Chapter 53 Branch Vein Occlusion
Introduction
Branch retinal vein occlusion (BRVO) is a common cause of retinal vascular disease.1 The Beaver Dam Study estimated the 15-year cumulative incidence of retinal vein occlusions (RVO) at 2.3% in the population, with a majority of these (78%) being BRVO.2 BRVO affects males and females equally and occurs most frequently between the ages of 60 and 70. The pathologic interruption of venous flow in these eyes almost always occurs at a retinal arteriovenous intersection, where a retinal artery crosses over a retinal vein.3–7 Systemic vascular diseases such as hypertension and arteriosclerosis are risk factors for BRVO, probably because they lead to thickening of the retinal artery.3,7 Other risk factors for BRVO include diabetes, smoking, hyperlipidemia, glaucoma, and ocular inflammatory disease.8 Antiphospholipid antibodies, elevated plasma homocysteine levels, and low serum folate levels have also been associated with increased risk of vein occlusion.9–11 A decreased risk is present in those with higher serum levels of high-density lipoprotein and light to moderate alcohol consumption.8 Other studies have suggested an increased risk of BRVO in eyes with shorter axial lengths.12–15 This section discusses the pathophysiology, clinical features, evaluation, and treatment of patients with BRVO.
Pathogenesis
Because BRVO mostly occurs at arteriovenous crossings,4,8,16 underlying arterial disease may play a causative role. In 99% of 106 eyes with BRVO, the artery was located anterior to the vein at the obstructed site.7,8 Histopathologically, the retinal artery and vein share a common adventitial sheath, and in some cases, a common medium.17 The lumen of the vein may be compressed up to 33% at the crossing site.7,18 The vitreous may also play a role in compression of susceptible arteriovenous crossing sites, as evidenced by studies demonstrating that eyes with decreased axial length and higher likelihood of vitreomacular attachment at the arteriovenous crossing are at increased risk of BRVO.13,19
Some have postulated that turbulent blood flow at the crossing site causes focal swelling of the endothelium and deeper vein wall tissue, leading to venous obstruction.17,19,20 Other reports have demonstrated actual venous thrombus formation at the point of occlusion.3,18 The resulting venous obstruction leads to elevation of venous pressure that may overload the collateral drainage capacity21 and lead to macular edema and ischemia by mechanisms that are still under investigation. Unrelieved venous pressure can also result in rupture of the vein wall with intraretinal hemorrhage.17 Vision loss from RVOs is typically due to macular ischemia, macular edema, or complications from neovascular disease.
Clinical features
Signs
Patients typically present with a wedge-shaped distribution of intraretinal hemorrhage that is less marked if the occlusion is perfused (or nonischemic), and more extensive if the occlusion is nonperfused (or ischemic) and associated with retinal capillary nonperfusion. The Branch Vein Occlusion Study Group defined ischemic BRVO as those with greater than a total of five disc diameters of nonperfusion on fluorescein angiography (FA).1 The location of the venous blockage determines the distribution of the intraretinal hemorrhage; if the venous obstruction is at the optic nerve head, two quadrants of the fundus may be involved, whereas if the occlusion is peripheral to the disc, one quadrant or less may be involved with the intraretinal hemorrhage. If the venous blockage is peripheral to tributary veins draining the macula, there may be no macular involvement and no decrease in visual acuity. The most common location for BRVOs is in the superotemporal quadrant.5,6,22 This favored location may be attributed to a larger number of arteriovenous crossings in the superotemporal quadrant.
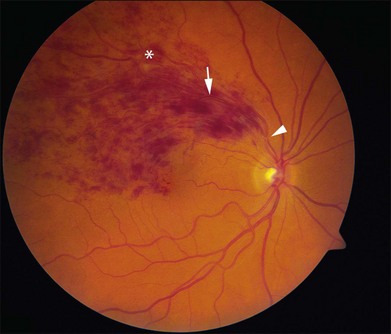
Figure 53.1 demonstrates the typical acute appearance of a BRVO involving the superotemporal quadrant of the right eye. A narrowed branch retinal vein passing under a retinal artery can sometimes be identified proximal to the hemorrhage. Rarely, a patient may present initially with very little intraretinal hemorrhage, which then becomes more extensive in the succeeding weeks to months. In these instances, it is presumed that an incomplete block at the arteriovenous crossing has progressed to more complete occlusion.
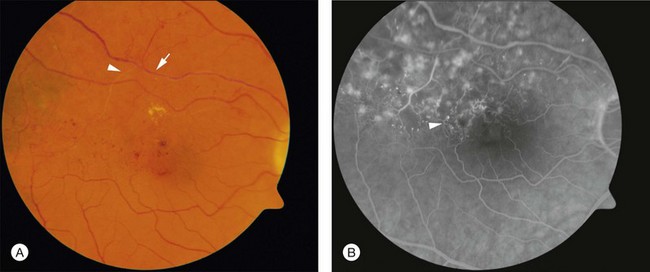
Over time the intraretinal hemorrhage may completely resorb. Without the characteristic segmental distribution of intraretinal hemorrhage, the ophthalmoscopic diagnosis may be more difficult, but the segmental distribution of retinal vascular abnormalities that occurred during the acute phase will persist and be apparent on FA. In many cases, macular edema may also be detected by optical coherence tomography (OCT). Consequently, in the chronic phase of the disease, after intraretinal hemorrhage absorption, the diagnosis may depend on detecting a segmental distribution of retinal vascular abnormalities that may include capillary nonperfusion, dilation of capillaries, microaneurysms, telangectatic vessels, and collateral vessel formation (Fig. 53.2).
Nonocular signs such as systemic hypertension have been associated with BRVOs.2,8,23 Thus, systemic blood pressure may be elevated. In bilateral cases or cases involving young patients, systemic manifestations of infectious disease, inflammatory or autoimmune conditions, neoplasm, or hypercoagulable states may be present.
Complications
There are three common vision-limiting complications of BRVO: (1) macular edema; (2) macular ischemia; and (3) sequelae of neovascularization. It is important to appreciate the variability of these complications before considering the benefits of treatment.24–26
Retinal and iris neovascularization, vitreous hemorrhage, traction retinal detachment, and neovascular glaucoma are complications that manifest late in the course of the disease due to ischemia. With the exception of macular ischemia, these complications can largely be treated or prevented. Thus, it is important that patients with BRVO be closely followed. From the Branch Vein Occlusion Study, 31–41% of patients with ischemic BRVO (defined as >5 disc diameters of nonperfusion on FA) developed neovascularization or vitreous hemorrhage, compared with 11% of patients with nonischemic BRVO.1
Clinical evaluation
Clinical examination
Fluorescein angiography
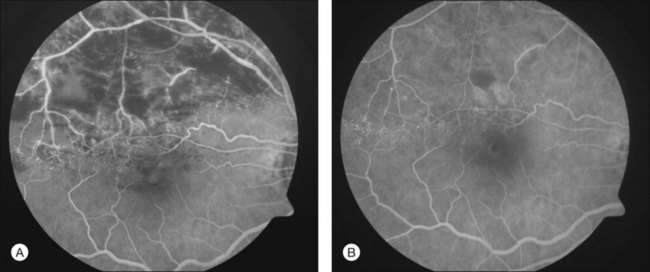
To help verify the diagnosis and evaluate for complications, FA should be obtained to delineate the retinal vascular characteristics that may have prognostic significance: macular leakage and edema, macular ischemia, and large segments of capillary nonperfusion that may portend eventual neovascularization. FA is the only technique that will accurately define the capillary abnormalities in BRVO; it is therefore particularly important that high-quality angiography be obtained (Fig. 53.3).
When FA demonstrates macular leakage and edema with cystoid involvement of the fovea, but no capillary nonperfusion, it is presumed that the macular edema is the cause of vision loss. Under these circumstances, about one-third of patients will spontaneously regain some vision. However, patients who have had decreased vision for over 1 year as a result of macular edema are much less likely to regain vision spontaneously. When macular edema is present ophthalmoscopically within the first 6 months after a BRVO and there is little or no leakage on FA, macular ischemia may be the cause of the macular edema. In such circumstances, the edema almost always spontaneously resorbs in the first year after the occlusion, often with return of vision.27
Wide-field angiography
Wide-field angiography is not a commonly used imaging modality for patients with BRVO, but a recent study supports its utility. Ultrawide-field FA using the Optos C200MA revealed a correlation between nonperfusion in the peripheral retina with macular edema and retinal neovascularization.28 Future studies to determine if laser photocoagulation of the nonperfused peripheral retina decreases macular edema and neovascularization may alter the therapeutic paradigm. In patients with recalcitrant macular edema or retinal neovascularization, wide-field angiography may reveal peripheral areas of nonperfusion helping to guide targeted laser photocoagulation.
Optical coherence tomography
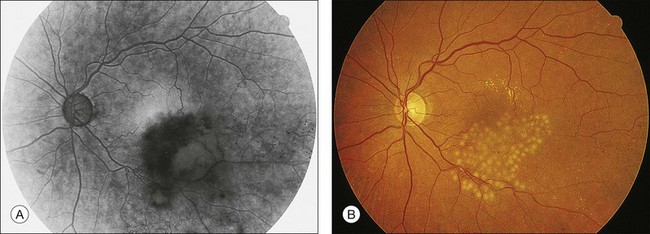
OCT has arguably become the most important imaging modality in the treatment of patients with BRVO and macular edema. OCT offers a noninvasive and rapid method of quantitatively measuring macular edema. OCT is frequently used to monitor the response to treatment of macular edema and has been used in place of FA in some treatment trials for BRVO. Unlike FA, intraretinal hemorrhages have a minimal effect on the interpretation of OCT, making this imaging modality helpful, even in the acute setting with foveal hemorrhage. The characteristic findings of BRVO on OCT are cystoid edema, intraretinal hyperreflectivity from hemorrhages, shadowing from edema and hemorrhages, and occasionally subretinal fluid29,30 (Fig. 53.4A). In chronic cases, photoreceptor inner-segment–outer-segment junction abnormalities from long-standing macular ischemia and macular edema may also be seen (Fig. 53.4B).
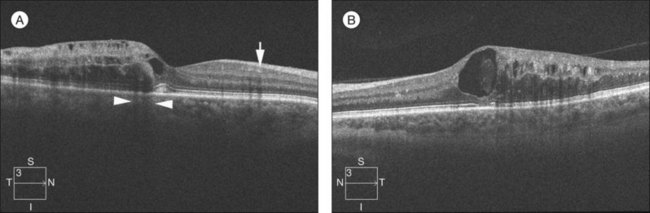
Fig. 53.4 Spectral-domain optical coherence tomography of an eye with a branch retinal vein occlusion (BRVO). (A) Raster scan of the BRVO in Figure 53.1 reveals cystoid macular edema, intraretinal fluid, and shadowing (between the arrowheads). Intraretinal heme (arrow) appears hyperreflective and produces a shadow on optical coherence tomography. (B) Raster scan of a chronic BRVO with inner-segment–outer-segment abnormalities and a large cyst.
Diagnostic workup
Young patient
BRVO typically occurs in patients beyond their sixth decade of life. When they present in younger patients, an underlying predisposing condition may be suspected. In this younger patient population, a detailed history should be taken, including the use of oral contraception in females, or the use of other medications that can promote a hypercoagulable state or thromboembolism. Workup should be performed in consultation with an internist. Systemic blood pressure should be checked and the patient should be screened for diabetes. In suspected cases, infectious causes such as Lyme disease, syphilis, or human immunodeficiency virus should be screened. In young patients with other systemic findings suggestive of inflammatory disease or coagulopathy, workup should include a complete blood count, prothrombin time/partial thromboplastin time/international normalized ratio, lipid panel, serum homocysteine, anticardiolipin antibodies, antinuclear antibodies with lupus anticoagulant, protein C/S, and activated protein C resistance (factor V Leiden).10,11,31
Bilateral or numerous BRVO patients
In bilateral cases or in cases with a history of multiple BRVOs, searching for an infectious or inflammatory disorder or hypercoagulopathy may be warranted. There are numerous case reports of patients with bilateral vein occlusions and systemic inflammatory disorders or hypercoagulopathies; however the vast majority can be attributed to systemic hypertension.32–37 The workup should proceed in the manner described for young patients.
Treatment options
Laser treatment
Branch Vein Occlusion Study for macular edema
The collaborative Branch Vein Occlusion Study (BVOS),38 a multicenter randomized clinical trial supported by the National Eye Institute, reported that argon laser photocoagulation may reduce visual loss from macular edema for those eyes that meet study eligibility criteria and are treated according to that protocol. Important eligibility criteria included fluorescein-proven perfused macular edema involving the foveal center, absorption of intraretinal hemorrhage from the foveal center, recent BRVO (usually 3–18 months’ duration), no diabetic retinopathy, and vision reduced to 20/40 or worse after best refraction.
In the BVOS,38 argon laser photocoagulation was applied in a grid pattern throughout the leaking area demonstrated by FA (Fig. 53.5). Coagulation extended no closer to the fovea than the edge of the capillary-free zone and no further into the periphery than the major vascular arcade. Recommended treatment parameters included a duration of 0.1 second, a 100-µm diameter spot size, and a power setting sufficient to produce a “medium” white burn. FA was repeated 2–4 months after the treatment, and additional photocoagulation was applied to residual areas of leakage if reduced visual acuity persisted. Improvement in visual acuity was assessed in several ways.38 When improvement was defined as reading two or more Snellen lines (beyond baseline) at two consecutive visits, treated eyes showed visual improvement more often than untreated eyes. After 3 years of follow-up, 63% of treated eyes gained two or more lines of vision, compared to 36% of untreated eyes. The average gain in visual acuity for treated eyes was one more Snellen line than in untreated eyes.
Before laser photocoagulation is performed, it is important to obtain high-quality FAs of the macula; the FA must demonstrate that the macular edema involves the center of the fovea and that there is not a large amount of capillary nonperfusion adjacent to the capillary-free zone that could explain the visual loss. In addition, it is important to follow patients for a length of time sufficient to ascertain that macular edema is not resolving spontaneously. During this period of follow-up, it should be demonstrated that there is clearing of intraretinal hemorrhage and that there is no hemorrhage in the center of the fovea that could account for a spontaneously reversible cause of visual loss. In the application of the grid photocoagulation, laser absorption occurs at the level of the pigment epithelium; photocoagulation is not applied to close the leaking and dilated capillary vasculature directly and immediately. Although it is not understood how the laser treatment may act in lessening edema, it is interesting to note that preliminary experimental studies in the normal primate have shown a decrease in capillary diameter when this form of therapy is used and when laser absorption occurs at the level of the pigment epithelium.39 One explanation for the effect of grid pattern photocoagulation is that it results in a thinning of the retina (in particular the outer retina), reducing oxygen consumption and increasing choroidal delivery of oxygen to the inner retina, producing a consequent autoregulatory constriction of the retinal vasculature in the leaking area and thereby decreasing the edema.
For the grid treatment used in the BVOS, the argon blue-green wavelength was employed.38 This is the only wavelength that has been proven effective and it is unknown whether argon green and krypton red photocoagulation are equally effective. In other diseases, when laser treatment is applied inside the capillary-free zone, it is recognized that krypton red and argon green laser photocoagulation are absorbed less than blue-green by the xanthophyll pigment of the inner retina that is present in increasing concentrations close to the foveal center. However, because the grid treatment never comes closer to the fovea than the capillary-free zone, the BVOS did not encounter any problems with the argon blue-green laser in this region; consequently, this laser continues to be recommended.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree