Chapter 77 Autoimmune Retinopathies
Introduction
Autoimmune retinopathies (AIR) represent a group of inflammatory mediated retinopathies with otherwise unexplained vision loss associated with visual field deficits, photoreceptor dysfunction as evidenced on electroretinography (ERG), and the presence of circulating autoantibodies targeted against retinal antigens. Clinically, the fundus often appears normal, but some patients may show retinal vascular attenuation, diffuse retinal atrophy with or without pigmentary changes or waxy disc pallor. There are usually few or no intraocular inflammatory cells.1,2
AIR presumably results from an immunologic attack on the retina by antibodies directed against retinal antigens. The first case of vision loss and photoreceptor degeneration associated with cancer was described by Sawyer et al. in 1976.3 Paraneoplastic retinopathy, as a term, was first used by Klingele and associates in 1984, and has become the more general term used for autoimmune retinopathies associated with systemic malignancy since then.4 Several forms of autoantibody-mediated retinopathy are described: cancer-associated retinopathy (CAR) syndrome, melanoma-associated retinopathy (MAR) syndrome, or autoimmune retinopathy of other types.3,5–11 AIR can be categorized in two groups: (i) autoimmune retinopathy associated with cancer or other malignancies (paraneoplastic retinopathy or paraneoplastic autoimmune retinopathy); (ii) autoimmune retinopathy without any evidence of malignancy (nonparaneoplastic autoimmune retinopathy). Cancer-associated retinopathy and other paraneoplastic retinopathies will be covered in Chapter 134 (Remote effects of cancer on the retina). Autoimmune retinopathy is the preferred term for an acquired, presumed immunologically mediated retinopathy caused by antiretinal autoantibodies in the absence of a malignancy. This chapter will emphasize the latter.
Epidemiology and mechanisms
The prevalence of AIR is unknown, but the condition is believed to be rare. Clinical reports of nonparaneoplastic AIR consist only of case reports and a few small cohorts.12,13 AIR remains an ill-defined disorder and the lack of standardized diagnostic criteria may be contributing to the underestimation of its prevalence. Though circulating autoantibodies to retinal antigens have been shown to be associated with retinal dysfunction, the mechanisms by which these antibodies cause dysfunction are not entirely understood.14 Multiple retinal proteins have been found to be antigenic, including recoverin, carbonic anhydrase, α-enolase, arrestin, transducin-β, carbonic anhydrase II, TULP1, neurofilament protein, heat shock protein-70, photoreceptor-cell-specific nuclear receptor (PNR), Müller-cell-specific antigen, transient receptor potential cation channel, subfamily M, member 1 (TRPM1) and many other yet-unidentified antigens15,16 (Table 77.1). Some of these antigens are retina-specific, such as recoverin and rhodopsin, while others can be found in nonretinal tissues as well, such as enolase. Among these, recoverin and enolase are the most extensively studied antigens in the context of AIR. Recoverin is a 23 kDa calcium-binding protein found in both rods and cones. Enolase is a 48 kDa glycolytic enzyme whose α- and β-isoforms are found in many tissues, and γ-isoform in neuronal tissues.39 In a large series, more than 30% of patients with antiretinal antibodies tested positive for antienolase antibody.40 Antibodies against α-enolase appear to be fairly ubiquitous; they are found in multiple autoimmune diseases and even in healthy subjects.41–44 This has been, in part, attributed to the multifunctional nature of α-enolase.14,25,45
Table 77.1 Proteins targeted by antiretinal antibodies
| Recoverin (23 kD) | CAR, npAIR6,17–24 | |
| Alpha-enolase (46 kD) | CAR, npAIR20,21,25–28 | |
| Carbonic anhydrase II (CA II) (30 kD) | npAIR20,21 | |
| Tubby-like protein 1 (TULP-1) (65 kD) | CAR29 | |
| Heat shock protein 70 (HSC 70) (70 kD) | CAR19 | |
| Transducin (35 kD) | MAR30 | |
| Arrestin (S-Antigen) (48 kD) | MAR, npAIR21,31 | |
| Interphotoreceptor binding protein (IRBP) (141 kD) | npAIR21 | |
| Unknown proteins | 22 kD, 34 kD, 35 kD, 37 kD, 40 kD, 68 kD | npAIR32–34 |
| 34 kD, 40 kD, 46 kD, 60 kD, 70 kD | CAR 22,35–38 | |
Antibodies against the listed proteins were identified in the serum of patients with AIR or AIR-like clinical findings. CAR, cancer-associated retinopathy; MAR, melanoma-associated retinopathy; npAIR, non-paraneoplastic autoimmune retinopathy.
Of all these antigens, recoverin and CAR has the strongest association. Recoverin has been shown to be expressed by the tumor cells of patients with CAR.46 While antirecoverin antibody is most specific to CAR, it has also been found in patients with nonparaneoplastic AIR, as well as patients with small-cell lung carcinoma without any retinopathy.17,18,47 Recoverin and α-enolase have been shown to be highly expressed in cancer cells in patients with paraneoplastic AIR. It is plausible that the disease is triggered by molecular mimicry between retinal proteins and tumor antigens in cases of paraneoplastic AIR and presumed viral or bacterial proteins26 in the case of nonparaneoplastic AIR.26,48,49 Regardless of the presence or absence of malignancy, autoimmune retinopathies appear to share common clinical features.
Experimental studies have attempted to shed light on the pathogenic mechanisms of autoimmune retinopathy. In vitro studies have demonstrated that some of the antiretinal antibodies are, indeed, cytotoxic to retinal cells, and that the cellular internalization of the antibody leads to apoptosis.19,25,27,50–52 This antibody-mediated apoptosis is independent of complement and involves caspase pathways and intracellular calcium influx. Similarly, in vivo studies showed that intravitreal injection of antirecoverin antibody causes apoptosis of retinal cells and a decrease in ERG responses.19,53,54 Recoverin also acts as a uveitogenic antigen and can induce autoimmune retinopathy-like disease in animal models leading to reduced scotopic and photopic ERG responses.55,56 Both in vitro and in vivo studies showed that antirecoverin antibody-triggered apoptosis occurs only in recoverin-positive cells.57 Antirecoverin antibodies target photoreceptor cells and anti-α-enolase antibodies appear to target ganglion cells. Antibodies targeting retinal bipolar cells have been associated with MAR. Additionally, injection of immunoglobulins isolated from serum of patients with MAR into the vitreous of monkeys led to ERG changes similar to those observed in MAR, indicating the pathogenicity of the antiretinal antibodies.58 Evidence from these studies suggests that antiretinal autoantibodies can target virtually any retinal cell type – photoreceptor cells, ganglion cells, bipolar cells – and cause retinal dysfunction.20,50,59 It is still unclear, however, why some patients with antiretinal antibodies develop retinopathy while others do not.
Clinical features
Clinical features of AIR are quite variable. Serum antiretinal autoantibodies have been associated with loss of vision and visual field defects as well as electrophysiologic changes in patients with autoimmune retinopathy, but the exact mechanism has not been fully understood. In particular, the pathogenicity and specificity of these autoantibodies with respect to clinical findings have yet to be determined. Nevertheless, patients with AIR appear to share common clinical features despite the heterogeneity in the detectable circulating antiretinal antibodies.12,20,60
The clinical manifestations of nonparaneoplastic AIR is quite variable, there are no clear guidelines or consensus on the diagnosis of autoimmune retinopathy. Commonly recognized clinical manifestations include:12,13
1. Symptoms: acute, subacute or chronic onset of photopsia, dyschromatopsia, nyctalopia, photoaversion, scotomas, sometimes central vision loss.
2. Fundus findings: normal-appearing fundus, or waxy disc pallor, attenuated retinal vasculature and retinal pigment epithelial (RPE) atrophy or mottling. There are few or no intraocular inflammatory cells.
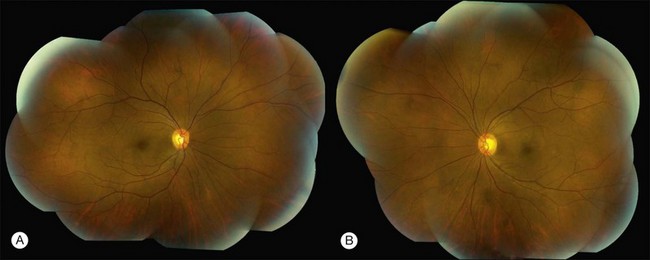
Patients with nonparaneoplastic AIR present with subacute or chronic vision loss. Patients typically complain of color vision changes, photosensitivity or photoaversion, and varying degrees of nyctalopia. Other presenting symptoms include floaters, photopsias, scotomas, most commonly paracentral, and constricted visual fields. Of these, photosensitivity, photoaversion, difficulty seeing in bright light, reduced visual acuity, dyschromatopsia and central scotomas are suggestive of cone dysfunction, whereas nyctalopia (night blindness) and midperipheral scotomas are suggestive of rod dysfunction. Depending on the extent and the distinct retinal cell involvement, visual acuity, particularly in the earlier stages, may be deceivingly good. On presentation to a tertiary care center, most patients may have been diagnosed with nonspecific retinal degeneration or isolated cases of retinitis pigmentosa (RP). Fundus examination can be unremarkable or may show signs of retinal degeneration such as waxy disc pallor, attenuated retinal vasculature with or without pigmentary changes, or diffuse atrophy. There may be mild or no inflammatory cells in the anterior chamber or the vitreous12,13,61 (Fig. 77.1).
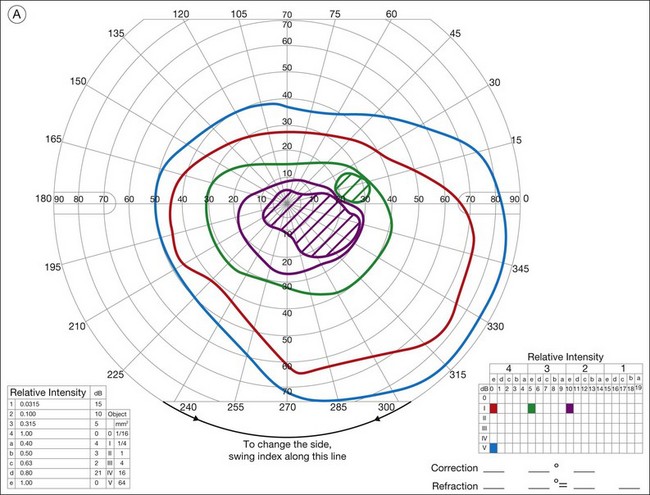
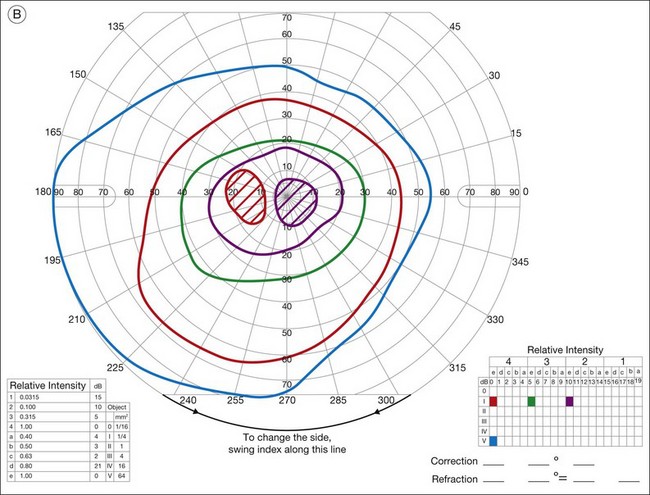
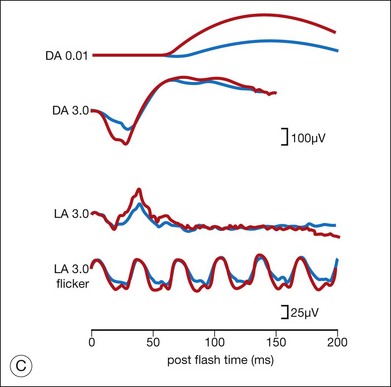
Ancillary testing with fluorescein angiography (FA) or optical coherence tomography (OCT) may show mild retinal vascular staining or leakage, or cystoid macular edema (CME) in some cases.12 Visual field testing may confirm scotomas and constricted visual fields. While static visual field testing may be better for demonstrating and following central and paracentral scotomas, kinetic visual fields are better for measuring peripheral field constriction. However, kinetic visual fields have the disadvantage of examiner dependence, the need for patient cooperation, inter-tester variability, and lack of standardization of parameters.62,63 ERG may demonstrate abnormal rod, cone, Müller cell, and bipolar cell responses and delay in implicit times.12,13,26,32 However, the majority of studies involving ERG findings in AIR come from paraneoplastic retinopathies (Fig. 77.2).
AIR is almost always bilateral, although involvement can be asymmetric. A family or a personal history of systemic autoimmune disease can be common among patients with nonparaneoplastic AIR.12,33 There is a female preponderance (63–66%), and average age at diagnosis appears to range from 51 years to 56 years.12,13,20 The typical patient would be a middle-aged or older adult in their fifth to sixth decades with no history of visual problems prior to the onset of photopsias, scotomas, and other symptoms consistent with AIR, and no family history of RP.
As might be expected for an entity with no consensus in diagnosis, retrospective studies in patients with nonparaneoplastic AIR showed that clinical features vary considerably. In one study, diffuse retinal atrophy was seen in the majority of patients (83%) and pigment deposits in only a small proportion (13%), macular edema was present in approximately half of the cases, while another study showed pigmentary changes in approximately half of the patients and macular edema in 24%. The most common symptoms at onset were subacute vision loss, photopsias, and nyctalopia. Similarly, in a series of 12 patients with antienolase-associated retinopathy, clinical characteristics included visual loss, normal-appearing fundus in most except for vascular attenuation, optic nerve head pallor in some, and mainly abnormal cone responses. In all of these studies, however, there was a female predominance and the age at onset was similar.12,13,20,26,
Diagnosis
The diagnosis of AIR is difficult as there are no definitive or standardized tests. The presumptive diagnosis relies on the presence of the above clinical manifestations, usually more than one, along with demonstration of serum antiretinal antibodies. Most patients will have more than one antiretinal antibody. If the clinical features of autoimmune retinopathy and the circulating antiretinal antibodies are present, and if there is no malignancy at presentation or following a thorough investigation, a diagnosis of nonparaneoplastic AIR is made (Table 77.2).
Table 77.2 Diagnostic criteria for nonparaneoplastic AIR (proposed by the authors)
| Must have all of the criteria | Must have ≥2 of the criteria |
|---|---|
| No evidence of malignancy | Symptoms: Photopsias or scotomas or dychromatopsia or nyctalopia or photoaversion |
| No family history of RP | Fundus changes* |
| (+) serum antiretinal antibodies | Visual field abnormality* |
| ERG abnormality* |
* Fundus changes, visual field or ERG abnormalities as described in clinical manifestations and diagnosis section (see text).
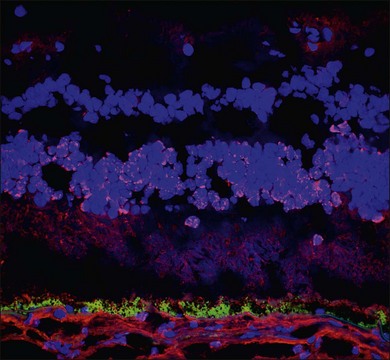
Demonstration of serum antiretinal antibodies can be done using various methods, including Western blot (WB), immunohistochemistry (IHC) or enzyme-linked immunosorbent assay (ELISA). Western blot is a protein immunoblot of patients’ serum incubated with extracts of a normal donor human retina and shows antiretinal IgG bands. It identifies antibody activity based on size of the protein, and the interpretation relies on intensity of protein bands on a photographic film. The results may be affected by multiple technical factors and lack specificity. For example, a 23 kDa band on WB does not necessarily mean antibody against recoverin; there may be other antigens with the same size. The immunohistochemical detection of antiretinal antibodies, on the other hand, involves fixing patient serum against frozen donor human retina (or monkey or mouse retina) using immunohistochemical staining. Sections are then analyzed using light microscopy to determine which layers of the retina show binding of the antiretinal antibody. The advantage with IHC is the ability to localize the specific site of binding within the retina (Fig. 77.3). ELISA is similar, in principle, to WB and IHC and involves adding various dilutions of patient sera into wells coated with specific retinal proteins and binding is detected using secondary antibodies.10,64 All of these techniques, however, lack standardization.15 Moreover, it is important to remember that the mere presence of these autoantibodies does not warrant a diagnosis of AIR, nor does it mean they are pathogenic. Antiretinal antibodies have been detected in patients with other retinal diseases, uveitis, retinal degenerations including age-related macular degeneration, and even in normal controls.21,65–68 Conversely, they can be negative in patients with clinical features of what would be considered AIR.20 Despite the caveats, there are few centers in the United States that perform antiretinal antibody testing. One center provides these services commercially through a CLIA (clinical laboratory improvement amendments) certified laboratory.69 WB and IHC are the more commonly performed techniques.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree