The authors present two case studies on pediatric nasal obstruction that highlight the consequences of a delayed diagnosis and the complexities of managing obstructed lesions in children.
A blocked or “stuffy” nose represents one of the most common patient complaints. The list of possible diagnoses is long. The problem can be self-limited or constant, a stand-alone concern or part of an array of symptoms. Children begin life as obligate nasal breathers and nasal obstruction in neonates can present as a dramatic airway insult. As the child ages, nasal blockage is a symptom commonly attributed to simple rhinitis or adenoid hypertrophy with little consideration given to the possibility of a significant underlying problem. This article presents two cases that highlight the consequences of a delayed diagnosis and the complexities of managing obstructed lesions in children.
Case report 1: 21-month-old female with bacterial meningitis
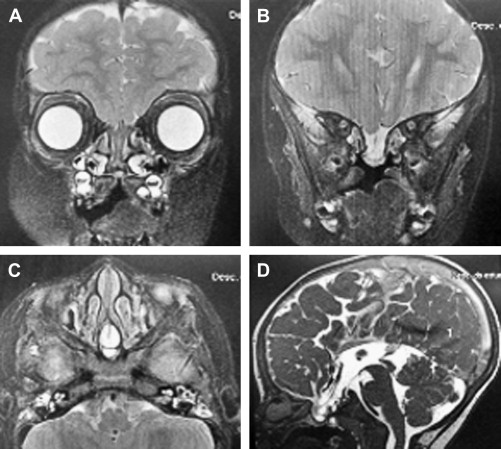
A 21-month-old female patient presented to a community hospital with a bacterial meningitis. She had mild hypertelorism and a stuffy nose attributed to a cleft soft palate. After appropriate medical intervention, the patient was discharged home without further assessment of her nasal stuffiness. She was then readmitted within a few weeks with a recurrence of her meningitis. After resolution of the second episode, a CT scan was performed that demonstrated a skull-base defect in the region of the planum sphenoidale. An MRI was performed and showed an encephalocele originating from the third ventricle protruding through the skull base into the posterior nasal space abutting the back of the septum ( Fig. 1 ). The patient was then transferred to Edmundo Vasconcelos Hospital in Brazil for definitive management.
Preoperative Considerations
The presentation of a child with a midline defect, such as a cleft palate and hypertelorism, should alert the clinician to the possibility of other midline defects and prompt the investigation of “trivial” complaints, such as a stuffy or blocked nose. Aberrant embryogenesis leads to three main types of anomalies: nasal dermoid, anterior encephalocele, and a nasal glioma. Each may present in a similar manner, including with nasal obstruction. The optimal timing for surgical repair of congenital skull-base defects has not been definitively determined with consideration given to the risks and benefits for each patient. In the case of an encephalocele, intact mucosal membrane and dura separating the intracranial contents from the nasal cavity may not always be adequate to prevent meningitis, although the risk for meningitis in the absence of a cerebrospinal fluid (CSF) leak has been reported as low for the first 5 years of life and it may be safe to delay definitive surgical repair until the child is of sufficient size to make endoscopic repair feasible (typically age 2–3 years). Actively leaking defects should be repaired earlier if possible, because the risk of meningitis in the presence of dural and mucosal defects is probably higher. The presentation in this case, with recurrent meningitis and the associated midline defects, prompted us to choose a more definitive surgical approach. It has also been suggested that a child with a sincipital or basal defect and mild hypertelorism should have the encephalocele treated in early childhood to allow the facial skeleton to remodel with growth. However, in this case, the encephalocele was situated in a posterior and completely intranasal position, the hypertelorism was not likely related directly to the intranasal mass, and the encephalocele was unlikely to affect facial growth.
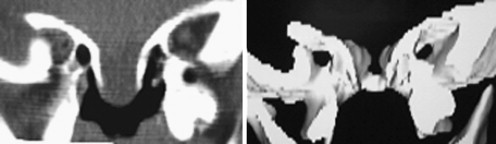
The position and size of the mass need to be considered preoperatively. In this case, although the mass was relatively small, it was in a nonfavorable position, arising from the ethmoid roof in the midline with an intact non-displaced septum and inferior turbinates. Often, large intranasal masses displace the septum and turbinates, making access easier. Basal encephaloceles may harbor vital herniated structures. This makes it important to consult with a neurologist/neurosurgeon before surgery to make every effort to preserve tissue during repair. Any element of hydrocephalus should be dealt with first, followed by an elective single-stage endoscopic repair.
Surgical Technique
The surgery was performed under general anesthesia, with the patient positioned with 30° head elevation. An image-guidance system was set up and used during the surgery. The nose was prepared with topical vasoconstriction using 1:1000 adrenalin-soaked neuropatties for 10 minutes and then the anterior part of the nasal septum was infiltrated with xylocaine and adrenaline (1:100,000). A traditional 4-mm, 0° nasal endoscope was used for the majority of the procedure to optimize the visual fields. Smaller 2.7-mm endoscopes are avoided unless anatomic constraints prevent use of the wider endoscopes. Access was gained through a hemi-transfixion incision on the right side of the nasal septum and the mucosa was raised along a subperichondrial and subperiosteal plane to the back of the septum. Superior and inferior parallel releasing incisions were then made along the maxillary crest and superior septum toward the posterior choanae and sphenoid ostium respectively, creating a long vascularized mucosal flap pedicled on the sphenopalatine artery. A similar flap was created on the left side, although this was not extended as far anteriorly, thus preserving the mucosa over the caudal septum. To maximize access and working space, the middle turbinate on the right side was resected. The mucosa was preserved in case additional free mucosal grafts were required to close any skull-base defects. Nasal septal cartilage was removed, preserving an appropriate caudal strut for tip support, and saved for possible use in the reconstruction of the skull-base defect.
To make use of all possible avenues, we passed a nasoendoscope through the cleft palate as an alternative way of visualizing the mass in the posterior nasal cavity as the lesion was situated at the posterior part of the nasal septum. We used special pediatric instruments, such as pediatric dissectors, aspirators, sickle knifes, and small Kerrison punches.
The meningoencephalocele was identified and resected from the nasal floor up to the skull base using bipolar cautery ( Fig. 2 ). The abnormal herniation of the meninges and brain was carefully repositioned.
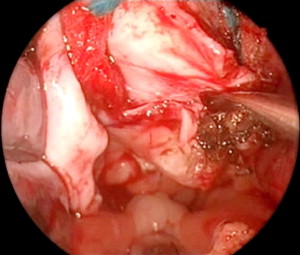
The bony skull base had a V- or funnel-shaped defect ( Fig. 3 ). This did not support a cartilage graft to hold the meningoencephalocele. So, instead we used fascia lata for an intracranial support and the two sphenopalatine artery septal vascularized flaps were laid over the defect ( Fig. 4 ). Fibrin glue was placed principally around the edges of the flap ( Fig. 5 ) and then Gelfoam was placed over the reconstruction, completely covering it to prevent the dressing or catheter from accidentally pulling on the graft when removed. A Foley catheter was inserted and inflated to keep the nasal packing in place. A lumbar drainage catheter was inserted at the start of the procedure and kept in situ postoperatively.
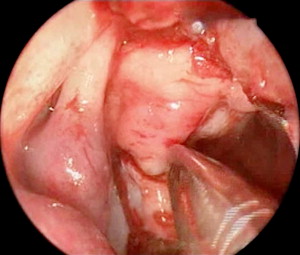

After the procedure, the child was sedated for 2 days to facilitate the use of the nasal packing. Antibiotics were given to the patient before the procedure and for 10 days after the surgery.
On the third postoperative day, the Foley catheter was removed and all sedation ceased. There were no signs of a CSF leak. The child was discharged from the hospital with no signs of CSF leak or infection on the sixth postoperative day.
One minor complication postoperatively was a representation with nasal obstruction. Again a broad differential needs to be considered, including such postoperative complications as bleeding with resulting clot, crusting, recurrence of the encephalocele, septal hematoma or collection, mucosal congestion, and synechiae. In this case, nasal obstruction was due to the Gelfoam, which slipped back into the nasal cavity, obstructing airflow but not causing respiratory distress. This was removed under vision in the clinic and no other problems were encountered.
During follow-up each week, the nasal area was examined and suctioned in the clinic. These nasal toilets were performed with great care to avoid causing any undue stress to the patient. Imaging studies were performed 30 days after the procedure.
There was no evidence of recurrence, any complications, or CSF leaks.
Discussion
Transnasal endoscopic techniques can now be performed relatively safely in fairly young children for the treatment of complex pathology, such as choanal atresia, orbital cellulitis, and meningoencephaloceles. However, pediatric patients require meticulous preparation, specialized instrumentation, and specialized staff involved in their perioperative care. The two main challenges in this case were access and closure. For access, we used a binostril technique and made use of the septal cleft to maximize exposure. To ensure adequate skull-base closure, we used nasal septum flaps pedicled at the sphenopalatine artery. These vascularized flaps receive direct blood supply from the sphenopalatine artery, making them much more robust as grafts than alternative graft sources in the closure of skull-base defects. In adults, the use of these flaps is almost routine in skull-base surgeries performed at many medical centers; however, their use has not been well described in pediatric patients. The creation of the flaps bilaterally not only allowed for plenty of tissue to cover the skull-base defect but also facilitated access.
Potential flap complications include mucoceles, synechia, infection, and failure, such as failure stemming from retraction of the graft over time, especially in anterior-based defects.
Another point to consider is the nasal packing. There are few nasal packs specially designed for pediatric patients. We try to minimize the use of packing. We used Gelfoam soaked in antibiotics in the nasal cavity to protect the graft from additional packing and offer some support. Although this is generally a safe and reliable option, the displacement of the packing postoperatively demonstrates a potential complication, such as the nasal obstruction seen in this case. Rarely it can pose a greater risk, such as aspiration of the packing material. Part of the reason a Foley catheter was inserted was not only to support the grafts, but also to prevent the displacement of the packing into the nasopharynx. Specific to this patient was the cleft soft palate, which allowed for improved access during the case. However, postoperatively the cleft palate did not provide any support for the packing, which probably made displacement more likely.
There is no consensus in the literature for the length of time nasal packing should remain in place. A balance between needing to keep the patient sedated to tolerate the packs and removing the packs too soon needs to be considered. We removed the Foley catheter balloon 2 days after the surgery, but the absorbable material was left in place to be resorbed or washed out.
A multidisciplinary team is also fundamental to a good outcome. The team managing this patient was made up of a pediatrician, a pediatric anesthesiologist, an otolaryngologist, a neurosurgeon, an intensive care pediatrician, and a radiologist, who were all instrumental in the success.
Case 2: 14-year-old male with nasal bleeding and onset nasal obstruction
A 14-year-old male presented with repetitive episodes of minor nasal bleeding and a gradual onset nasal obstruction over 3 years. Initially, the nasal obstruction was intermittent and fluctuated from side to side, but then the nasal obstruction became more persistent on the left side. The bleeding was considered minor and he underwent chemical cauterization with trichloroacetic acid 70% on several occasions. He also had a number of left middle-ear infections over the last year. Eventually he presented with poor nasal airflow interfering with his sporting activity. An endoscopy was performed, which demonstrated a purplish nasal mass occluding the posterior nasal cavity ( Fig. 6 ). There were no orbital or visual changes, cranial nerve function was normal, and the only positive finding on examination was in the left middle-ear.
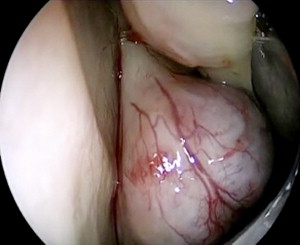
In a younger child, such as the child discussed in case 1, the top differential diagnoses include a glioma, dermoid, encephalocele, or a dacryocystocele. In an older child, especially a male, then a juvenile nasopharyngeal angiofibroma (JNA) must feature high on the differential list. There are other possible diagnoses. If the mass exhibits rapid growth or malignant potential, possible diagnoses include hemangioma, a lymphoma, craniopharyngioma, and rhabdomyosarcoma. If the mass arises from the posterior nasopharynx, then possible diagnoses include a Rathke’s pouch or Thornwaldt’s cyst. See Box 1 for summary.



