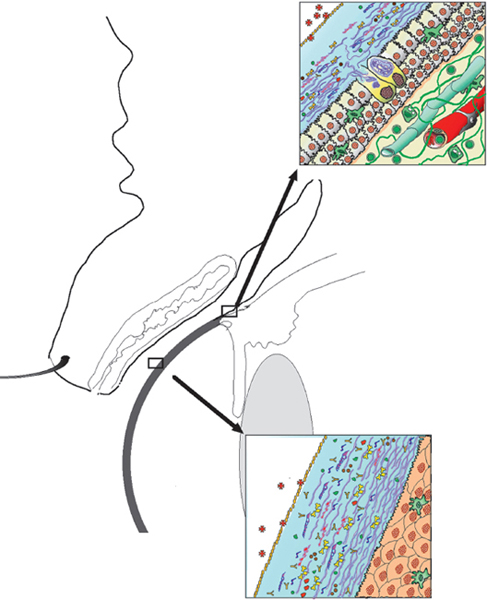
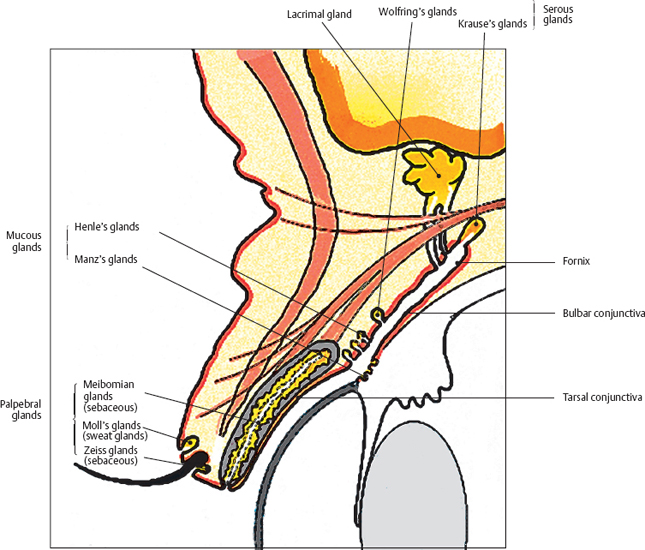
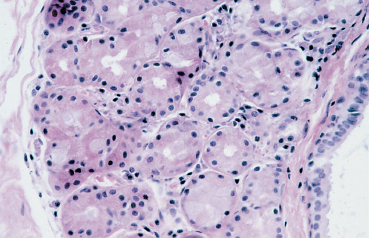
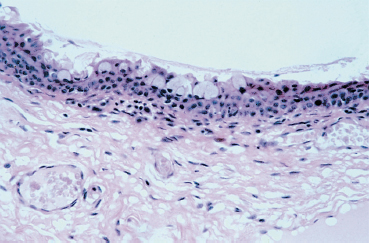
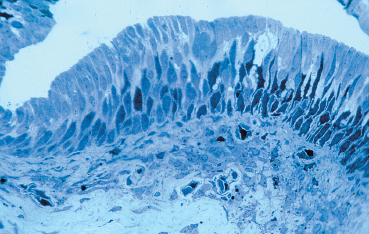
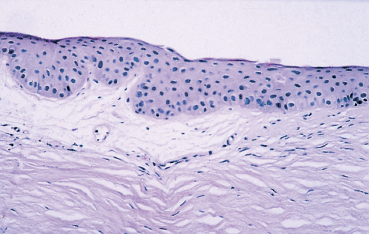
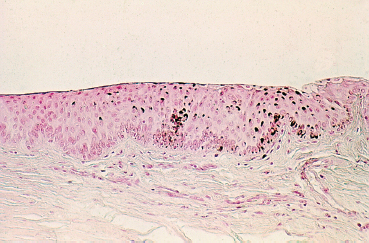
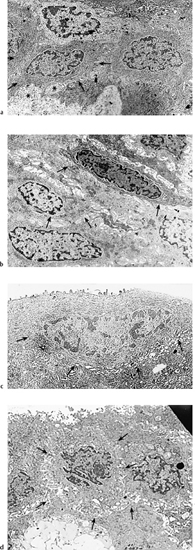
Chapter 1 Anatomy Introduction This chapter is a thorough analysis of conjunctival anatomy, zooming in from the lowest to the highest magnification. The conjunctiva is a vascularized mucous membrane that covers the anterior surface of the eyeball and the posterior surface of the upper and lower eyelids. It is responsible for secreting the mucus required for tear film stability and corneal transparency. It also plays an important defensive role. It is richly vascularized and contains many immunocompetent cells capable of initiating and mediating inflammatory reactions, and of synthesizing immunoglobulin. The morphological characteristics (microvilli) and biochemical properties (enzyme activity) of conjunctival epithelial cells means that they can phagocytose foreign particles such as viruses. Anatomy The Palpebral Conjunctiva In continuity with the skin, the palpebral conjunctiva consists of three parts (Fig. 1.2): —The mucocutaneous junction is located behind the row of meibomian gland openings. It is the transition zone between the stratified keratinizing squamous epithelium of the lid margin and the stratified nonkeratinizing squamous epithelium of the conjunctiva; —The tarsal conjunctiva is strongly adherent to the tarsus; —The orbital conjunctiva extends from the tarsal plate to the fornix. The Conjunctival Cul-de-sac, or Fornix This is the transition zone between the palpebral and bulbar conjunctivas. —The superior fornix, which is the deepest, is situated at the level of the orbital rim, about 8-10 mm from the limbus: Fig. 1.1 Diagrammatic representation of lid anatomy Fig. 1.2 The lid margin represents the transition zone between the epidermis and the conjunctival epithelium. The meibomian glands are located in the tarsus (light microscopy, HES x 25) Fig. 1.3 The structure of the serous glands is identical to that of the lacrimal glands. The serous glands are composed of acini formed by cylindrical secretory cells (light microscopy, HES x 400) —The inferior fornix is 8 mm from the limbus; —The lateral fornix is 14 mm from the limbus; —On the medial side, there is no fornix, which is replaced by the caruncle and the plica semilunaris (a rudimentary third lid). The Bulbar Conjunctiva Thin and transparent, the bulbar conjunctiva consists of two parts: —The scleral part extends from the conjunctival fornix to the limbus. It is readily cleavable, because it is separated from the Tenon capsule by subconjunctival connective tissue. The conjunctiva, the sclera, and the Tenon’s capsule are firmly attached approximately 3 mm from the limbus, and the conjunctiva is more difficult to mobilize; —The limbal part forms a ring about 3 mm wide at the junction between the conjunctival and corneal epithelia. The Conjunctival Glands2,3 (Fig. 1.1) These participate in tear film secretion. There are two groups of accessory conjunctival lacrimal glands, which differ according to the type of secretion (serous or mucous). The superficial lipid layer of the tear film is secreted by palpebral sebaceous glands known as meibomian (Fig. 1.2) and Zeiss glands. Serous Glands (Fig. 1.3) —Krause’s glands are located in the deep conjunctival tissue of the superior fornix. There are about 40 such glands in the superior fornix and six to eight in the inferior fornix. Their histopathological aspect is identical to that of the orbital lacrimal glands; —Wolfring’s glands: between two and five Wolfring glands are found in the upper lid (along the upper edge of the tarsus), and another two are present along the lower edge of the inferior tarsus. Their excretory ducts are lined by cubic basal cells identical to those of the conjunctival epithelium. Mucous Glands Mucus is secreted by goblet cells present in the conjunctival epithelium and by structures with a glandular architecture (Henle’s glands and Manz’s glands). —Henle’s glands, or crypts, are situated on the upper edge of the superior tarsus. They correspond to epithelial invaginations within the chorion and are composed of goblet cells; —Manz’s glands, present in the limbus of many animal species, are absent in humans.2 Histology and Ultrastructure General Architecture The conjunctival epithelium is composed of between two and 8-10 layers of cells, according to the precise location. As in all mucous membranes, the epithelium comprises a single layer of basal cells, a variable number of layers of intermediate cells, and superficial cells of variable shape. The flattening of the superficial cells appears to be an adaptation to mechanical pressure. Besides epithelial cells, the epithelium contains melanocytes with a morphology identical to that of cutaneous melanocytes (Fig. 1.4). They are located among the basals cells of the epithelium. Immunocom-petent cells (especially Langerhans cells) are also present within the epithelium. Their function is discussed in detail in Chapter 2. The entity referred to as the conjunctival epithelium in fact has four distinct morphological components. —The epithelium of the eyelid margin is a transition zone between the epidermis (keratinizing stratified squamous epithelium) and a nonkeratinizing stratified squamous epithelium with flattened superficial cells (Fig. 1.2); —The conjunctival epithelium of the tarsal and bulbar conjunctivas is a stratified epithelium with more or less cylindrical superficial cells. In these two sites the epithelium gradually changes to resemble the epithelium of the fornix (Fig. 1.5); —The conjunctival epithelium of the fornix is a stratified cylindrical epithelium (Fig. 1.6); —The limbus is a particular transition zone between the conjunctival and corneal epithelia. It is covered by a stratified epithelium with flattened superficial cells (Fig. 1.7). Fig. 1.4 Limbal conjunctival epithelium; note the melanocytes in the basal cell layer (light microscopy, Fontana-Masson staining x 400) Fig. 1.5 Bulbar conjunctiva; the epithelial cells become cylindrical when they reach the surface. Note the abundant goblet cells (light microscopy, HES x 25) Fig. 1.6 Forniceal conjunctiva; the superficial epithelial cells are cylindrical (light microscopy, semi-thin section, toluidine blue x 400) Fig. 1.7 Limbal conjunctiva; the superficial cells have a squamous appearance; the chorion is thin (light microscopy, HES x 250) The basement membrane zone (BMZ) separates the epithelium from the chorion or conjunctival stroma. Besides its key role in epithelial adhesion, it appears to have major antigenic potency, as reflected by its involvement in many autoimmune diseases (see Chapter 4). Finally, the chorion, in addition to its collagen framework, possesses abundant vessels and immunocompetent cells, explaining the rapid and violent inflammatory reactions that can occur there. The cellular actors in these immunological responses and their corresponding cytokines will be examined in Chapter 2. Epithelium Basal Cells (Fig. 1.8a) Basal cells have a cuboidal shape and form a single layer separated from the chorion by the BMZ, to which they are connected by hemidesmosomes and adhesion complexes. The lateral cell membranes possess abundant extensions into the intercellular spaces and are connected to one another by desmosomes. The cytoplasm of these cells contains bundles of intersecting tonofilaments corresponding to keratin fibers (Focus Intermediate Cells (Fig. 1.8b) This cell layer is only present in areas where the conjunctival epithelium is thickest (especially the limbus and fornix). The cells are interconnected by desmosomes, which are themselves connected to intracytoplasmic tonofilaments. Their cytoplasm contains intermediary filaments that measure 10 µm and are grouped together in bundles thinner than those found in basal cells. As in basal cells, the organelles are present in the clear perinuclear space and beneath the cytoplasmic membrane. The organelles include mitochondria, a rough ergastoplasmic reticulum, many polysomes, and, occasionally, a well-developed Golgi apparatus. In the juxtacaruncular location, Breitbach and Spitznas4 observed light cells (about 15%) within the layer of spinocytes, with little cytoplasm, no intermediary filaments, and abundant polysomes. Fig. 1.8 The conjunctival epithelium comprises: a Cubic basal cells (arrows) distributed as a monolayer above the basement membrane zone (asterisk) (TEM, x 10 000) b Squamous intermediate cells (arrows); the number of layers varies according to the conjunctival site (TEM, x 10 000) c Squamous superficial cells (arrows) (TEM, x 10 000) d Cylindrical superficial cells (arrows) (TEM, x 10 000) Fig. 1.9 The apical membrane of superficial cells possesses a multitude of microvilli (asterisks) which are covered by the tear film (arrows) (TEM, x 35 000) Superficial cells correspond to all those cells whose apical pole is in contact with the tear film. Superficial epithelial cells take i on a variety of shapes: flattened at the limbus, cubical at the tarsus, and prismatic at the fornix (Figs. 1.8c and 1.8d). Cell Membranes Apical membrane The apical membrane of superficial cells is covered with microvilli (Fig. 1.9). They measure 0.5 µm in diameter and 0.5-1 µm in height, and are themselves covered by the glycocalyx directly in contact with the tear film (Focus The apical pole of goblet cells looks like a “hole” which, according to the stage of the secretory cycle, is either empty or filled with a mucus plug.7 In the tarsal conjunctiva the crypts are recognizable by their large opening (10-60 µm in diameter), and the microvilli covering their inner surface.7,8 Lateral and basal membranes The lateral cytoplasmic membranes of all cells have impermeable tight junctions at their apical part; these junctions ensure both epithelial coherence and the impermeability of intercellular spaces (Fig. 1.11a). All the cells are interconnected by desmosomes (Fig. 1.11b), toward which cytokeratin fibers converge. Superficial Cell Types The cell type is defined according to its secretory activity and the main cytoplasmic organelles. The distribution of the different cell types varies according to the region of the conjunctiva and with age. Epithelial cell cytoplasm contains intermediate filaments which are structural proteins known as cytokeratins. Cytokeratins are coexpressed in the form of acid and basic “pairs” of different molecular weights that can be detected histopathologically by using specific antibodies. Mucosa-associated lymphoid tissue (MALT)-type ocular epithelia include the conjunctiva and the lacrimal gland. A study of the expression of seven cytokeratins1 showed heterogeneous immunoreactivity that allowed epithelial cells to be distinguished according to the site of the conjunctival biopsy. Applications The susceptibility of MALT epithelia to infection can depend not only on the degree of differentiation of epithelial cells, but also on their receptors for infectious agents. For example, the binding site for the Epstein-Barr virus is the CD3 or CD21 molecule. Levine et al.2 have shown that CD21 expression by MALT epithelial cells depends on their degree of differentiation, and correlates with cytokeratin 3 expression. Cytokeratin immunolabeling can be used to study the cellular cytoskeleton in diseases such as bullous pemphigoid and Sjögren syndrome, in which histopathological examination of conjunctival biopsies shows squamous metaplasia, keratinization, and loss of goblet cells. Cytokeratins can be used as markers to type epithelial cells in culture and to assess their degree of differentiation. References Functional Anatomy The tear film is in direct contact with the conjunctiva. It is the second line of ocular surface defense, after the lids. The total volume of the tear film is 7-9 μL, and the basal secretion rate is 1-2 μL/min. Tear film stability depends on three factors: tear secretion, spread of the tears over the ocular surface, and tear clearance, partly by the lacrimonasal route and partly by evaporation. Tear flow has been estimated at 0.9-1.2 μL/min by means of fluorophotometry, but some authors have found lower values. The tear film was considered to consist typically of three layers of about 7 µm thickness. In this model the mucus layer has an estimated thickness of 1 µm. However, Prydal et al.1,2,3 recently showed that the tear film had a real thickness of 35-40 µm, and was composed mainly of a gel containing mucins, the key element in tear film stability.4 Whatever the true thickness of the tear film, it is now clear that there is no clear-cut barrier between the three layers. On the contrary, the three tear components—lipids, water, and mucus—are intricately mingled. The mucus is diluted in the water and adheres tightly to the superficial epithelial cells, while the lipid outer layer limits tear evaporation. Nevertheless, for the sake of simplicity, we will describe the three components of the tear film separately (Fig. 1.10 and Fig. 9 [Chapter 9]). 1. Lipid Layer The lipid layer is mainly produced by the meibomian glands. Its thickness varies during the day, attaining its maximum on awakening. Its precise composition may differ from one individual to the next. It is composed of triglycerides, free fatty acids, waxes, and esterified cholesterol. Adherence of the tear film to the ocular surface is ensured by lipids known as lipocalins.5 These appear to prevent the penetration of sebaceous lipids from the skin that might disrupt the fragile tear film stability,6 and also hinder evaporation.7 Secretion of meibomian lipids is influenced by several factors: —Mechanical (blinking reflex), —Nervous (as shown after trigeminal nerve sectioning), —Hormonal (stimulatory action of androgens), —Physical (feedback regulation according to surface tension). 2. Aqueous Layer The aqueous layer, which is about 8 µm thick, represents the bulk of the precorneal tear film. It is composed of 98 % water but also contains water-soluble gases, electrolytes, hormones, organic molecules, and proteins (5-8 g/L), together with live and dead desquamated cells.8 In normal conditions, 99% of these proteins are synthesized in the main lacrimal gland. Basal secretion of the aqueous layer is ensured by Krause and Wolfring accessory lacrimal glands, while reflex secretion is provided by the main lacrimal gland.9 The pH varies from 7.14-7.82, and osmotic pressure is 305 mOsm/kg. Tears play a key defensive role against microbial agents through their high lysozyme,10 betalysin, lactoferrin, and antibody contents.11 IgA and IgG are secreted by the lacrimal glands which are rich in plasma cells and lymphocytes. Secretory IgA, synthesized by the plasma cells in the outer part of lacrimal gland acini, is detected in the mucus film and also is a major component of the defenses against viral and bacterial infections. Serum proteins derived from the vascular compartment by filtration represent 1 % of total tear proteins, in the absence of infections. These proteins include albumin, haptoglobin, IgG, IgA, IgM, and IgE, α2-macro-globulins, complement-derived proteins, transferrin, α1-antitrypsin, and β2-microglobulin. Other proteins also can be synthesized by connective tissue, mainly during inflammatory conditions. 3. Mucus Layer The mucus layer is the deepest tear film layer and adheres firmly to the underlying epithelial cells. Its thickness varies from 0.8 µm over the cornea to 1.4 µm over the conjunctiva. It is mainly composed of a mucin gel.2 It is partly admixed with the overlying aqueous layer and thus facilitates the adherence of the latter to the hydrophobic membrane surface of the conjunctival and corneal epithelial cells. The tear film is ruptured and reformed with each blink. When the lids close, the mucus adheres to the epithelial cells by their microvilli. At the same time, contraction of the orbicular muscle flushes the tears toward the canaliculi, which are distended during lid opening, allowing tear drainage. The lipid film secreted by the meibomian glands then spreads over the aqueous layer. The lipid film is absent at the level of the lids, where the aqueous layer is sandwiched between the two mucus layers.12 The tear film is regulated by both the central nervous system (sympathetic and parasympathetic control) and the peripheral nerves (irritation of nerve endings by fumes, smoke, etc.). Each layer of the tear film plays an essential role: the aqueous layer hydrates the cornea and prevents its opacification; the mucus layer allows the aqueous layer to adhere to the epithelial cells, whose surface area is increased by microvilli and invaginations; and the lipid layer plays a stabilizing role (Tables A and B). In rabbits, corneal thickness decreases immediately when the tear film is removed. In contrast, it remains unchanged if the lipid layer is preserved, even when lids remain open. Some epithelial cell abnormalities may be mainly caused by defective synthesis of the glycocalix, leading to increased hydrophobia of the epithelial surface and to increased evaporation and osmolarity.13 Failure of the tear film to adhere to underlying epithelial cells that have lost their microvilli leads to the emergence of “dry spots.”14 Table A Functions of the tear film15 Table B Factors of tear film stability (according to Tseng17) Laboratory Investigations Tests used to examine the tear film are based on direct analysis. Tear collection often requires mechanical instrumental stimulation of secretion, but this can alter tear composition.18 Thus, lacrimal levels of IgG, albumin, and lactotransferrin often dramatically drop after stimulation of the ocular surface.19 This illustrates the importance of distinguishing between basal and reflex tearing.20 Schematically, two techniques are used to collect tears, neither of which is perfect. The first uses a capillary tube, but leads to a dilution of tear components by reflex tearing; the second collects tears with a strip of filter paper. Both methods are highly dependent on the collection conditions, and especially evaporation. Norn applied a simple glass cylinder to the conjunctiva, and considered this the best method for assaying blood, nitrites, albumin, glucose, and pH. In contrast, collection based on Schirmer test, cotton thread, sponge, and microcapillary tubes seems more appropriate for harvesting mucus and desquamated cells. Each tear collection method therefore has its limitations, and a knowledge of these is essential when interpreting assay results, especially in patients with lacrimal insufficiency.21 The various components of the tear film can be assayed: protein electrophoresis and lactoferrin assay reflect the functional capacities of the lacrimal glands; the lysozyme level reflects the defensive capacities of the tear film (the lysozyme concentration in tears is higher than anywhere else in the body); the lipid film can be studied by high-pressure liquid chromatography (HPLC), although the meibometer seems to be more suitable in practice.22 Conjunctival impression cytology is a simple and noninvasive technique which is useful for evaluating the pathological consequences of tear film disorders on the underlying ocular surface epithelium. The value of conjunctival impression cytology is well documented in dry eye syndromes.23,24 References 7. Norn M. Pollution keratocon-junctivitis. Acta Ophthalmol (Copenh), 1992; 70: 269-73. 8. Van Haeringen NJ. Clinical biochemistry of tears. Surv Ophthalmol 1981; 26: 84-96. 17. Tseng SCG. Evaluation of the ocular surface in dry eye conditions. 21. Int Ophthalmol Clin, 1994; 34: 57-69. 21. Norn M. Sampling methods for tear stix tests. Acta Ophthalmol (Copenh) 1992; 70: 754-7. 23. Nelson JD. Impression cytology. Cornea 1988; 7: 71-81.
 1). The latter converge toward dense plaques formed by intracytoplasmic constituents of desmosomes and hemidesmosomes. The cellular organelles (mitochondria and rough endoplasmic reticulum) are mainly situated in a perinuclear space free of tonofilaments. Basal cells are undifferentiated and can divide. Breitbach and Spitznas4 observed about 10% light basal cells in paralimbal and juxtacaruncular sites, the remaining basal cells being described as dark. The cytoplasm of light basal cells is free of tonofilaments and contains many mitochondria grouped together at the apical part of the cell and characterized by a dense matrix and fine crests. These cells are not connected to neighboring cells by desmosomes.
1). The latter converge toward dense plaques formed by intracytoplasmic constituents of desmosomes and hemidesmosomes. The cellular organelles (mitochondria and rough endoplasmic reticulum) are mainly situated in a perinuclear space free of tonofilaments. Basal cells are undifferentiated and can divide. Breitbach and Spitznas4 observed about 10% light basal cells in paralimbal and juxtacaruncular sites, the remaining basal cells being described as dark. The cytoplasm of light basal cells is free of tonofilaments and contains many mitochondria grouped together at the apical part of the cell and characterized by a dense matrix and fine crests. These cells are not connected to neighboring cells by desmosomes.
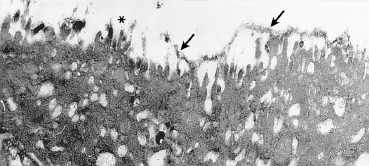
 2) (Fig. 1.10, and Fig. G [Chapter 9]).6 In places, the microvilli coalesce to form microplicae. Scanning electron microscopic examination of the conjunctival surface shows hexagonal cells that are classified light, medium, or dark. Light cells—the most numerous—have few large microvilli, while dark and medium cells have more microvilli which are often compact.7
2) (Fig. 1.10, and Fig. G [Chapter 9]).6 In places, the microvilli coalesce to form microplicae. Scanning electron microscopic examination of the conjunctival surface shows hexagonal cells that are classified light, medium, or dark. Light cells—the most numerous—have few large microvilli, while dark and medium cells have more microvilli which are often compact.7
 Conjunctival Epithelial Cytokeratins
Conjunctival Epithelial Cytokeratins

—To protect the cornea from desiccation;
—To maintain the refractive power of the cornea by smoothing its surface for refraction of incoming rays;
—To protect against infections16;
—To allow oxygen to penetrate into the cornea;
—To avoid corneal dehydration due to hyperosmolarity.
—Preocular tear film
1: Constitutive factors
a Lipid layer produced by meibomian glands;
b Aqueous layer produced by lacrimal glands;
c Mucus layer mainly produced by goblet cells.
2: Hydrodynamic factors
a Corneal sensitivity (controls factors 1b, 2b, and 2c);
b Tear spreading by blinking;
c Tear clearance during blinking.
—Ocular surface epithelium
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


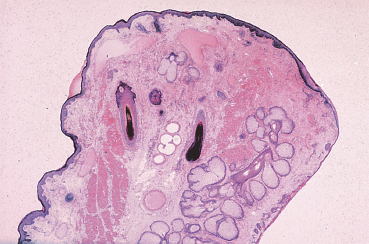
 The Tear Film
The Tear Film