13
HIV and Intraocular Inflammation
John J. Huang and Dawn S. Russell
Overview
The management of ocular inflammation in HIV patients is often complicated. This is related to the wide range of infectious etiologies and their unique presentation in HIV and especially AIDS patients. HIV and AIDS patients are often on a variety of systemic medication commonly associated with noninfectious ocular inflammation. In addition, HIV is associated with an immunocompromised clinical state as well as a state of immune dysregulation. The common infectious etiologies associated with HIV patients include syphilis, herpes simplex virus (HSV), herpes zoster virus, cytomegalovirus (CMV), fungal infections, tuberculosis, and toxoplasmosis. Noninfectious etiology of uveitis may be related to the masquerade syndromes such as lymphoma, immune recovery uveitis (IRU), or autoimmune phenomenon associated with HIV infection.
Clinical factors in the workup of uveitis in an HIV patient should always include a detailed medical history with a thorough examination. Medical history should include (i) CD4 count and viral load, (ii) current medications, and (iii) past ocular and systemic infections. Ocular examination should assess the degree and location of ocular inflammation. Infectious etiologies commonly associated with anterior uveitis include all the viral etiologies and syphilis. Retinitis is commonly seen with toxoplasmosis, syphilis, herpes family of viruses, and ocular fungal infection. Choroidal infiltration is seen in Pneumocystis carinii and in Cryptococcus infections. In patients with a negative workup for infectious etiologies, noninfectious causes such as IRU, ocular lymphoma, and autoimmune etiologies should be considered.
The clinical course and management of human immunodeficiency virus and the associated acquired immune deficiency syndrome (HIV/AIDS) have changed significantly since the era of highly active antiretroviral therapy (HAART). Once considered a fatal disease, it is now managed as a chronic condition. HIV is caused by a retrovirus in the Lentivirinae family. The virus is also known as human lymphotropic virus type III and is a RNA retrovirus with one of its key characteristics being its ability to replicate at a rapid rate. HIV has devastating effects on its host by a gradual destruction of the host immune function. HIV has two unique strains, HIV-1 and HIV-2. HIV-1 has worldwide prevalence, whereas HIV-2 is seen predominantly in Central and West Africa. There is a 40% homology in the nucleotide sequence between HIV-1 and HIV-2.
The HIV virus has a strong affinity to host cells through binding of CD4 cellular receptor on T lymphocytes, monocytes, and macrophages (Fig. 13.1). The virus is able to further gain entry into the host cell through additional binding of the CCR5 and CXCR4 receptors. The virus enters the host cell using the viral gp41 protein. Upon entry into the host cell, the viral RNA is translated into DNA by the viral reverse transcriptase. New viral particles are synthesized in the infected host cell, leading to cell death and the release of additional viruses. Infected T cells decrease in number leading to overwhelming immune deficiency and vulnerability to opportunistic organisms.
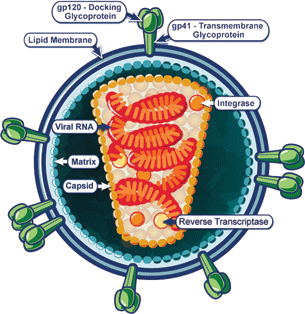
Figure 13.1 Molecular structure of the HIV virus with surface receptors for binding T lymphocytes.
STAGES OF HIV INFECTION
- Primary infection or acute retroviral syndrome: occurs within 1–6 weeks after initial infection with symptoms of fever, rash, myalgias, headache, or gastrointestinal symptoms.
- Clinical latency: lasts, roughly 10 years with gradual decline of CD4 T lymphocytes level of 75 cells/µL/year.
- Opportunistic infections: immunocompromised stage of HIV infection due to the severely depressed levels of CD4 T lymphocytes. The host is predisposed to a variety of infections such as toxoplasmosis, cryptococcus, Pneumocystis carinii, HSV, varicella-zoster virus (VZV), and CMV (Table 13.1).
Table 13.1 Major causes of intraocular inflammation in HIV patients

HIV RETINOPATHY
HIV retinopathy is one of the most common noninfectious ocular findings seen in HIV/AIDS patients. The clinical findings of cotton-wool spots and retinal hemorrhage seen in HIV retinopathy are associated with microvasculopathy due to HIV viral particles and antibody deposition. It has been shown histologically that the cotton-wool spots seen in HIV/AIDS patients are identical to that seen in these other retinal conditions. At the histological level, the cotton-wool spots represent a localized area of blocked axoplasmic flow in the retinal nerve fiber layer. There is subsequent localized edema and an accumulation of cytoid bodies (swollen axons) that form cotton-wool spots. HIV retinopathy is seen in approximately 50% to 70% HIV patients (Fig. 13.2).
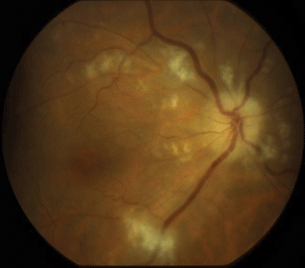
Figure 13.2 Diffuse cotton-wool spots in the posterior fundus in a patient with advanced HIV disease with HIV-associated retinopathy.
HERPES SIMPLEX VIRUS
HIV/AIDS patients are particularly susceptible to recurrent infections with the herpes family of viruses. These are DNA viruses of which there are two strains, HSV-1 and HSV-2.
HSV-1 typically causes oral and ocular diseases, whereas HSV-2 causes genital infections; however, this is not a strict rule as there is some overlap between these viral strains. The mode of spread of this virus is via direct contact of infected secretions with skin and/or mucous membranes. Viral infection of the ciliary body, cornea, or iris leads to herpetic iridocyclitis. The presence of anterior chamber inflammation in these patients may be clinically apparent in association with dendritic lesions of the cornea or the presence of disciform keratitis. More importantly, the presence of anterior segment infection may be an early sign of HSV retinitis.
Clinical Findings
- Dendritic epithelial keratitis can progress to large geographic ulcers (Fig. 13.3).
- Neurotrophic ulcers.
- Stromal keratitis.
- Endothelitis with keratic precipitates, stromal or epithelial edema, and iritis.
- Hyphema is a common presentation in herpetic iridocyclitis with associated damage to the iris and ciliary body. This is associated with iris atrophy and transillumination defects.
- Elevated intraocular pressure.
- Viral retinitis.
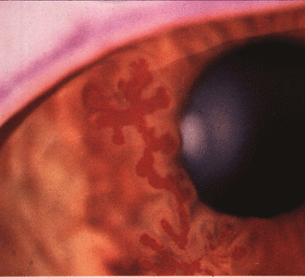
Figure 13.3 Corneal dendrite due to HSV infection of the cornea stained by fluorescein.
Differential Diagnosis
- VZV
- Bacterial or fungal keratitis
- CMV
Diagnostic Testing
- Clinical evidence of characteristic eyelid or skin lesions and corneal epithelial dendrites
- Giemsa stain of corneal scrapings, which would show multinucleated giant cells
- Immunohistochemistry (viral antigens)
- Viral culture
- Polymerase chain reaction (PCR) for corneal scrapping, anterior fluid, or vitreous fluid
- Papanicolaou stain, which would show the intranuclear eosinophilic inclusion bodies
Treatment
Treatment options for ocular HSV may vary according to the anatomical location of the infection and the need to prevent more extensive involvement.
- Herpetic keratouveitis, corneal epithelial disease
- Trifluridine 1% eyedrops nine times a day for 14 days or
- Oral acyclovir 400 mg five times a day
- Cycloplegic agents (e.g., scopolamine 0.25% t.i.d)
- Corneal debridement of loose, infected epithelium at the slit lamp as adjunctive therapy. Administer topical anesthetic eyedrops prior to procedure (0.5% proparacaine) and use a moistened cotton tip applicator to remove the epithelium.
- Trifluridine 1% eyedrops nine times a day for 14 days or
- Anterior uveitis
- Topical corticosteroids (careful examination of the cornea to rule out evidence of infective epithelial keratitis should be done prior to initiation of topical corticosteroids)
- Cycloplegic agents (e.g., scopolamine 0.25% t.i.d)
- Severe uveitis cases require oral antiviral therapy
- Acyclovir 400 mg, five times a day
- Valacyclovir 1 g, two times a day
- Cycloplegic agents (e.g., scopolamine 0.25% t.i.d)
- Topical corticosteroids (careful examination of the cornea to rule out evidence of infective epithelial keratitis should be done prior to initiation of topical corticosteroids)
- Disciform keratitis
- In mild cases, only a cycloplegic agent may be necessary (e.g., scopolamine 0.25% t.i.d)
- Prednisolone acetate 1% eyedrops four times a day
- Prophylactic antiviral agents (e.g., acyclovir 400 mg orally b.i.d)
- In mild cases, only a cycloplegic agent may be necessary (e.g., scopolamine 0.25% t.i.d)
- HSV retinitis
- Aggressive treatment with induction dose of intravenous acyclovir at 1,500 mg/m2 total medication in three divided doses × 10 to 14 days followed by oral acyclovir 1,000 mg PO twice a day for 2 to 4 weeks.
- Intravitreal ganciclovir or foscarnet may provide immediate control of the disease.
- Peripheral laser retinopexy in the area of atrophic necrotic retina for RD prophylaxis early in the recovery period.
- Aggressive treatment with induction dose of intravenous acyclovir at 1,500 mg/m2 total medication in three divided doses × 10 to 14 days followed by oral acyclovir 1,000 mg PO twice a day for 2 to 4 weeks.
HIV/AIDS patients may require long-term oral antiviral prophylaxis (e.g., acyclovir 400 mg orally b.i.d) given the increased frequency of recurrence in these patients.
VARICELLA-ZOSTER VIRUS (VZV)
“Chicken pox” is the result of primary infection with the herpes zoster virus. The virus remains dormant in the sensory nerve ganglia and then reactivation occurs resulting in “shingles” in the corresponding dermatome. Herpes zoster ophthalmicus (HZO) is the result of the involvement of the first division of cranial nerve V (CN V). HZO can be the initial or presenting feature of HIV infection. In HIV patients, VZV retinitis commonly occurs after the resolution of dermatologic and anterior segment disease. Patients should be carefully monitored even after the resolution of HZO and anterior uveitis.
Clinical Findings
- Vesicular skin rash, along a dermatome of CN V (Fig. 13.4).
- Upper eyelid lesions/extension of the rash.
- Hutchinson sign is the presence of vesicles on the tip of the nose due to the involvement of the nasociliary nerve of CN V.
- Conjunctivitis.
- Elevated mucous plaques or “pseudodendrites” of the corneal epithelium.
- Stromal keratitis.
- Neurotrophic keratitis.
- Patchy or sectoral iris atrophy secondary to vasculitis of the iris vessels and associated iritis and iridocyclitis.
- Anterior segment inflammation that can be severe and give rise to posterior synechiae.
- Trabeculitis with subsequent development of secondary glaucoma.
- Severe viral retinitis with large areas of rapidly progressing infected retina with vitreous cells and white edematous retina.
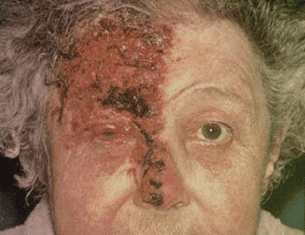
Figure 13.4 A patient with HZO with the involvement of the nasociliary nerve.
Differential Diagnosis
HSV CMV
Diagnostic Testing
- Clinical evidence of classic skin lesions in dermatomal distribution
- Viral culture
- PCR with sample or swab of the base of one of the lesions
- Tzanck smear—unfortunately does not differentiate herpes zoster from herpes simplex
- Monoclonal antibody test
- Direct immunofluorescence test
- Tzanck smear—unfortunately does not differentiate herpes zoster from herpes simplex
Treatment
- Acyclovir at a dose of 10 mg/kg IV every 8 hours. Give at a frequency of every 12 hours if creatinine is greater than 2.0 mg/dL. Continue this regimen for 7 to 10 days. Maintenance therapy is dosed at 800 mg orally five times a day.
- Famciclovir is given orally at a dose of 500 mg every 8 hours. Renal dosage adjustments are made for creatinine clearance lesser than 60 mL per minute
- Valacyclovir 1 g orally every 8 hours. Renal dosage adjustments are made for creatinine clearance lesser than 60 mL per minute.
- Intravitreal ganciclovir or foscarnet for immediate rapid disease control.
CYTOMEGALOVIRUS RETINITIS
CMV is a double-stranded DNA virus in the herpes family of viruses and is associated with opportunistic infection in immunocompromised and AIDS patients. Prior to the advent of highly active antiretroviral therapy (HAART), disseminated CMV infection was the leading opportunistic infection in patients with AIDS. Patients with the diagnosis of CMV retinitis have a life expectancy of 9 months. It most commonly occurs in those patients whose CD4 count has fallen to a level less than 50 cells/mm3. CMV predominantly affects the retina/posterior segment causing a posterior uveitis in the form of a viral retinitis. CMV retinitis accounted for more than 95% of viral retinitis in studies done on autopsy specimens of AIDS patients. The disease has a very slow course with the rate of progression or growth of approximately 250 µm per week. Patients may present with complaints of floaters, scotomas, and rarely ocular pain or discomfort. There can be gradual visual decline if there is involvement of the fovea I region or optic nerve.
Clinical Findings
- Fine keratic precipitates with minimal or no anterior chamber reaction.
- Papillitis.
- Active retinitis occurs at the borders or edges of the retinal lesions. Retinal Hemorrhage
Three main retinal manifestations are the following:
Classic, fulminant type
- Initially small, perivascular white-yellow lesions that can resemble cotton-wool spots.
- These initial lesions then progress to large areas of retinal opacification and necrosis.
- Typically involves the posterior pole and can involve optic nerve and extend from arcade to arcade (Fig. 13.5).
- Hard exudates, intraretinal hemorrhages.
- In AIDS, minimal vitritis.
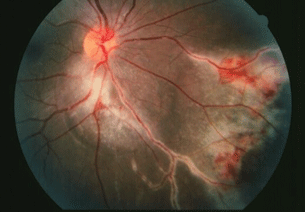
Figure 13.5 CMV retinitis with infection of the peripapillary retina, retinal vessels, and associated retinal hemorrhage.
Granular type
- Seen predominantly in peripheral lesions
- Few retinal hemorrhages and hard exudates
- Mottling of retinal pigment epithelium
- Active disease predominantly at lesion borders
- Retinal atrophy
Perivascular type
- This was initially described in healthy, immunocompetent children.
- Resembles a frosted branch angiitis in its appearance and perivascular distribution.
Differential Diagnosis
- Progressive outer retinal necrosis (PORN)
- Acute retinal necrosis (ARN)
- Toxoplasmosis retinochoroiditis
Diagnostic Testing
- Serology—CMV antibodies, although of limited value given large percentage of general population with anti-CMV antibodies
- CMV PCR—anterior chamber tap or vitreous sample
- Viral culture
- Clinical appearance—of highest value
Treatment
Intravenous Ganciclovir
- Traditionally, the first mode of therapy used for CMV retinitis
- Start with induction therapy at a dose of 5 mg/kg IV b.i.d for 2 weeks
- Maintenance therapy follows at a dose of 5 mg/kg daily for 1 week
Side Effects of Ganciclovir
- Myelosuppression—if the patient is also on zidovudine (ZDV), the dose of ZDV must be decreased as one of its major side effects is bone marrow toxicity.
Oral Ganciclovir
- Mainly used as maintenance therapy for patients who have responded favorably to intravenous ganciclovir.
- There is lower drug toxicity with oral ganciclovir.
- Disadvantage of oral ganciclovir therapy is that median time to progression of disease is less with oral than with intravenous ganciclovir.
- Also, the risk of developing CMV retinitis in fellow eye is greater in patients on oral therapy.
Intravitreal Ganciclovir
- Indicated in patients who have demonstrated progression of CMV retinitis even after treatment with intravenous ganciclovir or other agents at high dosages, particularly foscarnet.
- Also indicated in patients unable to take intravenous therapy.
- The dose for intravitreal ganciclovir is 200 μg to 2.0 mg in 0.1 mL administered twice weekly for 2 to 3 weeks for induction and then once weekly for maintenance therapy.
Intravenous Foscarnet
- Start with induction dosing at 90 mg/kg every 12 hours for 2 weeks.
- This is followed up with maintenance dosing at 90 to 120 mg/kg daily.
Side Effects of Foscarnet
- Renal toxicity.
- Foscarnet has no effect on the bone marrow and hence can be given along with ZDV with no required adjustment in ZDV dose.
Intravitreal Foscarnet
- The indication for intravitreal foscarnet is the same as for intravitreal ganciclovir—patients who have failed systemic therapy or those who are unfit for high-dose systemic therapy.
- Foscarnet intravitreal dosing is 2.4 mg (in 0.1 mL) twice weekly for 2 to 3 weeks for induction.
Oral Valganciclovir
- This is a “prodrug” of ganciclovir.
- Start with induction dosing of 900 mg twice daily for 21 days.
- This is followed up with maintenance dosing of 900 mg once daily.
- Similar side effect and toxicity profile to intravenous dosing.
Advantages of Oral Valganciclovir
- Oral dosing of this drug achieves blood levels similar to levels achieved with intravenous ganciclovir.
- Convenience of oral dosing, eliminating the need for catheter placement
Intravenous Cidofovir
- Start with induction therapy at a dose of 5 mg/kg once weekly for 2 weeks.
- Follow up with maintenance dosing at 5 mg/kg every 2 weeks.
- Comparable success rates to intravenous ganciclovir.
- With intravenous cidofovir administration, intravenous hydration is essential to prevent renal impairment.
- Concurrent administration of probenecid is also required to prevent renal toxicity.
- Comparable success rates to intravenous ganciclovir.
Side Effects of Cidofovir
- Renal toxicity
- Anterior uveitis
- Hypotony
Ganciclovir Intravitreal Implant (Vitrasert)
The ganciclovir implant delivers 4.5 mg of the drug over a period of 5-8 months. The implant is surgically placed in the pars plana region. Phase III clinical trials of 188 patients with vitrasert demonstrated significant delay of CMV retinitis progression in AIDS patients compared to intravenous ganciclovir.
Advantages of Ganciclovir Implant
Concurrent use of oral valganciclovir decreases the risk of development of CMV retinitis in the fellow eye.
Implant insertion is a relatively low-risk outpatient surgery.
The ganciclovir implant achieves local drug concentrations significantly higher than that with intravenous ganciclovir.
Disease Course
Overall disease activity is assessed with the following parameters:
- Evaluation of borders of lesion. Careful evaluation of the borders of the lesion is essential. At each visit, retinal photographs can be taken to determine any change in terms of overall size of the lesion.
Hemorrhages and retinal whitening assessment of borders for the presence of new retinal whitening and hemorrhages.
The resolution of disease activity can be expected in most patients within 6 weeks of initiation of therapy. Ganciclovir and foscarnet have a synergistic effect when used together in the treatment of CMV retinitis. Furthermore, it is helpful in the prevention of disease progression if there is initial failure with the use of either drug alone.
Patients on HAART have demonstrated more successful long-term results with decreased recurrence of active retinitis. Approximately 50% of patients not on HAART, CMV retinitis recur within 2 months of being on maintenance CMV therapy. This also translates to lower rates of overall progression and spreads to the fellow eye in patients on HAART.
Patients with CMV retinitis may develop viral resistance, in which case a change in the mode of therapy (from intravenous to intravitreal) is indicated. Viral resistance can be roughly determined by evaluating the blood CMV viral load. If this number is undetectable, then it is highly unlikely that there is viral resistance. It should be noted, however, that partial viral resistance can still develop in patients on HAART with CD4 levels greater than 100 cells/mm3.
Disease activity is also indirectly monitored using the CD4 count in patients on HAART. With successful treatment, we can see an increase in the CD4 count to a level where discontinuation of the therapy can be achieved. It has been advised that anti-CMV therapy not be discontinued unless there is a consistent CD4 count greater than 100 cells/mm3 for a period of at least 3 to 6 months. These patients need to be followed closely, and it is suggested that they be seen every 6 to 12 months, given the likelihood of disease recurrence even with higher CD4 levels (above 100 cells/mm3), and even more often if there is a decrease in the CD4 level.
Complications of CMV Retinitis
- Development of atrophic holes at the edge of the lesions
- Retinal detachment, which can be seen in approximately 50% of patients
- IRU
Progressive Outer Retinal Necrosis
This is a form of posterior uveitis or retinitis secondary to the VZV but has also been seen with the HSV. PORN occurs typically in patients who are severely immunocompromised and can often be seen in patients with AIDS, usually when the CD4 count is less than 50 cells/µL, but typically at a median CD4 level of 21 cells/µL. Quite a number of patients may have recent history of ipsilateral HZO (67% cases). The overall prognosis is very poor and patients have a rapid visual decline.
Clinical Findings
- Very little, if any anterior chamber inflammation.
- Minimal to no vitritis.
- Initially, isolated areas of retinal whitening in the periphery that rapidly coalesce together in a circumferential fashion.
- Lesions are deep and multifocal (Fig. 13.6).
- Involvement of the posterior pole may occur early.
- Retinal vessels are not involved.
- Can rapidly spread to the fellow eye.
- Can also develop retinal detachment.
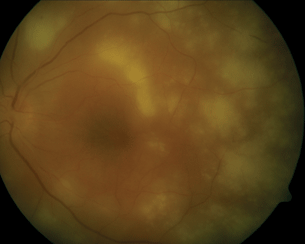
Figure 13.6 PORN with extensive multifocal areas of outer retinal inflammation with minimal vitreous cells. The retinal vessels are uninvolved.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


