Chapter 3 Allergic Conjunctivitis In 1906 Von Pirquet described “allergy” as host reactivity to a previously encountered antigen, and in 1921 Prausnitz and Kustner showed that a serum factor (IgE) was responsible for passive transfer of the allergic reaction to a normal recipient. Our understanding of the pathogenic mechanisms underlying allergies has greatly advanced. In contrast, progress in the treatment of allergies, especially those affecting the eyes, is somewhat disappointing. The reasons are multifactorial. It is known, for example, that the incidence of allergy is 50% and 70% respectively, when one or both parents are affected, and that nothing can presently be done about this hereditary component. Other factors are environmental changes and migration, both of which generate exposure to increasingly numerous potential allergens which cannot always be avoided. Clinical diagnosis of allergic conjunctivitis can be difficult. The condition can be missed or overdiagnosed because of the frequently intricate links with other causes of ocular inflammation, such as qualitative changes in the tear film and drug toxicity. Allergy tests are not sufficiently sensitive to formally incriminate an allergen in the onset of ocular disorders. Finally, symptomatic treatments are often disappointing and can have adverse effects. The term allergic conjunctivitis covers several entities that differ not only by their clinical expression and prognosis, but also by their pathogenic mechanisms and, consequently, their treatment. The aim of this chapter is to help the reader to diagnose the different forms of allergy, to make best use of appropriate clinical and laboratory tests, and to choose the most appropriate therapeutic strategy. Ocular manifestations of allergy, and especially conjunctivitis, are highly frequent, accounting for 5% of visits to allergy specialists. While sometimes isolated, they usually occur in subjects with a history of allergic rhinitis, asthma, or eczema. The immunological mechanisms underlying ocular allergy are a variable mixture of immediate and delayed-type hypersensitivity. The functional symptoms of allergic conjunctivitis are often multiple, and the degree of morbidity depends on the clinical form. Although some cases of allergic conjunctivitis share certain features, five separate forms are generally recognized on the basis of their clinical manifestations, course, and prognosis (Table 3.1): —Hay fever conjunctivitis (or seasonal conjunctivitis) —Perennial conjunctivitis —Giant papillary conjunctivitis (GPC) —Vernal conjunctivitis —Atopic keratoconjunctivitis (AKC) The latter two clinical forms are usually complicated by corneal involvement and can be sight-threatening. The frontier between these different types of allergic conjunctivitis is not always clear-cut, some patients developing several different forms either simultaneously or consecutively. Hay Fever Conjunctivitis Epidemiology Hay fever conjunctivitis is the most frequent clinical form of ocular allergy. The allergens involved depend on the geographical area. Clinical Features Ocular symptoms occur during exposure to the allergen, often in spring, and consist of itching, a tingling sensation, a burning sensation, tearing, and photophobia. Hot conditions and eye dryness aggravate the functional discomfort. Although not specific to ocular allergy, itching strongly points to this diagnosis.1 Hay fever conjunctivitis is usually bilateral. It manifests with papillary hypertrophy at the level of the tarsal conjunctiva (Fig. 3.1), sometimes associated with chemosis (Fig. 3.2), or violaceous palpebral edema. Corneal involvement is moderate, often being limited to punctate epithelial keratitis. Differential Diagnosis The main differential diagnosis is acute papillary conjunctivitis (generally infectious). Pathogenesis Hay fever conjunctivitis results from a type I hypersensitivity mechanism. Perennial Allergic Conjunctivitis Epidemiology Symptoms are present all year round, although some patients have a seasonal exacerbation. The allergens are mites, animal danders, cotton, and feathers (Table 3.2). Clinical Features Ocular symptoms are usually moderate. Physical examination sometimes shows conjunctival hyperemia, papillary conjunctivitis with a few follicles, transient lid edema, or conjunctival atrophy. Unfortunately, none of these signs is specific and the examination can be normal. Differential Diagnosis The diagnosis of perennial allergic conjunctivitis is sometimes difficult to establish and to distinguish from other disorders of the ocular surface. Thus, ocular dryness is often associated with allergy, increasing the symptoms and favoring local hypersensitivity by increasing the concentration of the allergen in tears.2 Differential diagnosis also may include accommodative insufficiency in patients with chronic symptoms. Table 3.1 Clinical aspects of ocular allergy
| SAC/PAC | VKC | AKC | GPC | |
|---|---|---|---|---|
| Pathogenesis | Type I HS | Types I and IV HS | Types I and IV HS | Mechanical Types I and IV HS |
| Patient type | Allergic | Child Allergic | 30- to 50-year-old man Atopic dermatitis | Contact lenses Ocular prostheses Ocular surgery |
| Lids | Lid eczema Meibomitis | |||
| Clinical signs: Conjunctiva | Papillary conjunctivitis | Cobblestones upper tarsus | Papillary conjunctivitis Conjunctival fibrosis | Papillary conjunctivitis Giant papillae upper tarsus |
| Cornea | Epithelial punctate keratitis | Trantas’ dots, epithelial punctate keratitis, vernal ulcer, vernal plaque | Epithelial defects, ulcerations, scars, neovessels | Corneal involvement rare |
SAC: seasonal allergic conjunctivitis; PAC: perennial allergic conjunctivitis;
VKC: vernal keratoconjunctivitis; AKC: atopic keratoconjunctivitis;
GPC: giant papillary conjunctivitis; HS: hypersensitivity
| Seasonal Allergic Conjunctivitis | Perennial Allergic Conjunctivitis |
|---|---|
| Tree pollens (early spring) | House dust mites |
| Graminaceae | Indoor molds |
| Grasses (May-July) | Plant dust |
| Weed pollen (August-October) | Animal dander |
| Outer molds | Cockroaches Occupational (hairdressers, bakers, etc.) |
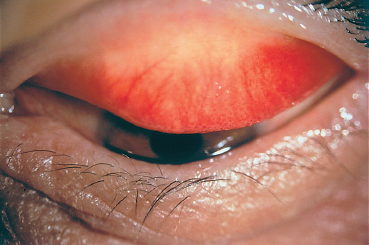
Fig. 3.1 Seasonal allergic conjunctivitis: papillary hyperplasia and hypertrophy
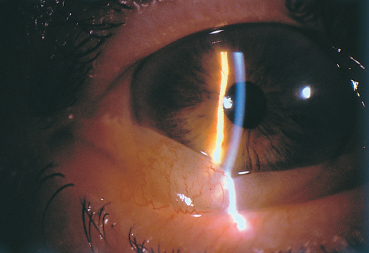
Fig. 3.2 Seasonal allergic conjunctivitis: chemosis
Pathogenesis
Perennial allergic conjunctivitis is due to an immediate hyper-sensitivity mechanism.
Vernal Conjunctivitis
Epidemiology
Vernal conjunctivitis is rare. It is most frequent in dry and hot climes, or in areas with polluted air. Symptoms are mainly perennial, with exacerbations in spring and summer (April-August). Vernal conjunctivitis often begins in boys under 10 years of age with a personal or family history of allergy. It usually stabilizes in adulthood but can become chronic or progress to AKC.3 As it very frequently involves the cornea, it would be better to term the condition vernal keratoconjunctivitis (VKC), regardless of the state of the cornea. It leads to a decrease in visual acuity in about 30% of cases, because of either corneal lesions or iatrogenic complications.4
Clinical Features
Vernal keratoconjunctivitis manifests with bilateral ocular pruritus, a foreign body sensation, tearing, and intense photophobia. The secretions are abundant and thick, and sometimes form pseudomembranes. These children have marked functional discomfort, with a risk of behavioral disorders and poor school performance. The parents may need psychological support to overcome their anxiety.
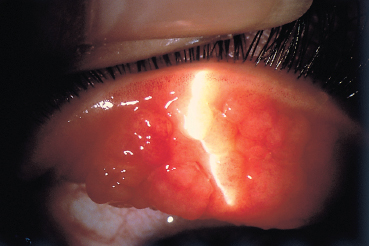
Vernal keratoconjunctivitis is characterized by giant papillae (diameter > 1 mm) on the superior tarsal conjunctiva, giving a cobblestone aspect (Fig. D, p. 51 and Fig. 3.3). The papillae can sometimes lead to palpebral thickening and pseudoptosis. Conjunctival scarring is rare in the absence of cryotherapy.
Highly characteristic vernal limbal involvement may be found concomitantly with palpebral involvement. Pure limbal forms are more frequent in darkly-pigmented patients.5 The limbal conjunctiva is thickened and forms a translucent gelatinous ring containing white excressences (Horner-Trantas dots) (Fig. 3.4) composed of eosinophil aggregates.6
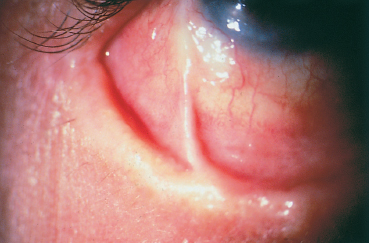
While corneal lesions were found in only 3 % of severe forms in one study,7 they were present in 50% of palpebral forms studied by other authors.8 Corneal lesions are more frequent in palpebral forms of VKC,5 but they are less often due to the mechanical effect of giant papillae on the cornea (albeit an aggravating factor9) than to the toxicity of inflammatory mediators (including eosinophilic major basic protein [EMBP]) released by mast cells and eosinophils.10,11 The most frequent form of corneal involvement is diffuse epithelial punctate keratitis (Fig. 3.5). A shallow oval epithelial macroerosion (“shield ulcer”) (Fig. 3.6) with slightly raised edges also can be seen. It is usually solitary and located on the upper third of the cornea. It is rarely painful. Cells and mucus are gradually deposited onto the bed of the lesion, leading to the formation of a homogeneous whitish vernal plaque firmly anchored to the underlying corneal stroma (Fig. 3.7). The vernal plaque maintains the inflammatory process and prevents reepithelialization. It rarely induces corneal neovascularization and, once healed, leaves anterior stromal opacities.12 These epithelial lesions are divided into three classes according to their clinical aspect and prognosis (see Table 3.8).13 There is a noteworthy risk of bacterial superinfection and of amblyopia secondary to corneal opacity. Pseudogerontoxon and superior pannus formation are other rare complications of VKC and are associated with superficial new vessels growing from the limbus into the corneal periphery.
Keratoconus and cataract are also frequently associated with VKC. Corneal ectasia may be favored by eyelid friction on the cornea.14
Differential Diagnosis
Vernal keratoconjunctivitis must be distinguished from atopic keratoconjunctivitis, which affects adults and combines palpebral skin lesions, fib rosing conjunctivitis with fornix foreshortening, and corneal neovascularization. However, some cases of VKC in children progress to atopic forms in adulthood.
Giant papillary conjunctivitis shares certain clinical characteristics with vernal conj unctivitis. The diagnosis of GPC is suggested, however, by the presence of giant papillae in adult contact lens wearers (see section on Giant Papillary Conjunctivitis, p. 56 ff).
Fig. 3.3 Vernal conjunctivitis: note the giant papillae on the superior tarsal conjunctiva
The giant papillae of vernal conjunctivitis must be distinguished from follicular conjunctivitis secondary to adenovirus, herpesvirus, or chlamydial infection.
Pathogenesis
Vernal keratoconjunctivitis involves two hypersensitivity mechanisms.
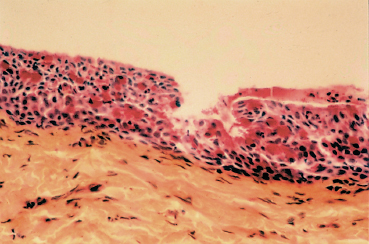
IgE detection in tears15,16 and conjunctival infiltration by B cells, plasma cells, eosinophils, and mast cells (Fig. 3.8) (see Chapter 2, section on Mast Cells, p. 33 and 48 ff)10,17,18 reflect type I immediate hypersensitivity. The conjunctival infiltrate contains 96% of chymase-positive mast cells (CTMC).19
Conjunctival infiltration by dendritic cells, T lymphocytes17 and basophils,18 and microvascular endothelial alterations,20 reflect type IV cell-mediated delayed-type hypersensitivity.
Eosinophils also have a pathogenic role in VKC.10 Serum levels of preformed eosinophil-derived mediators (eosinophilic cationic protein [ECP] and eosinophil-derived neurotoxin/eosinophil protein X [EDN/EPX]) are increased.21 Eosinophilic major basic protein (EMPB) is involved in the formation of corneal epithelial lesions22.
Atopic Keratoconjunctivitis
As atopic conjunctivitis is nearly always accompanied by corneal involvement, the term AKC is used to define the combination of severe chronic keratoconjunctivitis with atopic dermatitis.23 Cutaneous involvement is sometimes associated with rhinitis or asthma and is a diagnostic sign in AKC. Ocular complications, which combine palpebral lesions, fibrosing conjunctivitis, and corneal lesions, can be sight-threatening.
Epidemiology
Atopic keratoconjunctivitis is a rare, chronic, and severe condition. It usually begins in mid-adulthood (30-50 years) and generally affects men. It is reported in 25-40% of patients with atopic dermatitis.24,25
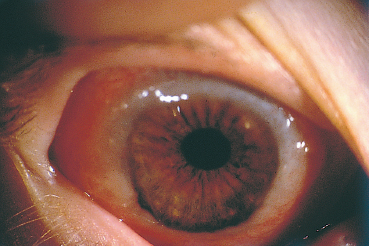
Fig. 3.4 Vernal conjunctivitis in its limbal form
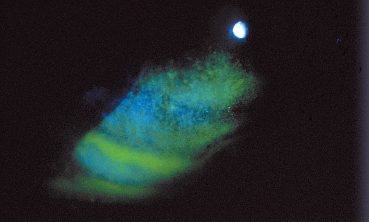
Fig. 3.5 Complication of vernal conjunctivitis: epithelial punctate keratitis
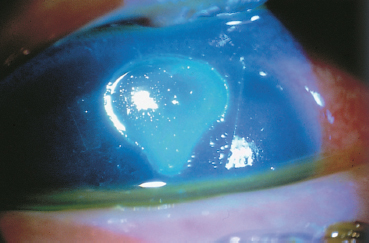
Fig. 3.6 Complication of vernal conjunctivitis: corneal shield ulcer
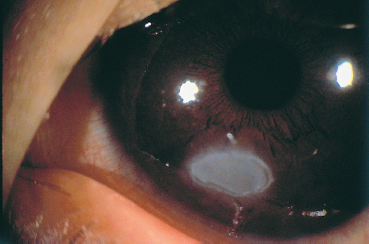
Fig. 3.7 Complication of vernal conjunctivitis: vernal plaque
Fig. 3.8 Vernal conjunctivitis: histological section of giant papillae. Note the presence in the chorion of a dense inflammatory infiltrate and abundant goblet cells stained red with PAS (courtesy of Dr F. D’Hermies, Hâtel Dieu, Paris)
Clinical Features
The symptoms are perennial, with aggravation during the hot season. The condition is bilateral and causes major functional discomfort. It leads to tearing, together with a burning and itching sensation in the eyes. Secretions are serous or thick and are abundant.
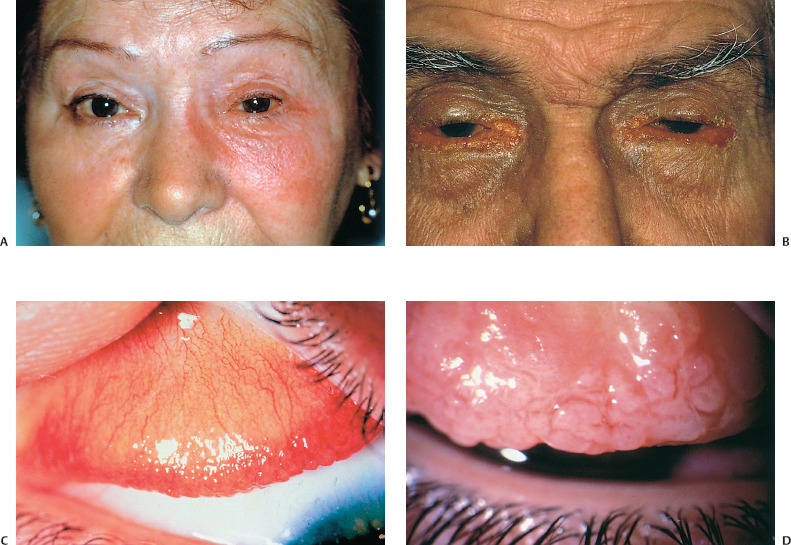
Eczematiform palpebral lesions lead to lichenification and induration of the eyelids, which become thick and lose their lashes (Fig. B, p. 51 and Fig. 3.9). Meibomitis is frequently associated with cutaneous lesions.
The conjunctiva is hyperemic, and the papillary hypertrophy observed in some cases usually affects the lower conjunctival fornices. The chronic conjunctival inflammation leads to conjunctival fibrosis, fornix foreshortening, and symblepharon formation (Fig. 3.10).26 The giant papillae and limbal involvement seen in VKC are rare.26
Corneal involvement is frequent (75% of cases),26,27 reflecting the severity of this sight-threatening condition. It is worsened by the associated ocular dryness and meibomitis. It begins with superficial keratitis and progresses to chronic epithelial ulceration, opacities, neovascularization (65% of cases) (Fig. 3.11 ),28 and corneal thinning that sometimes leads to perforation. The epithelial lesions carry a risk of Staphylo-coccus aureus superinfection, as this species frequently colonizes the eyelid margins in atopic subjects.29 Herpesvirus superinfection is also frequent (1-22% of eyes),26,28 and is often bilateral and extensive. This calls for close monitoring if topical steroid therapy is prescribed. There is also a risk of fungal superinfection if these patients are treated with topical steroids for long periods.30
Keratoconus is associated with a higher frequency of AKC and with atopic dermatitis, which is found in 16% of patients.31 Cataract complicates AKC in 10% of cases.32 It appears in patients with more than a 10-year history of the allergic condition. It is classically anterior subcapsular and shield-shaped, but posterior subcapsular cataracts are frequent and are favored by steroid therapy. High EMBP levels have been found in the aqueous humor of atopic eyes with cataracts.33 Rheg-matogenous retinal detachment has been reported in patients with AKC.34,35 It could be linked to vitreal degeneration or result from excessive eye rubbing due to itching (equivalent to traumatic detachment).35
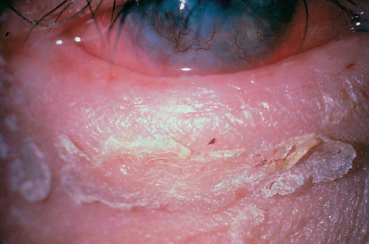
Fig. 3.9 Atopic keratoconjunctivitis: note the lichenified, thickened lid devoid of lashes
Differential Diagnosis
Chronic iatrogenic conjunctivitis, following the use of certain eyedrops, can mimic fibrosing conjunctivitis (see Chapter 8). The diagnosis of AKC is difficult to establish in advanced forms with symblepharons and neovascularized corneal opacities.36 The history and physical examination should seek to identify the slightest feature of allergic manifestations which may have been forgotten by the patient.36 Conjunctival biopsy with immunolabeling will rule out autoimmune fibrosing conjunctivitis (see Chapter 4) and will often show eosinophilic polymor-phonuclear cells in the epithelium and chorion (absent from the normal conjunctiva) and abnormally large numbers of mast cells in the chorion (Fig. 3.12).
Primary chronic meibomitis, like that associated with rosacea, can be complicated by conjunctival fibrosis and corneal inflammatory lesions. The diagnosis of AKC can be missed, especially as this condition is always accompanied by secondary meibomitis (see Chapter 5).
Pathogenesis
Atopic keratoconjunctivitis involves both type I and type IV hypersensitivity reactions.37 Mast cells and eosinophils infiltrate the conjunctiva (Fig. 3.13), and corneal damage is due to the toxicity of ECP,38,39 which contributes to conjunctival fibrosis by inhibiting proteoglycan breakdown.40
Giant Papillary Conjunctivitis
Epidemiology
The diagnosis of GPC should be raised in patients who wear contact lenses or ocular prostheses, and in those with a history of corneal surgery (unburried sutures)41 or surgery for retinal detachment with exposed indentation material.42 Giant papillary conjunctivitis has been reported in eyes with corneal deposits of keratin and calcium,43 band keratopathy,44 or a filtering bleb.44
Most patients are soft contact lens wearers (47.5%) who develop functional discomfort after tolerating the lenses well for an average of 2-3 years.45 Deposits of mucus, cell debris, and microorganisms46 are frequently found on the lenses47 and appear to play a pathogenic role in GPC. Hard contact lens wearers are more rarely affected (21.6 %)48,49 and GPC appears to occur after longer periods (up to 8 years).49 Risk factors for GPC in contact lens wearers are young age45 and poor lens hygiene,48,50 while the tear film break-up time,45 the type of ametropia, and keratometry51 do not seem to be involved.
Fig. 3.10 Atopic keratoconjunctivitis: symblephara
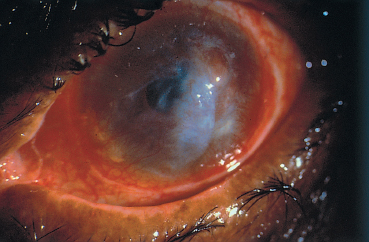
Fig. 3.11 Atopic keratoconjunctivitis: corneal ulcer, opacities, and neovascularization
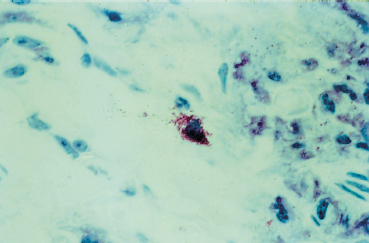
Fig. 3.12 Atopic keratoconjunctivitis: degranulating mast cell (Giemsa staining, original magnification x 60)
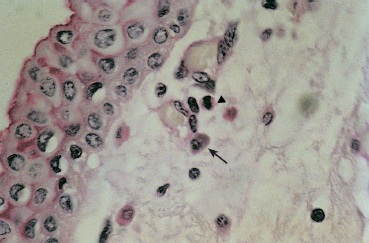
Fig. 3.13 Atopic keratoconjunctivitis: eosinophils (arrowhead) and mast cells (arrow) in the conjunctival chorion (PAS staining, original magnification x 100)
Clinical Features
Patients complain of itching that increases on removing the contact lens, together with blurred vision due to deposits on the lens, a foreign body sensation, or lens intolerance.
The examination initially shows mild papillary hypertrophy, which is followed by large papillae (>0.3mm) (Fig. C, p. 51) and finally giant papillae (> 1 mm) on the superior tarsal conjunctiva. There is also mucosal hypersecretion that leads to excessive lens movement. Contrary to VKC, limbal and corneal involvement are rare.
The prognosis is good, and symptoms resolve, albeit slowly, in 79 % of cases.50 Progression to fibrosis is extremely rare.
Differential Diagnosis
Vernal keratoconjunctivitis is also associated with giant papillae on the superior tarsal conjunctiva but is usually diagnosed in childhood.
Pathogenesis
Giant papillary conjunctivitis is due to a combination of mechanical irritation44 and hypersensitivity phenomena.52
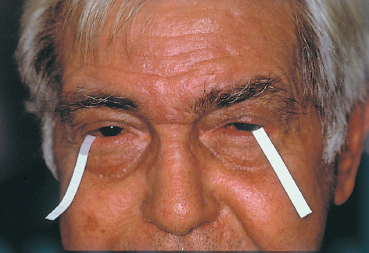
Fig. 3.14 Tear fluid IgE assay using filter paper strips for tear collection
There is usually a history of allergy, although pollen allergy does not seem to be more frequent than in the general population.53 Allergy to lens disinfection solutions (containing thimerosal, for example) is relatively frequent.53
The allergic hypothesis is based on the high levels of IgE and IgG in tears,54 IgM deposits on lenses,52 and conjunctival infiltration by eosinophils.55 The high prevalence of mucosal mast cells (MMC) could explain why most mast cell stabilizers are only relatively effective (see section on Mast Cell Stabilizers, p. 60 ff). The lack of corneal involvement could be linked to a lower degree of conjunctival infiltration by eosinophils than in VKC.55 A significant increase in tear leukotriene C4 levels has been found in patients with contact lens-associated GPC who might benefit from leukotriene inhibitor therapy in future.55a
The lens deposits appear to contain the antigens.54 The reduction in lactoferrin levels in tears56 would favor microbial deposition on the lens.57
Meibomitis has been associated with GPC.51,58 It would appear to be favored by an underlying allergic tendency. It provokes tear film instability but its role is uncertain.
Contact Conjunctivitis
Contact conjunctivitis is frequent. The allergen is usually an eyedrop solution (Table 3.3) or a cosmetic.
Table 3.3 Main drug molecules involved in contact allergy
| Beta-blockers Benzocaine Chlorhexidine Preservatives: benzalkonium chloride, mercury salts Neomycin Phenylephrine |
Contact conjunctivitis is linked to type IV hypersensitivity, and onset usually occurs 48 hours after exposure to the allergen. Itching and tearing are frequent. Eczematiform palpebral lesions (edema, hyperemia, and sometimes vesicles) orient the diagnosis (Fig. A, p. 51). They are associated with papillary or follicular conjunctivitis and sometimes with chemosis. Corneal involvement is rare and is usually limited to punctate epithelial keratitis.
Laboratory Work-up
(see Chapter 8, Laboratory Investigations, p. 137 ff)
IgE Assay in Tears (see Focus  4)
4)
Although semi-quantitative, total IgE assay in tears absorbed onto a nitrocellulose acetate filter paper59 placed in the lower conjunctival fornix is the simplest method, and one that can be used in the office by all ophthalmologists (Fig. 3.14). The only difficulties are with very young children and patients with severe dry eye. The presence of IgE in tears reflects a humoral hypersensitivity mechanism, but the test is neither sensitive nor specific. IgE is found in one third of eyes with chronic conjunctival conditions, and in 40-50% of cases of clinically diagnosed allergic conjunctivitis. The correlation between the presence of IgE in tears and the existence of recurrent hay fever conjunctivitis is therefore fairly weak.60
In practice, the presence of IgE in tears supports the diagnosis of allergy but the absence of IgE does not rule it out.
An immunoblotting method for specific IgE assay in tears has also been described but is not commonly used.61
Skin Tests
Skin tests can contribute to the diagnosis of ocular allergy and help to identify the allergen(s) involved. The choice of test allergens will be oriented by the history and clinical findings, the most frequently tested being animal danders, house dust mites, molds, tree and weed pollens, and grasses.
The prick test is used to identify allergens involved in immediate hypersensitivity phenomena, in which mast cell de-granulation plays a role. It cannot be done if the patient is receiving systemic treatment for allergy or has an extensive skin condition. The reaction is often weak in children and the elderly. The allergenic extract is deposited on the ventral forearm and the skin is pricked with a lancet or intradermal needle, to a depth of no more than 1 mm in the dermis. Controls are also tested (histamine, allergen solvent). An early reaction appears 10-15 minutes after the injection and disappears after a few hours. A papule more than 3 mm in diameter shows a positive reaction. A late reaction (5 hours after the test) can also occur. The sensitivity and specificity of the prick test depend on the allergenic extract used.
Skin testing is often used when an immediate hypersensitivity mechanism is suspected and prick tests are negative. They are very sensitive but give false-positive reactions when a high dose of allergen is used.62,63 Patch tests are used to detect delayed hypersensitivity. The allergen is deposited on the skin of the back or forearm, and the erythematous reaction is read after 48 hours. False-positive results can occur, and are due to irritation by the allergen (no allergic reaction).
Conjunctival Allergen Challenge
Allergen challenges are used to study how the conjunctiva reacts to contact with a potential allergen. They are applied to a noninflamed eye, in the absence of antiallergic treatment for at least 7 days.64 They are relatively noninvasive, and the acute conjunctivitis they induce generally abates within 12-18 hours.64 They can be used to confirm a diagnosis of allergy, to study clinical manifestations and the mediators involved,65,66,67 or to assess the efficacy of antiallergic treatments. Only one allergen can be tested at a time.
This test is sufficiently sensitive and reliable for the diagnosis of allergic conjunctivitis in 42 % of cases.64 The results correlate better with IgE levels in tears (87 %)68 than with the results of skin tests or serum IgE levels (71 %).69 The inflammatory reaction is dose-dependent.66,67 It can be long-lasting (more than 6 hours after the test) after a strong dose of allergen,70 and the reaction can be assessed objectively by means of digital imaging methods.71 In some countries, conjunctival allergen challenge is only used for experimental studies and not for diagnosis.
Impression Cytology (see Chapter 2, Focus 4 and 5)
Impression cytology can be used to analyze the morphology and architecture of cells forming the superficial layer of the conjunctival epithelium. Epithelial cells, goblet cells, and dendritic Langerhans cells can be identified. The study of deeper epithelial layers and the chorion necessitates conjunctival biopsy.
Impression cytology has mainly been used to study cellular alterations in dry eye syndromes.72
Morphological studies can be followed by immunocytological techniques that identify cell surface markers. Identification and quantification of cells expressing the HLA-DR antigen can be used to quantify the local inflammatory reaction. Normally, only dendritic cells express HLA-DR antigen, but inflammation leads to HLA-DR antigen expression by some epithelial cells. A subclinical inflammatory reaction can thus be demonstrated in glaucomatous eyes receiving multiple medications73,74 and in dry eye syndromes refractory to substitutive therapy.74
The detection of causal agents with specific antibodies can be used to establish the diagnosis of ocular chlamydiosis pending routine application of more sensitive gene amplification methods.76
Miscellaneous
Conjunctival scraping is a useful examination, as it can reveal the presence of eosinophils. In one study it was positive in 45 % of cases of hay fever conjunctivitis76a.
Conjunctival biopsy must only be used in exceptional circumstances. It may be indicated in AKC if the diagnosis is in doubt, especially when the presence of conjunctival fibrosis raises the possibility of autoimmune conjunctivitis (see p. 56 ff).
Assay of specific IgE in tears by means of the radioallergosorbent testing (RAST) is possible,76b but this in vitro test is only moderately sensitive.
Tryptase can now be assayed in tears to detect the high concentrations associated with allergic conjunctivitis. Tryptase, however, is only found in the early phases of allergic reactions, coinciding with mast cell degranulation.76c
Interleukin 2 receptor assay in the serum of atopic subjects is not routinely used.77
Interleukin 4 has been detected in cytological conjunctival samples from patients allergic to pollen.78
Therapy
Ocular allergies are recurrent acute, or chronic conditions. They necessitate long-term management combining environmental adaptation, pharmacotherapy, and immunotherapy.
The treatment aim in ocular allergy is to improve clinical tolerance and to avoid long-term sequelae in severe forms. Treatment should be adapted to the severity of the symptoms and prognosis. The drugs used in this setting act on processes involved in humoral and/or cellular hypersensitivity reactions. The risk of iatrogenic complications should not be overlooked, particularly as self-treatment is frequent in chronic allergy.
The efficacy of the different drugs is sometimes difficult to establish, for three main reasons: a placebo effect is found in 70% of cases of ocular allergy; spontaneous remission is possible; and signs and symptoms vary over time.
Environmental Control
When the allergen can be identified through the history or by skin testing, environmental control can be proposed. Eradication of the allergen is a useful measure but is not always feasible. The following simple measures can be recommended:
Mite allergy
—Stays at high altitudes (> 1200 m),
—Lowering room humidity (< 50%), installing air conditioning devices and air filtration systems in heating vents,
—Removing rugs and carpets,
—Scrupulous bedding hygiene,
—Use of antimite products (e.g., benzylbenzoate).
Mold allergy
—Regular airing of rooms,
—Avoidance of damp rooms (cellars, basements, etc.),
—Removal of indoor plants.
Cockroach allergy
—Repeated use of a powerful insecticide in the kitchen (and all kitchens in the building), especially when there is a garbage chute inside the apartment.
Weed allergy
—Avoid stays in the countryside during pollen-shedding periods,
—Avoid contact with lawns.
When the allergen cannot be eradicated, it may be possible to change its structure. For example, the use of tannic acid in sites harboring mites reduces the capacity of these allergens to induce hypersensitivity reactions in sensitized subjects.79
Symptomatic Measures
Regular use of preservative-free normal saline is recommended in all forms of ocular allergy. The aim is to wash out the conjunctival fornices, neutralize tear pH, and dilute antigen present in tears. It also has a noteworthy placebo effect.
Application of cold compresses to the eyelids can temporarily relieve symptoms.
Drug Therapy
Use of eyedrops containing preservatives should be avoided because there is a risk of conjunctival toxicity and sensitization. Increasingly, eyedrops are being sold in single-dose preparations.
Topical Vasoconstrictors
Topical vasoconstrictors (naphazoline, phenylephrine, oxy-metazoline, and tetrahydrozoline) can improve tolerance and reduce ocular hyperemia but must not be used excessively. They comprise sympathomimetics, alone or combined with antiseptics or an antihistamine (Table 3.4).
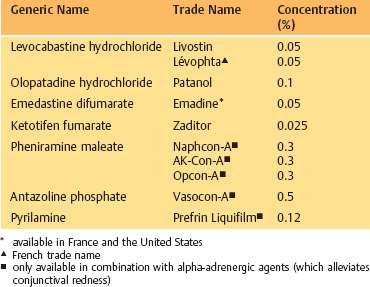
Table 3.4 Topical ocular antihistamines
Antihistamines (Table 3.4)
Antihistamines reversibly block the histamine receptors present on the surface of the eye. They compete with endogenous histamine, without interfering with its synthesis or release (Fig. 3.15).
Two types of histamine receptor have been identified on the ocular surface. Stimulation of type 1 receptors provokes vasodilation, increases vascular permeability, and is responsible for the itching sensation. At the cellular and molecular levels, it induces conjunctival fibroblast proliferation and migration,79a substance P and calcitonin release, and cytokine production (IL-6 and IL-8) by epithelial cells.79b Stimulation of type 2 receptors provokes vasodilation and conjunctival hyperemia.
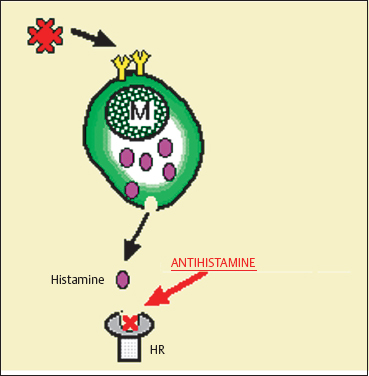
Fig. 3.15 Mode of action of antihistamines M: mast cell; HR: histamine receptor
Type 2 antihistamines do not seem to be indicated in the treatment of allergic conjunctivitis, even though they potentiate the action of type 1 antihistamines in some experimental models.80
Type 1 antihistamines can be administered orally or topically. The oral route (nonsedative drugs: astemizole, terfenadine, loratadine, and cetirizine) is effective on both ocular and extraocular symptoms, and permit milder topical treatment which, when used for long periods, is associated with conjunctival toxicity and/or sensitization. However, the response to systemic antihistamines is sometimes partial and there is a risk of adverse effects. For example, some such compounds have cholinergic and sedative actions, and there is also a risk of cardiac arrhythmias. In contrast, the sedative action can be beneficial, especially in children, if treatment is administered in the evening.
Pheniramine, pyrilamine, and antazoline are classical antihistamines which have been used for many years. They reduce chemosis and itching associated with allergic conjunctivitis. Topically, they are only available in combination with vasoconstrictors (Table 3.4).
Levocabastine 0.05%, a cyclohexylpiperidine derivative, is a topical antihistamine specific for type 1 receptors. It reduces the expression of adhesion molecules (intercellular adhesion molecule [ICAM-1]) at the surface of the conjunctival epithelium in vivo.80a It is rapidly effective (within about 10 minutes), its action lasts at least 4 hours,81 and it is well tolerated. A single instillation improves both the symptoms (itching and tearing) and the clinical signs (hyperemia, chemosis, and lid edema) that occur after conjunctival allergen challenges.81,82,83
Emedastine ophthalmic solution 0.1 % is a potent histamine H1 antagonist that is superior to ketorolac and levocabastine in controlling itching and redness in allergic conjunctivitis. 83a,83b,83c,83d
Olopatadine is a clinically effective dualaction topical ophthalmic antiallergic agent with selective H1 antihistaminic and mast cell stabilizing properties. It promotes decreased H1 receptor binding activity in vitro and functional H1 receptor antagonism in vivo,83e and inhibits mast cell histamine, tryptase, PGD2, and TNF alpha release in vitro.83f,83g Olopatadine hydrochloride 0.1 % ophthalmic solution is more effective and more comfortable than ketorolac ophthalmic solution (0.5%) in a clinical model of acute allergic conjunctivitis.83h
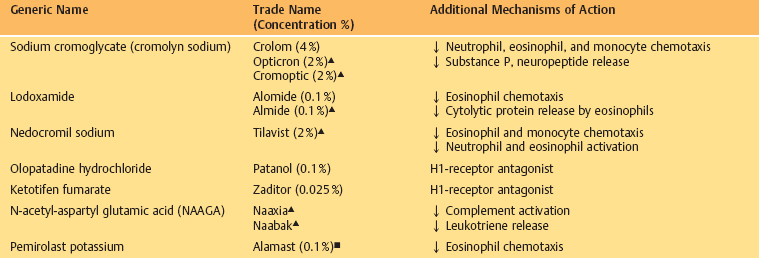
Mast Cell Stabilizers (Table 3.5)
Mast cell stabilizers are administered in the form of eyedrops because their gastrointestinal absorption is poor. Some are preservative-free. The products currently available are sodium cromoglycate, lodoxamide, N-acetyl-aspartyl-glutamic acid (NAAGA), nedocromil, ketotifen, and olopatadine.
Mast cell stabilizers act by inhibiting calcium channels in the mast cell membrane. They prevent the release of inflammatory mediators induced by allergen binding to IgE on the mast cell membrane (Fig. 3.16). Except for olopatadine they are all active on connective tissue mast cells (CTMC), but do not prevent de-granulation of mucosal mast cells (MMC), the latter being T cell dependent.83i The antidegranulating action varies according to the drug, and seems to be some 2500 times more potent with lodoxamide than with cromoglycate in animals.84 Additional mechanisms of action have been described with some of these compounds (Table 3.5), such as a reduction in the chemotaxis of polymorphonuclear neutrophils (cromoglycate), eosinophils (cromoglycate, lodoxamide, nedocromil, and pemi-rolast)84a,85 and monocytes (cromoglycate and nedocromil),86 a reduction in cytolytic protein release by eosinophils (lodoxamide), an inhibition of complement activation (NAAGA) and of the release of substance P, neuropeptides (cromoglycate),86 and leukotrienes (NAAGA, lodoxamide, and pimerolast),84a,86a and a selective H1 antihistaminic activity (olopatadine, ketotifen, and pemirolast).83e,83f,83g,86b,84a Nedocromil has a direct antiinflammatory action and inhibits polymorphonuclear eosinophil and neutrophil activation.86
Table 3.5 Topical ocular mast cell stabilizers
Mast cell stabilizers are more effective when administered before contact with the allergen.87
Topical Corticosteroids (Table 3.6)
Steroids act mainly on late hypersensitivity phenomena and do not inhibit the release of preformed mast cell mediators in vitro. They hinder inflammatory cell chemotaxis and reduce the number of eosinophils and mast cells in the conjunctiva. They inhibit phospholipase A2, thereby countering the production of arachidonic acid and its metabolites via cyclooxy-genase (prostaglandins and thromboxane) and lipooxygenase (leukotrienes) (Figs. 3.17 and 3.18).
Steroids increase the availability of ß-adrenergic receptors, whose activation provokes a rise in cAMP and inhibits mast cell degranulation. They also decrease corneal neovascularization.
Steroids have a potent antiinflammatory action and are still indicated in severe forms that carry a risk of functional sequelae (mainly AKC).
In rats, dexamethasone only prevents experimental allergic reactions (clinical and histological edema, conjunctival infiltration by eosinophils) when administered repeatedly 6,4, and 2 hours before the challenge.88
Steroid therapy should only be prescribed when other antiallergic treatments fail, and only for a short period. Its use is limited by the risk of induced cataract and glaucoma,89 her-pesvirus infection (more frequent in atopic subjects), and delayed corneal epithelial healing. These complications are more likely to occur when patients have become steroid-dependent and have self-treated for long periods. Ocular hypertension is, however, less frequent with fluorometholone,89 1 % rimexolone,90 and loteprednol etabonate 0.2%.90a
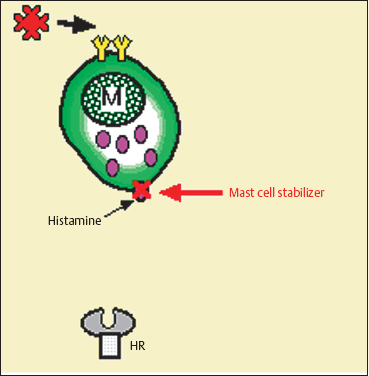
Fig. 3.16 Mode of action of mast cell stabilizers
Nonsteroidal Antiinflammatory Drugs
Mast cell degranulation is accompanied by arachidonic acid release from membrane phospholipids. The former is transformed into prostaglandin D2 and leukotrienes, respectively, by the cyclooxygenase and lipooxygenase pathways (Fig. 3.20). Once histamine has been released, prostaglandins and leukotrienes increase vascular permeability.
Nonsteroidal antiinflammatory drugs (NSAIDs) are cyclooxygenase inhibitors. They decrease conjunctival hyper-emia (PGD2, PGE2, and PGF2A), conjunctival edema (PGD2 and thromboxane A2), mucus hypersecretion (PGD2, PGJ2, and thromboxane A2), and eosinophil infiltration (PGD2 and PGJ2).91 Ketorolac is indicated for the treatment of allergic conjunctivitis. Its antiinflammatory action is associated with a dose-dependent antipruriginous effect.91
Topical NSAIDs (Table 3.7) often relieve symptoms, reduce conjunctival inflammation, and act as steroid sparing agents. Flurbiprofen seems to improve conjunctival hyperemia and itching after conjunctival allergen challenges.92 Therefore, NSAIDs should be considered as symptomatic treatments, even if they are insufficient to control severe forms of ocular allergy. They should be used cautiously because of the risk of epithelial toxicity.
Fig. 3.17 Mode of action of corticosteroids PAF: platelet-activating factor; MBP: major basic protein; ECP: eosinophilic cationic protein
Table 3.6 Topical corticosteroids
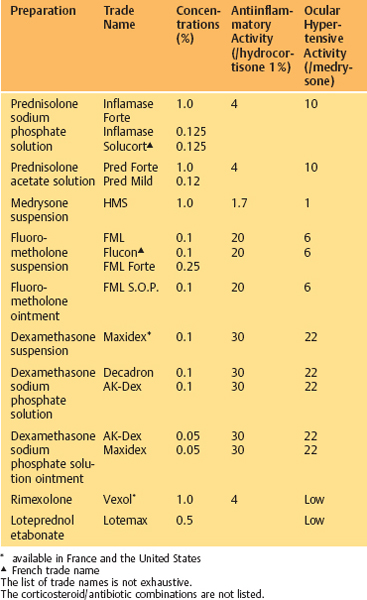
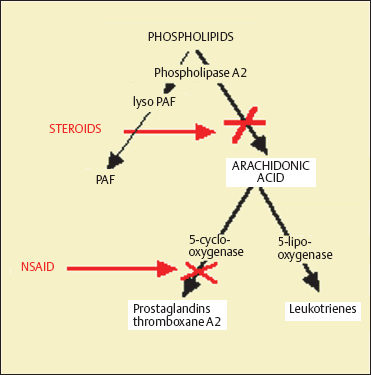
Fig. 3.18 Mode of action of corticosteroids and nonsteroidal anti-inflammatory drugs
Table 3.7 Nonsteroidal antiinflammatory drugs approved for use as topical ophthalmological preparations
| Generic Name | Trade Name | Concentration (%) |
|---|---|---|
| Diclofenac | Voltaren Voltarène* | 0.1 0.1 |
| Flurbiprofen | Ocufen* | 0.03 |
Ketorolac | Acular* | 0.5 |
| Suprofen | Profenal | 1.0 |
| Indomethacin | Indocid Indocollyre  | 1.0 0.1 |
* available in France and the United States
 French trade name
French trade name
 only nonsteroidal antiinflammtory drug approved for allergic conjunctivitis
only nonsteroidal antiinflammtory drug approved for allergic conjunctivitis
Oral aspirin has been used in VKC. The other oral NSAIDs do not seem to be effective on ocular allergic manifestations.93
Cyclosporine
Cyclosporine prevents T lymphocyte activation and interferes with cell-mediated hypersensitivity phenomena. It reduces the production of IL-2 and IL-2-receptors, and reduces the expression of class II major histocompatibility complex antigens at the surface of immunocompetent cells. It also inhibits cooperation between B cells and T cells. It may interfere with humoral hypersensitivity reactions linked to mast cell de-granulation94 and have an antipruriginous action.95
Cyclosporine A 2% eyedrops have been used successfully to treat necrotizing scleritis, Sjögren’s syndrome, ligneous conjunctivitis, and VKC. Experimental studies have shown that cyclosporine penetrates into the conjunctiva, epithelium, corneal stroma, and sclera after topical administration.96,97,98
Oral cyclosporine (3-5 mg/kg per day) is reserved for severe ocular inflammation99 such as that associated with uveitis and Mooren ulcers,100 and for the prevention of corneal graft rejection. It carries a risk of renal failure, arterial hypertension, hepatotoxicity, hypertrichosis, gingival hyperplasia, and lymphoma (a long-term adverse effect). Cyclosporine must be reserved for severe, sight-threatening forms of ocular allergy.
Miscellaneous
Synthetic Human IgE Pentapeptide
Synthetic human IgE pentapeptide (HEPP) has a poorly known mechanism of action. It appears to be more effective than some mast cell stabilizers on the symptoms and clinical signs of allergic conjunctivitis.101
FK506
FK506 (Tacrolimus) inhibits the signal transduction that leads to IL-2 gene transcription and IgE synthesis. Administered topically before a conjunctival allergen challenge in rats, it attenuates the experimental allergic conjunctivitis and reduces mast cell degranulation.102 It seems to be more effective than betamethasone and sodium cromoglycate103 especially when administered systemically.103a
Lipooxygenase Inhibitors
Lipooxygenase inhibitors (REV 5901 )104 and leukotriene D4 receptor antagonists (MK571 )105 are undergoing assessment. In animals, both compounds seem to be as effective as anti-histamines on immediate hypersensitivity reactions.106
Monoclonal Antibodies
Treatment with monoclonal antibodies directed against cell adhesion molecules (which play an important role in allergic conditions) are undergoing evaluation. Anti-ICAM-1 and anti-LFA-1 monoclonal antibodies reduced the clinical signs of allergic conjunctivitis and significantly inhibited cellular infiltration of the conjunctiva.106a,106b
As a general rule the different allergic treatments must be adapted to the severity of clinical manifestations. Anti-histamines are useful for controlling itching; mast cell stabilizers can be effective when administered before contact with the allergen; and NSAIDs improve clinical tolerance in moderate forms. Steroid therapy remains the most effective antiinflammatory measure but must be reserved for severe forms.
Immunotherapy
Immunotherapy is aimed at inducing host tolerance of the allergen. It has been used clinically since 1911,107 but its efficacy on ocular allergic manifestations is inconsistent.
The allergen must first be identified by skin tests, which must be interpreted with great care. Indeed, there are 20% false-positive results with pneumallergens, and the risk of cross-reactions between allergens is well documented.
The hyposensitization technique comprises repeated antigen administration, at gradually increasing doses, by the systemic (sublingual or subcutaneous) or topical (conjunctival) route. Hyposensitization by the conjunctival route seems to be poorly effective, even if good results have been reported in some cases.108 Hyposensitization by the systemic route carries a risk of anaphylactic shock, although this is a rare complication.109 To limit these risks, some allergens used in hyposensitization procedures are polymerized with glutaraldehyde or polyethylene glycol.107 Purer allergen preparations may become available in future.
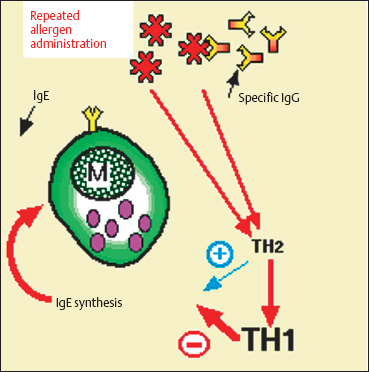
Fig. 3.19 Mechanisms of action of immunotherapy (hyposensitization)
The mechanism of action of immunotherapy is controversial, and several hypotheses have been proposed110:
—Repeated allergen administration at a low dose provokes a rise in specific IgG that competes with specific IgE and prevents the allergen from reaching mast cell IgE.
—Hyposensitization provokes a rise in CD8+ T cell numbers and transforms Th2 lymphocytes (which stimulate IgE production) into Th1 lymphocytes (which decrease IgE synthesis via gamma interferon). Serum IgE levels fall after a transient increase (Fig. 3.19).
—Finally, immunotherapy acts on T lymphocytes, monocytes, and platelets, and inhibits the production of mediators that favor histamine release.
Immunotherapy has proved effective in rhinoconjunctivitis111 due to weed pollens, grasses, and some fungi.
In animal experiments, oral antigen administration led to a reduction in conjunctival exudation, leukocyte infiltration and serum specific IgE levels after a conjunctival allergen challenge.112 Its efficacy in allergic conjunctivitis in vivo seems to be inconsistent.
Indications
Hay Fever Conjunctivitis and Perennial Allergic Conjunctivitis
This condition is linked to IgE-mediated humoral hypersensitivity. The symptoms are usually moderate and the risk of functional sequelae is negligible. Topical corticosteroids are very effective but are not essential in this mild, recurrent condition. Topical and/or systemic antihistamines and mast cell stabilizers are usually sufficient to control flare-ups.
Oral antihistamines improve both ocular and extraocular symptoms.
Topical levocabastine 0.05 % administered twice daily for 2-16 weeks is effective on functional signs in 71-87% of cases.113,114,115 It appears to be as effective as113 or more effective than115 mast cell stabilizers (cromoglycate),116,117,118 especially during periods of contact with the allergen.97 Likewise, it appears to be as effective as113 or more effective than118 oral antihistamines on ocular symptoms. It is well tolerated118 in 89% of cases, a Figure comparable to that obtained with a placebo.115 Treatment discontinuation for adverse effects is rare
Mast Cell Stabilizers
Topical disodium cromoglycate (2% cromolyn, four times daily, or 4% cromolyn, twice daily) is effective119 on signs and symptoms of hay fever conjunctivitis in 75% of cases.120 Its efficacy is comparable to that of oral antihistamines, even if terfenadine seems to be more effective on tearing.121 If mast cell stabilizers are ineffective, switching to another compound in the same therapeutic class can improve symptoms. For example, nedocromil sometimes improves the symptoms of conjunctivitis resistant to disodium cromoglycate.122
Nedocromil 2% topically, twice per day, administered before or during the period of allergen contact significantly improves symptoms (itching and conjunctival hyperemia).123,124,125,126 It seems to be more effective than oral antihistamines.124,127 It is more effective on symptoms when administered during periods of allergen exposure.125
Lodoxamide 0.1 % is more rapidly effective on the symptoms of hay fever conjunctivitis than is disodium cromoglycate 2%.128 Lodoxamide 0.1% seems to be more effective than NAAGA on symptoms (tearing, itching, and photophobia) and signs (papillary hypertrophy and epithelial lesions) after 1 or 2 months of treatment.129
Nonsteroidal Antiinflammatory Drugs
These can be administered topically in acute phases of allergic disease. They may act as steroid-sparing agents.
The efficacy of flurbiprofen, diclofenac, and ketorolac has been demonstrated in double-blind, placebo-controlled trials on the symptoms of ocular allergy (itching, hyperemia, and tearing).130,131,132
Topical Corticosteroids
A short course of topical corticosteroids may be effective in severe forms resistant to other treatments.
Immunotherapy
Immunotherapy has been attempted,133 although it is more effective in rhinitis than in allergic conjunctivitis. It appears to be especially effective on allergies due to weed pollens, grasses, and house dust mites.
Vernal Keratoconjunctivitis
Treatment of VKC is aimed at improving symptoms without necessarily normalizing the signs at all costs. Psychological management of the child and family is often necessary because of the frequent behavioral and developmental disorders in these highly symptomatic children.
The risk of iatrogenic complications must not be overlooked in VKC, which usually resolves in adulthood.4 Corneal complications call for aggressive management.
Identification of the allergen is useful, as its eradication can be beneficial. In some cases removal of a rug or carpet, installation of an air conditioning device or an air filtration system, and removal of household pets can improve clinical tolerance. Stays in cold, dry climates or at high altitudes are recommended. Cold compresses can be applied to the eyelids and eye patching reduces the friction of the tarsal papillae on the cornea. Patients must be discouraged from rubbing the eyelids because this maintains mast cell degranulation and favors epithelial lesions. Bandage contact lenses carry a risk of super-infection.
Hyposensitization is often disappointing and is not usually recommended in VKC.
Antihistamines
Oral antihistamines administered before bed have a beneficial sedative action in children with VKC.
Levocabastine is effective in 70% of cases (placebo 33%) on symptoms (photophobia [on D8], itching, and conjunctival irritation [D30]134) and signs (Horner-Trantas dots, lid edema, conjunctival hyperemia, and epithelial lesions).84 Its period of action seems to be shorter than that of disodium cromoglycate.84 It is as well tolerated as a placebo.134
Mast Cell Stabilizers
Mast cell stabilizers seem to reduce steroid requirements and can safely be used for maintenance treatment. Adverse effects are generally limited to a tingling sensation during eyedrop instillation.135
Disodium cromoglycate 2% administered for 6 weeks improves tearing, conjunctival hyperemia, and limbal edema during the active phase of inflammation,135 especially when patients have a history of allergy (randomized, double-blind, placebo-controlled trial). It is, however, less effective than topical steroid therapy (fluorometholone)136 (double-blind trial).
Nedocromil 2% administered for 6 weeks reduces itching, conjunctival hyperemia, and neutrophil, eosinophil, and lymphocyte counts in tears.137 The functional improvement is comparable to nedocromil 2% and disodium cromoglycate 2%,138 although nedocromil seems to be more effective on itching and chemosis.138
Lodoxamide 0.1 % administered for 90 days (double-blind, randomized, placebo-controlled trial) improves the epitheliopathy, tarsal papillae, and limbal involvement,139 although the functional improvement (photophobia and itching) was not statistically different from that obtained with the placebo.139 Lodoxamide 0.1 % seems to be more rapidly effective than disodium cromoglycate 4% on symptoms.84 It reduces the level of ECP in tears, a parameter that correlates with corneal involvement.140 Lodoxamide 0.1 % is more effective than NAAGA 6% (randomized double-blind trial) on symptoms (photophobia, tearing, and itching) and signs (reduction in papillary hypertrophy and epitheliopathy).129
Corticosteroids
Topical corticosteroids must be considered as a symptomatic treatment to control inflammatory exacerbations. They must not be prescribed for long periods because of the risk of adverse effects. Indeed, cortisone-induced cataract sometimes occurs a few months after beginning treatment in young patients.136
Topical corticosteroids are therefore prescribed for treatment periods of 10 days, with a gradually decreasing frequency of use. Supratarsal steroid injections given for resistant forms of VKC led to the disappearance of giant papillae in 13-15 days and an improvement in limbal involvement and corneal ulceration in 1-3 weeks.141
The clinical efficacy of topical dexamethasone 0.1 % correlated with a 58% reduction in ECP levels in tears.142
Nonsteroidal Antiinflammatory Drugs
Oral aspirin at a dosage of 25-50 mg/kg per day seems to prolong remissions and to reduce steroid requirements.143,144 Alone,143 or combined with disodium cromoglycate,145 it improves symptoms (itching and tearing) and palpebral, limbal, and corneal lesions significantly more effectively than a placebo. It reduces conjunctival inflammation without affecting papillary hypertrophy.146 The improvement begins from the second week of treatment143 in 88% of cases, sometimes after the failure of steroids or disodium cromoglycate.146 Aspirin therefore seems to be a useful adjuvant treatment for severe forms in which steroid therapy is indicated. The high recommended dosages (up to 1.5 g per day) are, however, associated with adverse effects (gastric intolerance and increased bleeding time) and cannot be administered for long periods.
A symptomatic improvement was reported after topical treatment with 1 % indomethacin in 84% of cases147 and with suprofen after day 7.148 Indomethacin is not sufficiently effective to control the inflammation associated with VKC.149
Cyclosporine
In severe forms necessitating steroid therapy, topical 2% cyclosporine rapidly controls the inflammation and reduces steroid requirements. It improves symptoms in 76% of cases from day 7,150 an effect that lasts throughout treatment.151 It reduces conjunctival hyperemia, papillary hypertrophy, and limbal and corneal involvement152 (double-blind, placebo-controlled trial). It remains effective in severe forms of VKC. It reduced the level of ECP in tears by 84 %142 and also the number of eosinophils in tears.153 It is not accompanied by systemic adverse effects because its passage into the systemic circulation is negligible.153 However, inflammation relapses within 2-4 months following treatment cessation.150,151
Other Drugs
Mucolytic agents (5, 10, or 20% acetylcysteine) fluidify thick mucous secretions and can improve patient comfort.
Mipragoside is a ganglioside ester that inhibits histamine release by human basophils and rat mast cells after antigenic stimulation.154 It reduces the number of eosinophils in the rabbit conjunctiva.155 In VKC, 0.5% mipragoside significantly reduces symptoms (itching and tearing) and some signs (hyperemia)156 (randomized placebo-controlled trial).
Surgical Treatment
In VKC, surgery can contribute to controlling inflammation and corneal lesions. Surgical procedures that have become obsolete because of their inefficacy, such as oral mucosal grafting, will not be dealt with here.
Cryotherapy
Cryotherapy of the superior tarsal conjunctiva provokes massive mast cell degranulation and induces a period of remission. The regression of giant papillae is not always associated with an improvement of corneal lesions, for they are linked to the epithelial toxicity of the mediators released. The cryode is applied to the superior tarsal conjunctiva at a temperature of -60° to -80°C for 30 seconds. The procedure is repeated two or three times.
Cryotherapy applied to 30 cases of active VKC improved symptoms and decreased papillary hypertrophy, conjunctival hyperemia, limbal nodules, and punctate epithelial keratitis. However, relapse occurred in 3.3% of the cases after 1 year. In this study cryotherapy was combined with oral aspirin administration (0.5-1.5 g per day).157
Table 3.8 Vernal ulcers and plaques in vernal keratoconjunctivitis (according to Cameron13)

MCS: mast cell stabilizer; TC: topical corticosteroids; TAB: topical antibiotics
Surgery of vernal plaque
In VKC, shield ulcers reepithelialize slowly, with a risk of superinfection and corneal scarring.
Clear-based ulcers (grade 1) usually respond to medical treatment, combining topical prophylactic antibiotic therapy, topical steroid therapy, and mast cell stabilizers.13
Vernal plaques (grade 3) and ulcers with an opalescent base (grade 2) reepithelialize slowly during medical treatment, and the epithelial defect may sometimes persist. The material deposited onto the bed of the ulcer may be toxic and may prevent complete reepithelialization. Scraping of the bed and edges of the ulcer, combined with medical treatment, leads to healing within 8 days13 in 87% of cases.158 A few pulses of excimer laser phototherapeutic keratectomy (PTK) can also be applied to the base of the ulcer after scraping, and leads to reepithelialization within a week (Fig. 3.23).158 Sometimes, superficial keratectomy12 is necessary to remove all the inflammatory material that interconnects with the underlying Bowman’s layer.159 In practice, medical treatment of grade 2 or 3 corneal lesions can be attempted first. In case of failure after 8 days of treatment, surgery is indicated (Table 3.8).
Surgical treatment of corneal complications
Corneal sequelae sometimes necessitate specific management. Thus, in corneal ectasias, contact lenses or penetrating kerato-plasty may be indicated. The results of penetrating kerato-plasty are still disappointing in this indication.14
Atopic Keratoconjunctivitis
Atopic keratoconjunctivitis is a severe form of ocular allergy because it carries a risk of severe corneal sequelae. It is usually associated with systemic allergic manifestations and calls for multidisciplinary management. Potential allergens, toxins, and irritants should be eradicated. When this is impossible (e.g., pollen), treatment with oral antihistamines and mast cell stabilizers should be initiated before the period of allergen contact. Hyposensitization can be attempted in refractory forms, even if the results are inconsistent.
Ocular discomfort is often severe, leading to the use of multiple medications including prolonged and frequent use of topical corticosteroids. The functional prognosis is poor, because of the severity of the condition and its iatrogenic complications. These patients should be advised not to use eyedrops containing preservatives.
Seborrheic and staphylococcal blepharitis are frequently associated with AKC. They worsen the corneal lesions and must receive specific treatment. Daily eyelid hygiene and systemic antibiotic therapy (cyclines) should be prescribed. Staphylococcal superinfection can be treated with topical antibiotics.
Washing the conjunctival fornices with normal saline dilutes the allergens and provides a tear substitute for these patients, who often have dry eyes. It also neutralizes local pH. The use of topical mucolytic agents has been proposed to liquefy the sometimes thick secretions.160
Local Treatment
Mast cell stabilizers are used as maintenance therapy for AKC.26 Disodium cromoglycate improves symptoms in two thirds of cases24 and reduces steroid requirements.
Topical corticosteroids are frequently necessary during exacerbations of the inflammatory reaction, but must only be prescribed for short periods.37
Cyclosporine 2% eyedrops are sometimes prescribed in severe forms requiring steroid therapy.26
Systemic Treatment
Local treatment is generally inadequate in AKC, which is frequently accompanied by cutaneous involvement. Anti-histamines can be administered systemically.26 The full dose regimen should be introduced gradually to improve tolerability. Sedative drugs are best administered in the evening.
Systemic steroid therapy is only indicated in those rare cases in which pulmonary or cutaneous allergic manifestations are not controlled by other antiallergic treatments.
Oral cyclosporine A (3-5 mg/kg per day) has proved effective in atopic dermatitis,161,162 and itself appears to have an anti-pruriginous action.94 Its efficacy in severe, steroid-resistant forms of AKC was recently demonstrated.163 An improvement in symptoms and regression of corneo-conjunctival inflammation was reported in a series of four patients. The need for regular monitoring of kidney function must be underlined. Any change in renal function necessitates adjusting the dose or ceasing treatment. Treatment discontinuation is followed by a relapse of the ocular inflammation.
Miscellaneous
In rare cases of AKC associated with a marked rise in serum IgE and the presence of antistaphylococcal IgE, plasmapheresis has been used with good results.26
Treatment of Complications
Penetrating keratoplasty
Corneal complications are frequent in AKC and are responsible for severe alterations of visual function. Penetrating keratoplasty is sometimes used to treat secondary corneal opacities but the prognosis is uncertain because of the major risk of graft rejection, ocular dryness, and associated palpebral conditions.165 The persistent conjunctival inflammation renders the sutures unstable.166 Corneal graft rejection occurs in more than 50% of cases.165
Surgery of palpebro-conjunctival scars
Atopic keratoconjunctivitis is associated with conjunctival fibrosis and symblepharon formation. These cicatricial lesions can benefit from reparative surgery consisting of margino-plasty and repair of the conjunctival fornices.
Giant Papillary Conjunctivitis
The treatment of GPC is based on a three-pronged approach: contact lens fit and type; patient education in lens care; and drug therapy. These measures are accompanied by the resolution of symptoms in 79% of cases,50 even if the papillae can persist for several weeks.
Withdrawal of contact lenses alone may not be sufficient to resolve the inflammation; it must be combined with other therapeutic measures.50
Contact Lens Care
Recommended daily care includes cleaning the lenses, rinsing in normal saline, and disinfection. Solutions containing potential allergens such as preservatives and mercury derivatives must be avoided. Daily wearing of contact lenses can improve the symptoms.
Ocular prostheses must be washed daily with soap, then rinsed in clean water and placed in a dry place before being used.
Contact Lens Fit and Type
During the inflammatory exacerbation it is usual to discontinue wearing the lenses for a few weeks before considering a new adaptation. Discontinuation of contact lens wear is often inadequate and must be combined with other therapeutic measures.50 Lenses with a different material and/or design can be adapted in 75% of cases. Gas-permeable lenses are sometimes better tolerated. The use of disposable lenses does not suffice to prevent the inflammatory reaction.167,168 Some authors recommend shortening the duration of lens wear.148 Soft lens wearers can be offered rigid lenses that are less susceptible to deposits and less traumatic for the superior tarsal conjunctiva given their smaller diameter and decreased edge lift.169
Medical Treatment
Mast cell stabilizers
Disodium cromoglycate is effective in the early stages, in which it improves symptoms and signs. It improves tolerance during the period of readaptation to the contact lens.170 An improvement in symptoms is obtained in 79 % of cases after adoption of hygiene measures and local treatment with disodium cromoglycate.50 Nedocromil 2% improves the sensation of itching during the initial phase of treatment (first 3 weeks), but has no beneficial action in the longer term.171 Some authors consider that the efficacy of mast cell stabilizers may be linked to the washing effect on the conjunctival fornices.172 The relative efficacy of mast cell stabilizers could be explained by the prevalence of MMCs in GPC.
Corticosteroids
Their efficacy is only moderate and their use is not essential in this indication. Steroids may be associated with more rapid regression of conjunctival papillae.173 They are not recommended for contact lens wearers. They may be indicated for patients with ocular prostheses.
Nonsteroidal antiinflammatory drugs
Suprofen administration for 28 days is associated with an improvement in symptoms and with regression of the papillae.174
References
3. Buckley RJ. Vernal keratoconjunctivitis. Int Ophthalmol Clin 1988; 28: 303-8.
4. Dart JKG. The epidemiology of vernal keratoconjunctivitis. In: Proceedings of the Second Visions International Ophthalmology Workshop, Bollington, Cheshire, England, 1989, Pennine Press.
6. Hoang-Xuan T. Pathologie inflammatoire du limbe. J Fr Ophtalmol 1993; 16: 560-5.
8. Beigelman MN. Vernal conjunctivitis. Los Angeles 1950, University of Southern California Press.
9. Jones BR. Vernal keratitis. Trans Ophthalmol Soc UK. 1961; 81: 215-28.
11. Udell IJ. Differential diagnosis of ocular allergy. Int Ophthalmol Clin 1988; 28: 275-81.
12. Buckley RJ. Vernal keratopathy and its management. Trans Ophthalmol Soc UK 1981; 101: 234-8.
16. Brauninger GE, Centifento YM. Immunoglobulin E in human tears. Am J Ophthalmol 1971; 72: 558-61.
23. Hogan MJ. Atopic keratoconjunctivitis. Trans Am Ophthalmol Soc 1952; 50: 265-81.
26. Foster CS, Calonge M. Atopic keratoconjunctivitis. Ophthalmology 1990; 92: 992-1000.
27. Braude LS, Chandler JW. Atopic corneal disease. Int Ophthalmol Clin 1984; 24: 145-6.
31. Copeman PW. Eczema and keratoconus. Br Med J 1965; 5468: 977-9.
32. Beethan WP. Atopic cataracts. Arch Ophthalmol 1940; 24: 21-37.
62. Pecoud A. Review of diagnostic methods in allergy. Schweiz Med Wochenschr 1987; 117: 252-5.
64. Giovanni M, Spada E, Broccoli MP. Conjunctival provocation tests in suspected allergic conjunctivitis: a clinical study. Ophthalmologica 1990; 201: 1-4.
92. Bishop K, Abelson MB, Cheetham J, Harper D. Evaluation of flurbi-profen in the treatment of antigen-induced allergic conjunctivitis. Invest Ophthalmol Vis Sci 1990; 31(Supp): 487.
107. Noon L Prophylactic inoculation against hayfever. Lancet 1911; 1: 1572.
110. Bousquet J, Michel FB. Advances in specific immunotherapy. Clin Exp Allergy 1992; 22: 889-96.
137. Bonini S, Barney NP, Schiavone M, Centofanti M, Berruto A, Bonini S, Allansmith MR. Effectiveness of nedocromil sodium 2 % eye-drops on clinical symptoms and tear fluid cytology of patients with vernal conjunctivitis. Eye 1992; 6: 648-52.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree