Chapter 29 Cellular Effects of Detachment and Reattachment on the Neural Retina and the Retinal Pigment Epithelium
Introduction
In retinal detachment the separation of the neural retinal from the retinal pigment epithelium (RPE) initiates a complex series of cellular and molecular changes.1 Left untreated, retinal detachment results in permanent visual loss; however, early intervention may be associated with good visual outcomes, suggesting that some of these molecular changes may be arrested or reversed.2,3
Use and limitations of animal models in the study of retinal detachment
Human studies of cellular changes following acute retinal detachment are limited to isolated case reports as surgical management does not routinely involve removal of retinal tissue. More recently data have become available from patients undergoing macular translocation surgery in which the retina is detached as part of the procedure, allowing sampling of the retina as early as 1 hour following detachment.4 In patients with advanced stages of retinal detachment and PVR, surgical management may involve excising areas of scarred retinal tissue allowing histopathological analysis. However, the data from human studies is still limited by small numbers, the challenges of sampling and analyzing small retinal specimens, and an inability to study cellular recovery following reattachment.4–7
Ideally the characteristics of an experimental detachment should closely mimic those found in humans while allowing for precise control over the extent of separation between the two layers (detachment height), the location of the detachment, its surface area, and the onset of detachment (or reattachment). A number of methods have been used to simulate human retinal detachment in animal models. These range from creating large retinal tears to subretinal injections of fluid or viscous substances. Experiments where retinal detachment induction is standardized with a micropipette provide a very controlled environment for analysis; however they differ from the clinical pattern of events in which acute retinal tears of variable size are induced by vitreoretinal traction at the time of posterior vitreous detachment. It is possible that retinal tearing may act as a more potent stimulus for cellular disorganization, loss, and remodeling, leading more rapidly to the advanced pathology usually seen following longer periods of retinal detachment in animal models. Although experiments involving animal models may differ in methodology, species used, and outcome measures, they have yielded similar results to give a relatively detailed profile of the changes that occur after detachment. Retinal tissue removed from human postmortem specimens and from patients undergoing retinal detachment surgery has demonstrated changes similar to those seen in animal models.6–8
Cellular changes in response to retinal detachment
Acute retinal detachment
A rapid response to retinal detachment has been shown to occur within 15 minutes, including phosphorylation of fibroblast growth factor receptor (FGFR-1) and increased expression by RPE and Müller cells of extracellular signal-regulated kinase and activator protein transcription factor.9 This initiates a cascade of events that leads to a number of molecular and cellular changes within the retina and RPE.
RPE–photoreceptor interface
The earliest structural effects of retinal detachment are seen at the interface of photoreceptor outer segments and the RPE.10 The mature RPE is a polarized monolayer of neuroepithelial cells that rests on Bruch’s membrane, between the choriocapillaris and the neural retina.11 The relationship of the apical surface of the RPE to differentiated photoreceptors is anatomically complex. There are no actual cellular junctions between the two layers in the mature eye, but the two are adherent, with the degree of adhesion varying among species.12 With the onset of retinal detachment changes to this interface include alterations in the RPE apical surface, proliferation of RPE cells, migration of cells into the subretinal space, degeneration of photoreceptor outer segments, and changes in photoreceptor outer-segment renewal.1
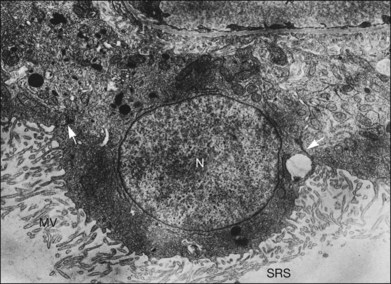
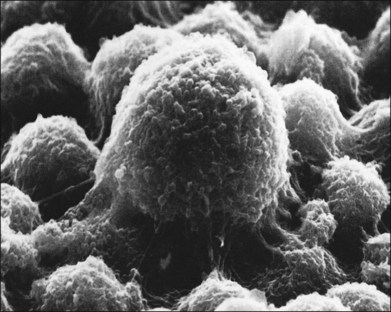
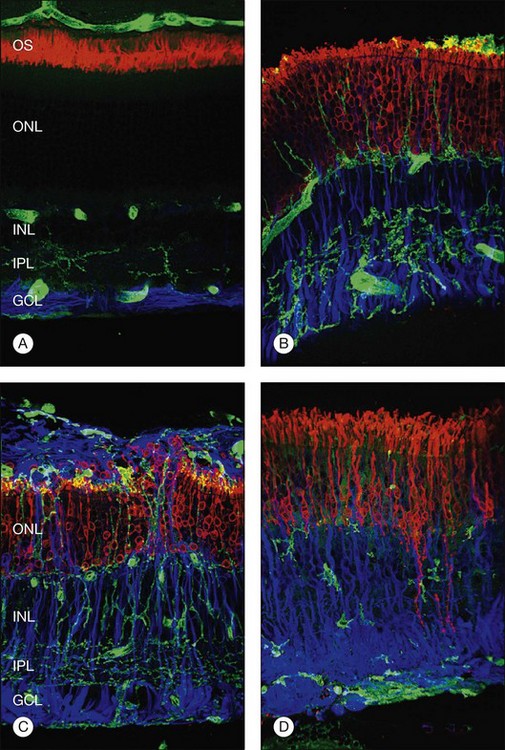
Within a few hours of retinal detachment, the long and elaborate sheet-like and villous processes that normally ensheath the outer segments are lost and replaced by a “fringe” of short microvilli (Fig. 29.1).13 At the same time, the overall surface morphology of the RPE cells changes into a rounded contour, as cytoplasm protrudes past the normal limits of the apical surface into the subretinal space, and the nucleus becomes displaced to a more apical position10,14 (Fig. 29.2).
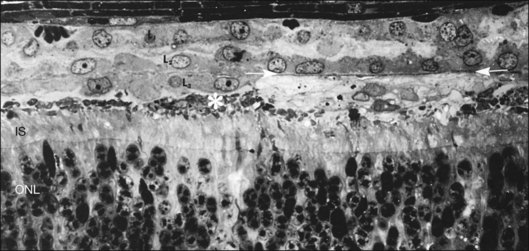
In the feline model, experiments using 3H-thymidine have shown that within 72 hours of retinal detachment the RPE has begun to proliferate and may be observed as areas of hyperplasia within the RPE monolayer.10 This proliferative response transforms the RPE’s uniform monolayer into a heterogeneous morphology in which strands of cells extend from the original monolayer into the subretinal space or result in the formation of multiple layers of cells whose polarity does not necessarily match that of the original monolayer (Fig. 29.3). This effect is limited to the region of detachment; in attached regions the RPE remains mitotically quiet, suggesting that attachment of the RPE to the neural retina acts to keep the RPE mitotically inactive and its apical surface highly differentiated.15–17 The proliferative response of the RPE cells also appears to be self-limiting with only low levels of proliferation observed after long detachment intervals (e.g., 12–14 months) in owl, monkey, and cat retinas10,16 (Box 29.1 and Fig. 29.4).
Box 29.1
Clinical correlates
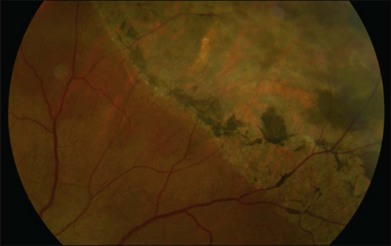
Retinal pigment epithelial (RPE) proliferation can be seen as subretinal pigment deposition in chronic retinal detachments. It is likely that the demarcation lines noted in human retinal detachments represent zones of proliferated RPE occurring at transitions between detached and attached regions of the eye (Fig. 29.4).
The subretinal space is usually free of cells; however within 24 hours of retinal detachment a number of cell types (polymorphonuclear neutrophils, monocytes, and macrophages) migrate into this space from the choroidal and retinal capillaries.10,18 Free RPE cells are also seen in the subretinal space within 72 hours of retinal detachment and frequently contain outer-segment fragments, indicating that they may play a role in phagocytosis of cellular debris.10,18
Photoreceptors
Within 12 hours of experimental retinal detachment, photoreceptor outer segments show evidence of structural damage. Initially, the distal end of the outer segment becomes vacuolated or distorted and by 24–72 hours all rod and cone outer segments are significantly shorter and distorted with disoriented discs.19 The degeneration of outer segments may proceed until those in the zone of detachment appear only as empty sacs of membrane attached to the connecting cilium.10
Outer-segment debris is shed into the subretinal space where it is phagocytosed by RPE cells and macrophages which have migrated into the area.10,18
Although retinal detachment interrupts the process of disc production and shedding, outer-segment specific proteins continue to be produced but localize to abnormal cellular locations. Opsin, normally concentrated in the outer segment, begins to accumulate in the plasma membrane vitread to the outer segment within a day following experimental retinal detachment (Fig. 29.5).19 Peripherin/rds, another outer-segment protein specific to the disc rims, is also redistributed and begins to appear in cytoplasmic vesicles.20 Cone outer-segment proteins appear to be more susceptible to damage, with redistributed cone opsins persisting for just 1 week following retinal detachment, after which their expression is downregulated.21
During the first day of a detachment the inner segments appear essentially normal, but between the first and third days they begin to show signs of degeneration: most commonly swelling, disruption, and loss of mitochondria (and loss of anticytochrome oxidase labeling) in the ellipsoid region,3,22 an overall disruption of the organized rough endoplasmic reticulum and Golgi apparatus in the myoid region, and, within a few days, an overall size reduction of the inner segment. It is interesting to note that the connecting cilium, which is essential for production of the outer segment, is retained even in severely affected inner segments in long-term detachments. This is crucial as its loss would prevent regeneration of outer segments following reattachment. Similarly, the loss of mitochondria also has the potential to affect the photoreceptors’ ability to regenerate significantly, because the metabolic rate in these cells is among the highest of any in the body.
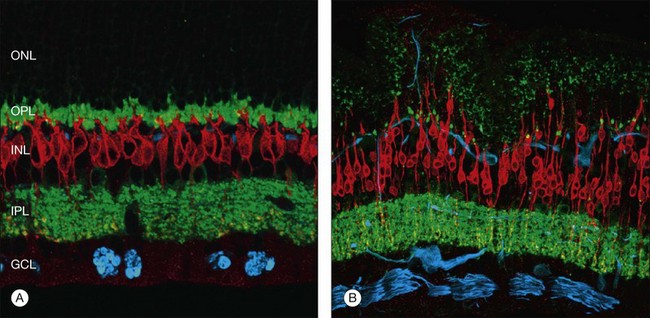
The outer nuclear layer contains the cell bodies of the photoreceptor cells. These cell bodies extend a process toward the outer plexiform layer, where they form synapses with second-order neurons. Rods and cones have characteristic synaptic terminals called spherules and pedicles respectively.23 The outer plexiform layer also contains the processes of second-order neurons, the cell bodies of which lie in the inner nuclear layer. These processes synapse with each other and with the photoreceptors. The photoreceptor cell bodies and synaptic terminals show a rapid response to detachment with extensive vacuolization, degeneration of mitochondria, and disorganization of the microtubules and actin filaments. Cell death via the apoptotic pathway peaks at day 3 following retinal detachment but continues at low levels for as long as the retina is detached, a process that appears to be mediated via caspases 3, 7, and 9.24,25 Recent studies have shown that, when caspase pathways are blocked, receptor interacting protein (RIP) kinases promote necrosis and overcome apoptosis inhibition. Therefore, targeting of both caspase and RIP kinase pathways is required for effective photoreceptor protection26 (Box 29.2).
Box 29.2
Clinical correlates
More recently, photoreceptor apoptosis has also been demonstrated in human retinal specimens, with a peak at day 2 following retinal detachment.27
Following cell death some photoreceptors are extruded into the subretinal space where they are phagocytosed by macrophages while others appear to undergo degeneration and phagocytosis within the outer nuclear layer.28
Not all photoreceptors degenerate at the same rate; areas of extensive degeneration coexist with areas of relatively intact photoreceptors.19 It does appear that rod cell bodies appear to degenerate quicker than cones following retinal detachment.21 In a region in which nearly all of the rod cell bodies show signs of degeneration and even cell death, neighboring cone cell bodies may look relatively intact. Consistent with this observation, the rod spherules appear to be particularly susceptible to the effects of detachment. These synaptic terminals are normally filled with synaptic vesicles and contain one or two large presynaptic ribbons. When the retina has been detached for 3 days, many of these terminals appear depleted of vesicles, except for a few that remain as a halo around a greatly truncated ribbon.29 Many terminals appear as if they have “retracted” into the cell body, and some synaptic structures generally associated with the outer plexiform layer now occur within the outer nuclear layer (Fig. 29.6).29,30 As with the cone and rod photoreceptor cell bodies, the cone synaptic terminals seem to survive the early effects of detachment better than the rod terminals do. Although their shape can change fairly dramatically, they do not appear to retract and by electron microscopy they remain filled with synaptic vesicles.30,31
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree