and choroidal vascular hyperpermeability are the main features that are shared among patients with CSC.3 The resultant increase in hydrostatic pressure in the choroid causes breakdown of the retinal pigment epithelial (RPE) barrier with subsequent leakage of fluid from the choroid through defects in RPE cell tight junctions into the subretinal space.4,5 Studies of ICGA of patients with CSC also revealed areas of hypofluorescence, indicating choriocapillary nonperfusion, which might also be one of the mechanisms that lead to choroidal venous dilation and congestion.
Horniker first suggested that psychiatric disturbances were linked to CSC in 1927.6 He described a mechanism in which mental disturbances would lead to retinal angiospasm and subsequent macular exudation. In the subsequent 60 years, however, there was a lack of study in the literature to confirm this link between psychiatric problems and CSC. It was not until 1987 that Yannuzzi’s cross-sectional study demonstrated the association of CSC with Type A behavioral pattern.7 The mechanism was thought to be related to elevation of catecholamine levels, which might trigger vasoconstriction of choroidal vessels by stimulating the sympathetic nervous system and adrenergic receptors.
Exogenous as well as elevated endogenous corticosteroids are well-known predisposing factors for CSC.8 Patients taking systemic steroid for diseases such as autoimmune conditions and after organ transplantation are at risk.4,9,10 CSC has been reported to occur in up to 6% of patients receiving corticosteroids after renal transplant.11 Other routes of exogenous corticosteroids such as intra-articular, intranasal, and topical have also been reported to put patients at risk of CSC.12–14 Diseases associated with an increased endogenous cortisol production such as Cushing’s disease and pregnancy also increase the risk of CSC.15–19 In some cases, CSC can even be the presenting symptom of Cushing syndrome.20 There are a number of proposed mechanisms as to how corticosteroids are related to CSC. Corticosteroids could induce choroidal vasoconstriction by reducing nitric oxide production. Direct increase in the permeability of the blood vessels might also occur, together with RPE cell tight junction damage.21–23 Corticosteroids could reverse the polarity of RPE cells, which causes them to pump ions into the subretinal space. Fluid will then enter the subretinal space by osmosis.24,25
Abnormal coagulation and platelet aggregation have also been proposed to be involved in the pathogenesis of CSC.26 Aqueous sample cytokine analysis showed that eyes with CSC have lower platelet-derived growth factor (PDGF) levels in the aqueous, suggesting that PDGF might play a role in the pathogenesis of CSC.27 The role of VEGF in CSC has been studied and intravitreal anti-VEGF injections have been tried. However, aqueous and plasma samples from patients with CSC showed no elevation in VEGF level when compared with normal controls.27,28
Helicobacter pylori infection has been reported to be associated with CSC and its treatment to hasten the rate of subretinal fluid resolution.29–31 Hypertension, smoking, antibiotic use, antihistamine use, alcohol consumption and allergic respiratory diseases have also been implicated in increasing the risk of CSC.16,32 Obstructive sleep apnea has been postulated to be associated with CSC and its treatment has been reported to lead to the resolution of CSC.33 A case-controlled study showed that patients with CSC have a less chance of glaucoma when compared with controls.34 This might be related to increased choroidal blood supply to the optic nerve but the exact mechanism remains unclear. While there have been isolated case reports of patients developing CSC after rhinoplasty35 and laser in situ keratomileusis,36 their relationship is unclear.
Clinical features
Demographics
CSC predominately affects males, with a male to female ratio of approximately 6 : 1.37 The age of onset is usually between 30 and 50 years but patients with chronic CSC might continue to suffer from the disease even though they are advanced in age. However, if a patient over 50 years of age presents a clinical appearance of CSC, one should be suspicious of differential diagnoses such as age-related macular degeneration (AMD) and polypoidal choroidal vasculopathy (PCV).38 CSC appears to have a low incidence in blacks when compared to whites and Asians, but it might behave more aggressively in blacks.39–41 At presentation, involvement is usually unilateral. However bilateral involvement is common in chronic cases and cases related to excessive endogenous or exogenous corticosteroids.
Signs
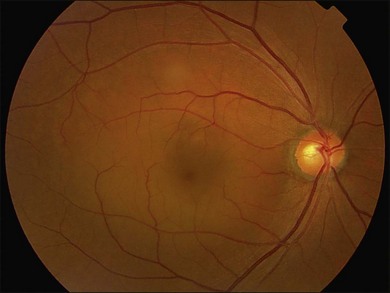
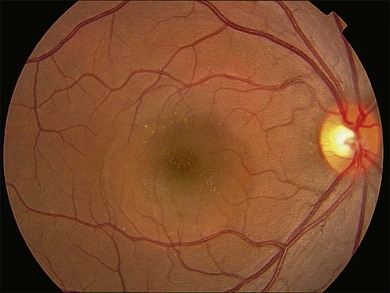
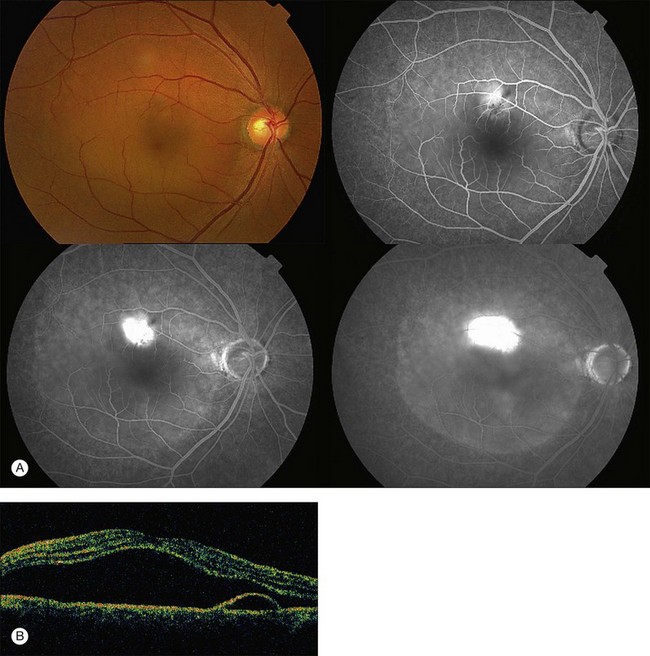
Fundus examination typically shows a well-demarcated oval-shaped area of neurosensory retinal detachment in the posterior pole (Fig. 72.1). Serous pigment epithelium detachment (PED) can also occur together or independently. The diagnosis is usually obvious from examination with indirect ophthalmoscopy. However, in cases with minimal subretinal fluid or small PED, slit-lamp biomicroscopy with fundus contact lens might be useful. In these cases, the loss of the normal foveal reflex might provide a good hint. Yellow dots are frequently observed on the posterior surface of the detached retina and are postulated to be associated with phagocytosis of shed photoreceptor outer segments (Fig. 72.2).42 Yellowish discoloration of the fovea is often seen and is caused by increased visibility of retinal xanthophyll.4,17,39,43 The subretinal fluid is usually transparent and colorless, but occasionally it can also appear cloudy.39,44 Fibrin can form in the subretinal and sub-RPE spaces and cause the subretinal fluid to become opaque. The fibrin usually dissolves spontaneously but rarely it could cause fibrosis and lead to permanent drop in vision. In chronic recurrent cases, RPE change and atrophy might develop. Patients with CSC can also present bullous neurosensory detachments,10,45–48 which are usually located inferiorly as the subretinal fluid drains down from the macula by gravity. In chronic cases, an atrophic RPE tract connecting the macula to the inferior detachment might be seen. Other complications of chronic CSC include secondary choroidal neovascularization (CNV) formation, cystoid macular edema, subretinal lipid deposition, and choriocapillaris atrophy.49
Investigations
Fluorescein angiography
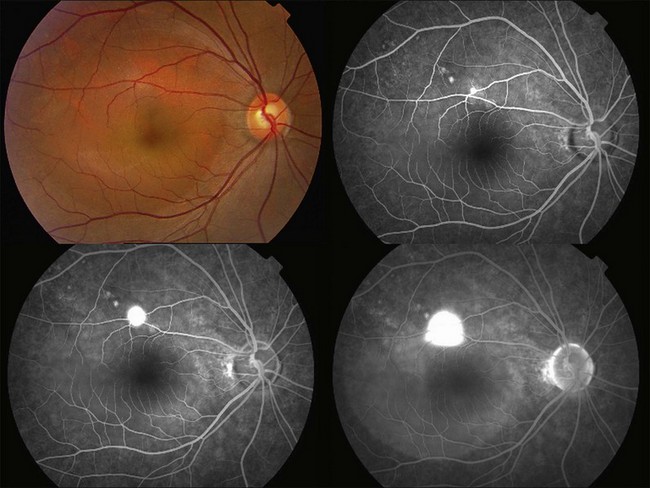
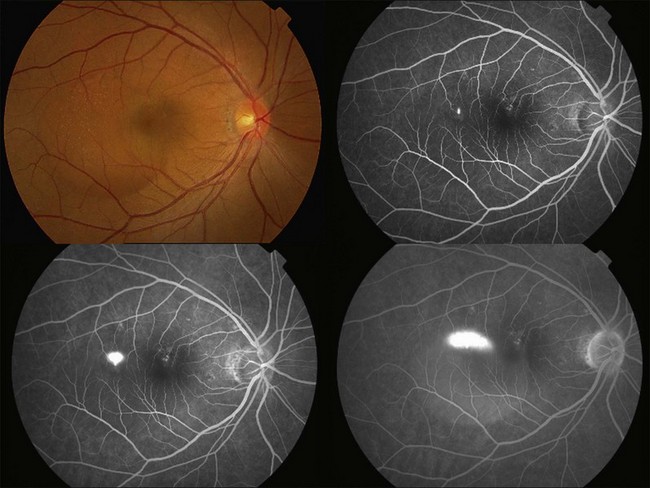
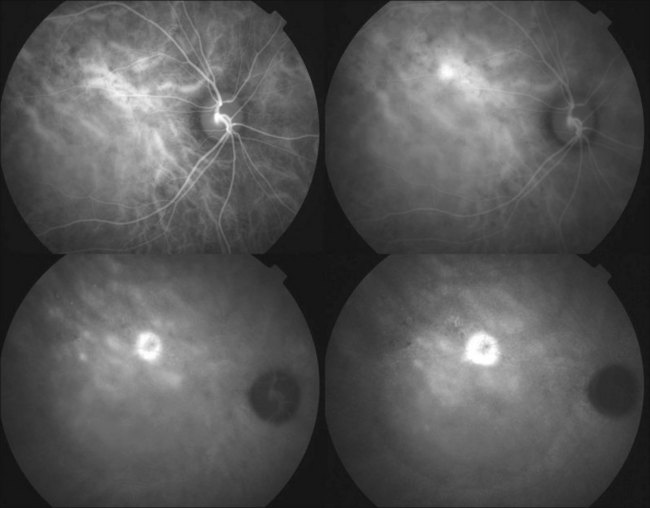
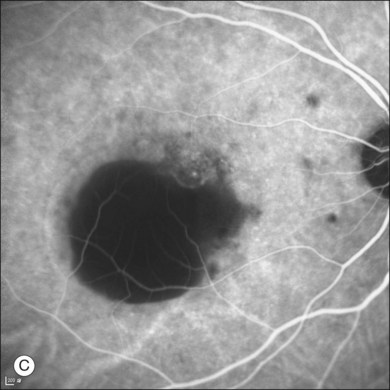
Fluorescein angiography (FA) in acute CSC typically shows one of two different types of leakage patterns: ink blot or smoke stack. In the former, the leakage starts as a pin point in the early phase and then concentrically diffuses out in the late phase and appears like an ink blot (Fig. 72.3). In the smoke stack appearance, the leakage again starts as a pin point in the early phase, but it gradually tracks upward and then expands to form a mushroom cloud or umbrella-like appearance (Fig. 72.4). Smoke stack appearance is less common and only appears in about 10–15% of patients with acute CSC.50 It is caused by an increased protein concentration in the subretinal fluid. In cases in which there is PED, the FA appearance would be the pooling of dye in the sub-RPE space (Fig. 72.5). Chronic CSC might show an RPE window defect due to RPE atrophy. Multifocal CSC would show multiple sites of leakage. FA is also useful in differentiating CSC from other diagnoses such as choroidal neovascularization (CNV) and Vogt–Koyanagi–Harada disease.
Indocyanine green angiography
ICGA is one of the most important investigations in CSC because it demonstrates the choroidal vascular abnormalities and can act as a guide to treatments such as photodynamic therapy. In CSC, delay in choroidal filling is usually present. Typical features include abnormally dilated choroidal vasculature in the early phase and choroidal hyperpermeability in the late phase (Fig. 72.6).51–54 The area of choroidal vascular abnormality is usually much more widespread than the leakage point on FA and is commonly present in the fellow eye as well. Punctate hyperfluorescent spots are often seen in the mid-phase.55 In the later phases of ICGA, the dye usually leaks into the deeper layers of the choroid to produce hyperfluorescent patches.39 Hypofluorescent areas can also be seen on ICGA. They represent areas of choriocapillary nonperfusion and this might be one of the mechanisms that lead to choroidal venous dilation and congestion.
Optical coherence tomography
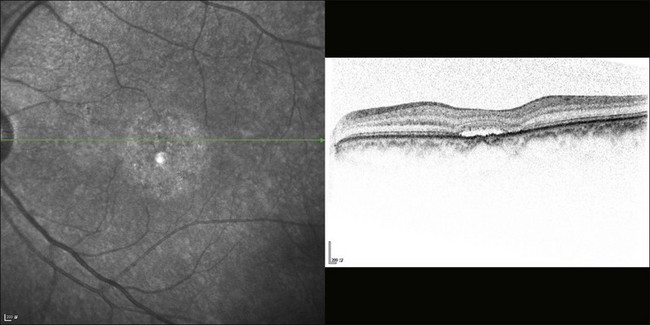
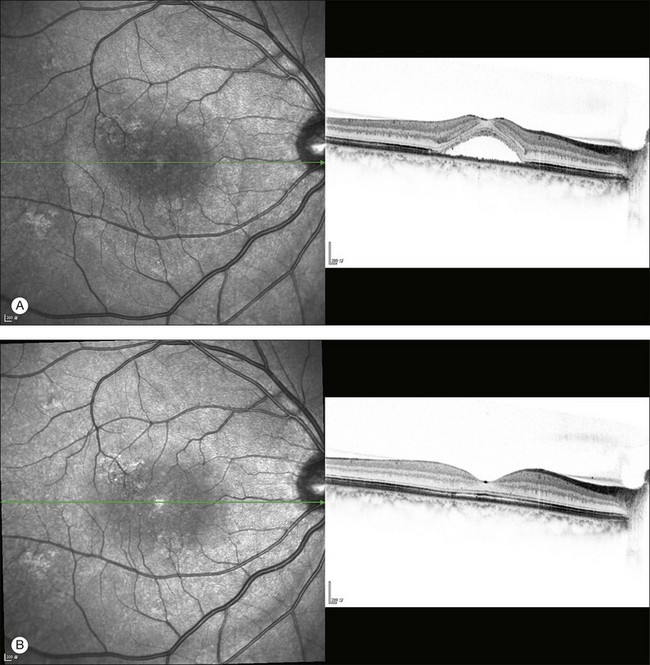
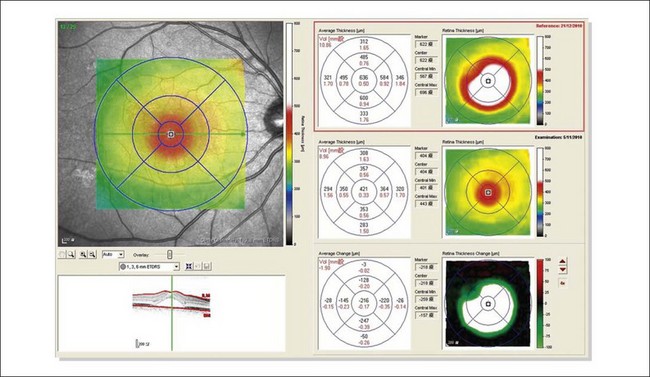
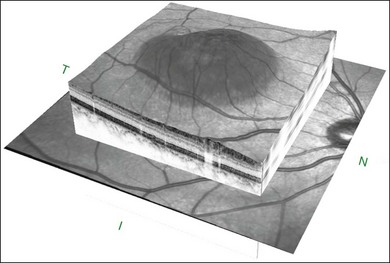
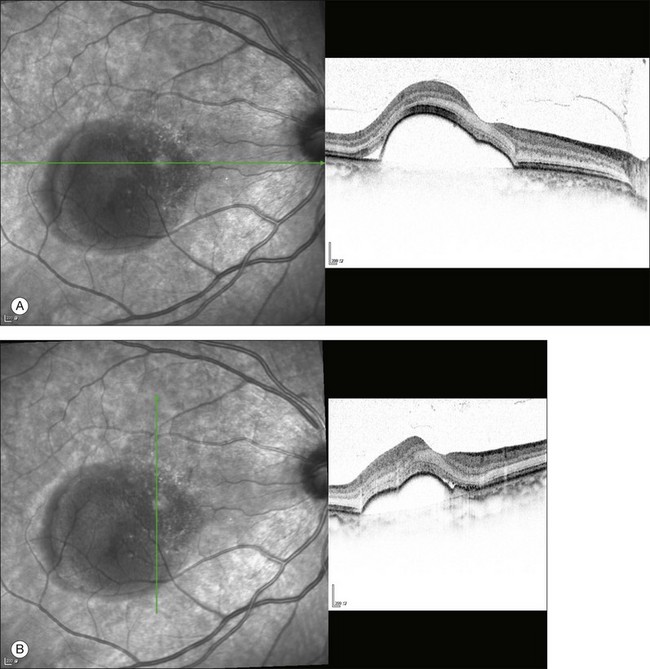
The availability of OCT has vastly enhanced the anatomical assessment and understanding of CSC by providing cross-sectional imaging of the macula. OCT demonstrates beautifully the presence of subretinal fluid or PED and helps in differentiation between the two.56,57 Subretinal yellow dots observed clinically, which typically show high reflectivity, can also be seen.42 Outer nuclear layer OCT is especially useful in detecting shallow subretinal fluid and small PED, which might be difficult to identify clinically (Fig. 72.7). OCT might be able to demonstrate lesions in the fellow asymptomatic eye, such as RPE bumps and small PED.58 Serial scans can be used to assess disease progression and treatment response (Fig. 72.8). Quantitative measurements and thickness maps can be generated and are useful for documentation as well as research purposes (Fig. 72.9). The software can also render three-dimensional images to allow better structural visualization and understanding (Fig. 72.10). Choroidal vascular hyperpermeability on ICGA is associated with an increase in subfoveal choroidal thickness on OCT.59,60 With the introduction of enhanced depth imaging, many researchers consider that increased choroidal thickening is a hallmark of CSC. OCT can also offer valuable prognostic information. Cystoid degeneration and disruption of the outer photoreceptor layer and the inner/outer-segment junction have been reported to be associated with poor visual outcomes.61 OCT can also assist in differentiating CSC from other diagnosis such as CNV and PCV by detecting lesions in the subretinal space (Fig. 72.11). Another reported distinguishing feature between PCV and CSC is that eyes with PCV might show thinning of the photoreceptor outer segments on OCT.62
Fundus autofluorescence
Fundus autofluorescence (FAF) is an adjunctive tool for the assessment of CSC. During the acute phase of the disease, FAF typically shows hypofluorescence at the leakage point and over the area of neurosensory detachment due to blockage by subretinal fluid.63 The subretinal yellow dots observed clinically might demonstrate hyperfluorescence.42 In chronic-recurrent CSC, hyperfluorescence is common in areas of residual neurosensory detachment. Therefore, FAF might give additional information on whether the disease is acute or chronic.64 Moreover, the pattern of FAF has been shown to correlate with visual acuity.65 After the resolution of subretinal fluid, areas of hyperfluorescence might become visible due to release of fluorophore materials into the subretinal space. Photopigment density has been studied with autofluorescence densitometry. It was found to be decreased in eyes with CSC and showed a delayed recovery after the resolution of subretinal fluid.66
Multifocal electroretinography
Multifocal electroretinography (mfERG) is useful in evaluating macular function in CSC. First- and second-order kernel mfERG response amplitudes have been shown to be reduced in patients with CSC.67,68 Reductions in response amplitudes appear to be localized in the center for the first-order kernel mfERG but predominately affect the more peripheral retina for the second-order kernel mfERG. These suggest that while outer retinal dysfunction is localized in the center, inner retinal dysfunction might be more widespread. Unlike OCT, mfERG response amplitudes were found to correlate with visual acuity.67,69 Therefore mfERG and OCT can complement each other in the functional and anatomical assessments of CSC respectively.
Microperimetry
Microperimetry is useful in the assessment of macular sensitivity in patients with CSC. Macular sensitivity is reduced in both the central and paracentral areas in the active phase of the disease when there is subretinal fluid.50 It has been shown to improve after the resolution of subretinal fluid with or without treatment.70–72 However, more often than not there are residual focal areas with reduced sensitivity that correspond to RPE irregularities or defects of the inner/outer-segment junction on OCT.73,74 Moreover, macular sensitivity was demonstrated to correlate with central macular thickness on OCT, which suggested the existence of structural and functional correlation.75
Natural history
The disease is usually self-limiting and 90% of the cases will show spontaneous recovery within a few months without significant visual loss.37,76,77 However, some patients may develop chronic or recurrent diseases that lead to areas of RPE atrophy or hypertrophy with visual loss. Poor visual acuity on presentation and a prolonged duration of serous macular detachment appear to be associated with poor visual outcomes.78 Up to 50% of patients might develop recurrence.79–82 Recurrence can develop at any time and CSC recurs in about 50% of the patients within the first year. A history of psychiatric illness is associated with a higher rate of recurrence.82 A small proportion of patients develop severe irreversible visual loss due to, for example, RPE atrophy, CNV development (in up to 6% of patients), and transformation into polypoidal choroidal vasculopathy (PCV) with exudation and hemorrhage. Adaptive optics scanning laser ophthalmoscopy was able to demonstrate reduced cone densities in eyes with resolved CSC.83 Even patients whose visual acuity has recovered to baseline might be left with residual symptoms such as metamorphopsia, scotoma, and reduced contrast sensitivity. Therefore, CSC should not be considered a benign disease.
Differential diagnosis
The diagnosis of CSC is usually clear and straightforward from clinical examination and is confirmed by FA, ICGA, and OCT. However, several diseases can mimic CSC and it is important to keep in mind the following differential diagnoses (Table 72.1).
Table 72.1 Important differential diagnoses of central serous chorioretinopathy and their differentiating features
| Differential diagnosis | Differentiating features |
|---|---|
| Optic disc pit | Presence of optic disc pit, absence of leakage on FA |
| AMD | Older age group, CNV on FA |
| PCV | ICGA shows polyps and branching vascular network |
| Inflammatory and infectious diseases | Systemic features and bilateral involvement in VKH; ultrasonic T-sign in posterior scleritis |
| Autoimmune and vascular disorders | Systemic features are usually evident |
| Intraocular tumors | Ultrasound is useful in the detection and differentiation between different types of tumors |
FA, fluorescein angiography; AMD, age-related macular degeneration; CNV, choroidal neovascularization; PCV, polypoidal choroidal vasculopathy; ICGA, indocyanine green angiography; VKH, Vogt–Koyanagi–Harada disease..
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree