Chapter 126 Artificial Vision
Introduction
More than 1 million Americans are legally blind and 10% have no light perception, from various causes.1 Certain experimental approaches, such as gene (see Chapter 34, Gene therapy) and drug therapy may be a preventative or therapeutic option.2,3 However, once photoreceptors are nearly completely lost, such as in end-stage retinitis pigmentosa (RP) (see Chapter 40, Retinitis pigmentosa and allied disorders) or age-related macular degeneration (AMD) (see Chapters 63–66 on epidemiology/risk factors for AMD, pathogenesis, and diagnosis and treatment of dry and wet AMD, respectively), very few approaches4 can restore useful vision to blind patients. Retinitis pigmentosa (RP) is the leading inherited cause of blindness with 1.5 million people worldwide affected and an incidence of 1/3500 live births.5 Also, AMD is the leading cause of visual loss among adults older than 65, with 700 000 newly diagnosed patients annually in the USA, 10% of whom become legally blind each year.6 With an increased mean lifespan, particularly in the developing world, the number of people with age-related eye disease and resulting visual impairment is expected to double during the next three decades.7
Few treatment options exist for outer retinal degeneration. The advent of the anti-VEGF therapy has shown effectiveness for neovascular-AMD patients. This therapy is capable of preventing visual loss and even returning vision to patients treated in the initial phases.8–10 The therapeutic agents inhibit the growth of new blood vessels in the retina. Nevertheless, like most new therapies, it has limitations and drawbacks and there is some evidence that there is disease progression in spite of injections, especially in polypoidal choroidal vasculopathy.11 Moreover, a number of patients seek consultation when the neovascularization is advanced and hence irreversible vision loss has already occurred. Non-neovascular AMD can also become advanced leading to atrophic AMD (e.g., geographic atrophy). Other than the Age Related Eye Disease Study (AREDS) trial showing benefit for non-neovascular AMD patients receiving a formula containing high levels of antioxidants and zinc (500 mg of vitamin C; 400 IU of vitamin E; 15 mg of beta-carotene; 80 mg of zinc as zinc oxide; and 2 mg of copper as cupric oxide),12 there have been no other approved therapies, albeit many companies are trying to develop a therapy for this slowly progressive variant of AMD. Although gene therapy has not become a proven therapy in AMD, gene therapy has shown some success in Leber’s congenital amaurosis by targeting a specific mutation of the RPE65 gene.13–16 This is a tremendous scientific breakthrough for this retinal degeneration, despite the fact that the total number of eligible patients is small in number (approximately 1000). Neither anti-VEGF nor many of the proposed pharmacological treatments or gene therapy can address vision lost due to photoreceptor loss, since photoreceptors are not regenerated by these approaches.
Background and history of artificial vision
The concept of electrically stimulating the nervous system to create artificial vision was first introduced in 1929, when Foerster, a German neurosurgeon, observed that electrical stimulation of the visual cortex caused his subject to detect a spot of light (phosphene). He further demonstrated that the spatial psychophysical location of this phosphene depended upon the location of the electrical stimulation point over the cortex.17 The first serious effort (by today’s standards) of establishing an electrical artificial vision system was undertaken less than 50 years ago by Giles Brindley. Brindley’s implantation of an 80-electrode device onto the visual cortex of a blind patient revealed the possibilities of electrical stimulation to restore vision and the barriers to implementation of a suitable device. Brindley’s pioneering work has influenced all subsequent major efforts in the area of electronic visual prostheses. In the past 50 years, exponential advances in our understanding of electronics, physiology, and medicine have enabled the development of implantable microelectronic systems that overcome the shortcomings of Brindley’s large, immobile visual stimulator.18
Whether useful vision can be rendered via artificial visual prostheses depends upon establishing a definition of useful vision that is based on the minimum number of pixels required for human beings to accomplish activities of daily living. Several researchers have completed psychophysical experiments designed to determine the minimum acceptable resolution for useful vision. Brindley originally suggested that 600 points of stimulation (pixels) would be sufficient for reading ordinary print.18 More recent studies have tested humans with normal visual function by pixelating their vision via a portable phosphene simulator, consisting of a small head-mounted video camera and monitor. Patients then walked through an obstacle course and read pixilated text. In this fashion, it was determined that 625 electrodes implanted in a 1 cm2 area near the foveal representation in the visual cortex could produce a phosphene image with a visual acuity of approximately 20/30 and reading rates near 170 words/minute with scrolled text and 100 words/minute with fixed text.19–21 Further, a degree of learning was noted as walking speeds increased five-fold during 3 weeks of training.20
Studies simulating electrodes placed over the entire macula rather than a foveal pixelization have assessed the ability of subjects to recognize faces through a pixilated square grid. Parameters included grid size (10 × 10 to 32 × 32 dots), dot size, gap width, dot dropout rate, and gray scale resolution. The subjects achieved highly significant facial recognition accuracies in both high- and low-contrast tests with a marked learning effect documented. These results suggest that reliable facial recognition is possible even with crude visual prostheses, and possibly makes the task of engineering the implant easier as it would require fewer data/stimulation channels.21 The ability of subjects to read using a pixilated visual simulator has been evaluated in a separate cohort which demonstrated that most subjects are able to read fonts as small as 36 point (with all at 57 point) using a 16 × 16 pixel array.22,23
Visual prostheses
Cortical prosthesis
Building upon earlier observations of phosphene perception with cortical stimulation, Brindley and Dobelle began work in the 1960s towards functional, visual cortex prosthesis. They demonstrated the ability to evoke phosphenes and patterned perceptions by electrically stimulating the occipital cortex via permanently implanted electrodes.18,24–30 Both researchers implanted arrays with over 50 electrodes subdurally over the occipital pole, thus providing evidence of the ability to return the sensation of vision to individuals who had severed visual pathway anterior to the visual cortex. Dobelle’s 64-channel platinum electrode surface stimulation prosthesis allowed blind patients to recognize 6-inch characters at 5 feet (approximately 20/1200 visual acuity).28,29,31 Difficulties encountered in these experiments included the following: (1) controlling the number of phosphenes induced by each electrode; (2) interactions between phosphenes; (3) use of high currents and large electrodes that induced pain from meningeal stimulation; and (4) occasional focal epileptic activity following electrical stimulation.28,32,33 Patients in these initial experiments complained of not being able to appreciate distinct phosphenes, but rather reported seeing “halos” surrounding each phosphene.34
Since most of the visual cortex is deep within the calcarine fissure and inaccessible to cortical surface electrodes, intracortical stimulation was introduced in hopes of remedying the shortcomings of surface cortical stimulation via a lower-current, higher-fidelity system. The intracortical devices employed smaller electrodes closer to the target neurons, thus requiring less current and resulting in a more localized stimulation. The stimulus threshold is 10–100 times lower for intracortical prosthesis as compared to surface stimulation. Further, this approach allows for closer spacing of electrodes at 500 µm apart and thus possibly higher resolution. Initial studies, during which the intracortical prosthesis was implanted in humans for a period of 4 months, demonstrated the ability to produce phosphenes which usually had color.35 Documented advantages of the intracortical versus surface cortical implants include: (1) predictable forms of elicited phosphenes; (2) absence of flicker phenomenon; (3) reduction in phosphene interactions: (4) increased number of electrodes; (5) reduced overall power requirement.33,35–37
Current models of the intracortical prosthesis include the Utah electrode array. This device consists of multiple silicon spikes organized in a square grid measuring 4.2 mm by 4.2 mm.36 A platinum electrode is at the tip of each spike. A pneumatic system, which inserts 100 electrode devices into the cortex in about 200 ms, is required for minimal trauma during insertion of this array.38 The cortical visual prosthesis is advantageous over other approaches because it bypasses all diseased visual pathway neurons rostral to the primary visual cortex. As such, this approach has the potential to restore vision to the largest number of blind patients.
There are some limitations to the cortical visual prostheses. First, histologic changes for chronically implanted prostheses need to be further investigated.39–41 In the case of silicon-doped penetrating electrodes, tissue reaction has ranged from none to a thin capsule around each electrode track to gliosis and buildup of fibrotic tissue between the array and meninges.42 Second, the organization of the visual field is markedly more complex at the level of the primary cortex than at the retina or optic nerve and is not easily reproducible between various patients.34 Next, there is a high level of specialization of every area of cortex for various parameters including color, motion, and eye movement, making it unlikely to garner simple phosphenes from stimulation.43 Finally, surgical complications of this approach carry significant morbidity and mortality for the patient. The future success of the intracortical prosthesis requires further investigation of these areas.
Optic nerve prosthesis
Investigators have targeted the optic nerve as a potential site for the implementation of a visual prosthesis.44,45 Veraart et al. was the most recent of such groups attempting this method, employing the concept of a spiral nerve cuff electrode.46–49 Essentially, an electrode cuff is surgically implanted circumferentially on the external surface of the optic nerve. As this device does not penetrate the optic nerve sheath, it relies on the principle of retinotopic organization within the optic nerve. One group has recently implanted a chronic, self-sizing cuff with four electrodes into a human patient.50 Preliminary reports have demonstrated that electrical stimulation of the optic nerve produces colored phosphenes broadly distributed throughout the visual field.46,51
The optic nerve is an appealing site for the implementation of a visual prosthesis, as the entire visual field is represented in a small area. This area can be reached surgically and presents a viable anatomic location for an implant; however, there are several hurdles to overcome regarding this approach. First, the optic nerve is a densely consolidated neural structure with approximately 1.2 million axons in a 2-mm-diameter cylinder. While this allows for the entire visual field to be represented in a relatively small area, it is difficult to achieve focal stimulation of neurons, and to garner the exact retinotopy of the optic nerve. The dense packing of neurons requires a large number of electrode contacts from the prosthesis in a small area, increasing the risk of damage to the nerve.52 Surgical manipulation of this area requires dissection of the dura mater, creating possible harmful CNS effects including infection and possible interruption of blood flow to the optic nerve. Fourth, intervention at this point within the optic pathway requires intact retinal ganglion cells (RGC) and therefore is limited to the treatment of outer retinal (photoreceptor) degenerations. The optic nerve and RGC represent higher-order structures than the bipolar cells targeted by the retinal prosthesis. As such, the processing power of the bipolar, horizontal, and amacrine cells is lost and therefore much more image processing must be achieved by the implant rather than relying on intact human physiologic pathways. Last, the nerve fibers from the macula lie most centrally within the optic nerve. Cuff electrodes, thus, are farthest away from macular fibers and this will dramatically limit the use of this approach especially for AMD as the peripheral fibers will get stimulated along with the central macular fibers. Future development of this technology must address the above issues. Investigators have also proposed intraneuronal stimulation devices in order to more accurately target individual neurons within the optic nerve.53
Veraart et al. published results of an optic nerve prosthesis implanted in a patient. A volunteer with retinitis pigmentosa and no residual vision was chronically implanted with an optic nerve electrode connected to an implanted neurostimulator and antenna. An external controller with telemetry was used for electrical activation of the nerve that resulted in phosphene perception. Open-loop stimulation allowed the collection of phosphene attributes and the ability to elicit perception of simple geometrical patterns. Low perception thresholds allowed for large current intensity range within safety limits. In a closed-loop paradigm, the volunteer was using a head-worn video camera to explore a projection screen. The volunteer underwent performance evaluation during the course of a training program with 45 simple patterns. Multiple bars (each 320 × 22 mm when projected on a screen) were combined to form letters on a 1 × 1 m screen, with the patient at 0.5 m from the screen. After learning, the volunteer reached a recognition score of 63% with a processing time of 60 seconds. The results were encouraging in that the blind volunteer was able to adequately interact with the environment while demonstrating pattern recognition and a learning effect for processing time and orientation discrimination.54
Retinal prostheses
Pathology of retinitis pigmentosa and selected macular disorders
Whereas the above prostheses are potentially useful for patients who have compromised visual pathways posterior to the retina, a microelectronic retinal implant is suitable for cases in which the patient is affected by an outer retinopathy as with RP or AMD. Potts and Inoue, some 40 years ago, demonstrated the ability to evoke an electrically elicited response (EER) via ocular stimulation using a contact lens as a stimulating electrode.55–57 This discovery was expounded upon by Knighton, who demonstrated that inner retinal layers could be electrically stimulated and would elicit an EER.58,59
For a retinal prosthesis to function properly, the retina must not be affected by disease to the point where not enough viable cells remain to initiate a neural signal. Postmortem morphometric analysis of the retina of patients with end-stage RP has revealed that 78.4% of inner nuclear and 29.7% of ganglion layer cells were retained compared to only 4.9% of photoreceptors.6,60–63 Also, 93% of RGC were spared and an increase in inner nuclear layer cells (by 10%) were noted in legally blind neovascular AMD patients.64,65 Furthermore, no statistical significance was noted between non-neovascular eyes with geographic atrophy and age-matched controls.64,65 This demonstrates limited transsynaptic neuronal degeneration in the aforementioned retinopathies, and as such, it is theoretically possible to electrically stimulate the remaining retinal neurons to elicit useful visual perceptions. It is important to understand the stages of outer retinal degeneration and the associated anatomical and physiological changes that occur. A comprehensive study has been done by Marc, in which three phases of degeneration and remodeling are classified.66 In the first two phases, photoreceptor stress and death and associated loss of tropic transport are observed. Both bipolar and horizontal cells can actually retract dendrites, while the latter can sprout axonal and dendritic processes that can reach the inner plexiform layer. Müller cells can form a dense fibrotic layer and seal off the subretinal space, electrically isolating implants placed there via the choroid. In phase 3, the number of viable cells of all classes is depleted. Bipolar and amacrine cells can migrate up to the ganglion cell layer and undergo neural rewiring.
Such anatomical changes manifest physiologically. Using a patch clamp technique in a degenerate mouse model, it has been shown that rod bipolar cells lose their sensitivity to the excitatory neurotransmitter glutamate while they increase their response to the inhibitory horizontal cell neurotransmitter GABA.67 Thus, the retinal circuitry is altered both anatomically and physiologically by degeneration.
In spite of these well-documented changes in the inner retina after photoreceptor loss, numerous studies have established the safety and efficacy of electrical stimulation of the retina. Early studies by Humayun and colleagues61–64 established the feasibility of electrical stimulation of the retina. In an operating room setting, hand-held electrodes were inserted into the eye of blind test subjects. The test subjects reported the appearance of small spots of light when the electrodes were activated. The apparent location of the spot of light in general corresponded with the retinal area stimulated. Similar experiments were repeated by other groups.68,69 While these experiments only allowed a few hours of testing in each subject, the critical findings led to the development of chronic implant system.
ARGUS I
At 2 weeks later, under general anesthesia, the implant is placed in a recess well created in the temporal skull, the same way it is done for the cochlear implants.70
To secure and protect the cable, a shallow groove is created along the temporal skull. The cable is then placed in the groove and delivered through a lateral canthotomy into the periocular space. Next, the cable and electrode array are implanted under the four rectus muscles. A complete pars plana vitrectomy is performed and the array introduced to the eye through a 5-mm circumferential scleral incision placed 3 mm posterior to the limbus. The array is placed temporal to the fovea and a single retinal tack inserted to secure the array in place.71 A clinical trial of the ARGUS I device began in 2002 and enrolled six RP patients.
Subjects were able to discriminate between different percepts, identify everyday objects such as a knife, a plate or a cup, and detect the direction of motion. Perceptual thresholds were within safe limits and were stable over time.72,73 Perceptual thresholds correlated with separation (i.e., lift-off) between the electrode array and the retina.73,74 In addition, increasing frequency of pulses lowered the charge per pulse in a predictable way.75
The best visual acuity using the ARGUS I was the maximum allowable by the spacing of electrodes on the array (i.e., 20/4000), but this was only demonstrated in one test subject.76 Adverse events included erosion of the conjunctiva over the cable at the sclerotomy and detachment of one array after one subject incurred blunt ocular trauma (subsequent re-tacking was successful).
ARGUS II
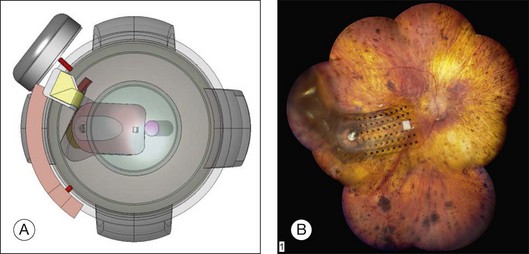
The ARGUS II System (Fig. 126.1) uses an external camera system very similar to ARGUS I, but the implanted part of the device is completely different. The ARGUS II System comprises an encircling band (sclera buckle), an inductive coil and a case containing the electronic components attached to the band, and an integrated ribbon cable and electrode array. The electrode array spans 20° of visual field corner-to-corner. All components fit inside the orbit. The dimensions of the ARGUS II are as follows: electronics case: height, 3.2 mm, diameter, 10.29 mm; receiving coil: height, 16.33 mm, width, 9.7 mm; wire diameter, 0.25 mm; two layers of winding; electrode array 5.5 mm wide and 6 mm long; electrode cable: length, 53.1 mm; width, 1.9 mm.
The ARGUS II is being evaluated in a single-arm, prospective, multicenter clinical trial. A total of 30 subjects were enrolled between June 2007 and August 2009 at 10 clinical centers.77 The electrode array extends across 20° of visual field, measured from corner to corner. All subjects were able to perceive light during electrical stimulation. Experiments documented improvement in object localization. Using a target of a 7 cm white square on a black LCD screen at 30 cm distance, 27 out of 28 subjects (96%) performed better in localizing the object with System ON versus OFF, and no subjects performed significantly better with the System OFF.75 Motion detection was also improved, but to a lesser extent as this is a more difficult task. Using a target of a white bar moving across a black LCD screen, 16 out of 28 subjects (57%) performed this test better with the System ON versus OFF.77 Some subjects report the perception of color, which can be reliably produced under certain conditions.
All subjects’ acuity was measured at worse than 2.9 logMAR in both eyes before implantation. To date, none of the subjects have been able to reliably score on the visual acuity scale in either eye with the System OFF. Seven subjects have been able to reliably score on the scale with the System ON in at least one follow-up time point. The best result to date is 1.8 logMAR (equivalent to Snellen 20/1262).77
Letter reading was tested in 22/30 subjects. Six of these subjects were able to identify any letter of the alphabet at a 63.5% success rate (versus 9.5% with the system off). In all 22 subjects, a small set of eight letters was identified 72.5% correctly, versus 16.8% with the system off. Subjects were free to take as much time as needed to make a judgment. Subjects provided answers after 100 seconds in the full alphabet and 44 seconds in the limited letter set.78 Some subjects were able to put the letters together into words and read sentences.79
Most subjects had no serious adverse events (SAE) and none had any unanticipated adverse events. Of the 30 subjects, 21 subjects (70%) had no SAEs. Three subjects experienced conjunctival erosion due to the extraocular device, this being the most common SAE. All but one was successfully repaired. One device needed to be explanted and this was accomplished without any complications. Other SAEs included three cases of endophthalmitis; each was treated with intravitreal antibiotics and the devices were not explanted and remain functional. There were three cases of hypotony that were resolved with surgical intervention. There was one intraoperative tear treated successfully during surgery with laser retinopexy and two retinal detachments that required subsequent surgery to reattach the retina.80
Intelligent Medical Implants (IMI, Bonn) is developing an active epiretinal prosthesis. The electronics are located in the same location as the ARGUS II implant.81 The device has 49 electrodes and fits entirely inside the orbit. Inductive coupling is used for power and an optical link is used for data. The IMI group has published its surgical approach in animals as well as acute testing procedures and results.
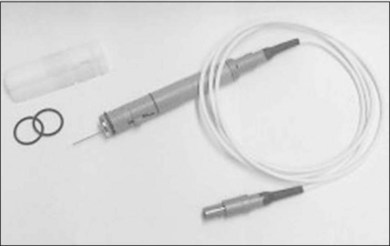
The acute testing surgery was performed under subconjunctival anesthesia. The four eye muscles were fixed, and a pars plana vitrectomy with complete removal of the vitreous and posterior hyaloid were performed. Afterwards, the electrodes were introduced into the eye using one of the sclerotomies with the MESE 12 system (Fig. 126.2). The MESE 12 system is a hand-held surgical instrument for controlled positioning of a microcontact film (BSA) on the surface of the retina. The system must be held by the surgeon during the entire procedure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree