(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
Retinal vein occlusion is a well-known clinical entity, but there is no corresponding detailed information about the clinical features of vortex vein occlusion, in man or in primates. There are some old studies dealing with experimental occlusion of vortex veins designed primarily to produce glaucoma [1–11], one study designed to produce retinal detachment [12], and others to study histopathological changes [13–15]. All the earlier studies were carried out in rabbits except for those by Adamiuk [1] and Macri [11] in cats, Müller [8] in the horse, Sachsenweger and Lukoff [12] in dogs, and Nishikawa et al. [15] in five Japanese macaque monkeys. Koster [9] described ocular signs of vortex vein occlusion in rabbits. To investigate the clinical features of vortex vein occlusion comprehensively, I [16] conducted an experimental study of vortex vein occlusion in 40 eyes of rhesus monkeys. All eyes had these examinations: external ocular, intraocular pressure, fundus, fluorescein fundus angiographic, and histopathological features or silicone rubber perfusion. Various vortex veins were occluded in different combinations, as shown in Table 27.1. The findings of the study are discussed in detail elsewhere [16], and following are the main features.
Vortex veins occluded | Number of eyes | |
|---|---|---|
Number | Name | |
All veins | All vortex veins | 14 |
3 | ST + SN + IT | 7 |
ST + IT + IN | 1 | |
ST + SN + IN | 1 | |
2 | ST + SN | 4 |
ST + IT | 5 | |
SN + IT | 3 | |
1 | ST | 3 |
SN | 1 | |
IT | 1 | |
In this study, the maximum brunt of vortex vein occlusion was borne by the anterior segment of the eye – most marked by the vessels in the ciliary processes.
Ciliary Processes and Ciliary Body Findings
Histological examination of the eyes, soon after the occlusion of the vortex veins, revealed marked vascular congestion and dilated vessels in the ciliary processes (Figs. 27.1 and 27.2); some eyes even showed the presence of thin-walled blood cysts on the ciliary processes (Figs. 27.3 and 27.4). The area affected depended upon the vortex vein occluded, because each vein had a segmental distribution in the ciliary processes as well. When all the veins were occluded, the fine blood vessels under the ciliary epithelium ruptured in the majority of the eyes within a period of from a few minutes to about 3 h after occlusion. This produced bleeding into the posterior chamber which escaped via the pupil and led to a hyphema in the anterior chamber (Figs. 27.1, 27.2, 27.3, 27.4, 27.5, and 27.6). Histological examination of some of these eyes revealed ciliary epithelial cysts projecting into the posterior chamber (Figs. 27.2, 27.3, and 27.5). When three vortex veins were occluded, such a bleeding was seen in four of nine eyes (or in four of five eyes followed up for more than 24 h), while it was seen in two of twelve eyes on occluding two vortex veins, but in none after occlusion of one vortex vein. In eyes with occlusion of two or three vortex veins, the hyphema was not only less common but also less marked than after occlusion of four veins. The hyphema cleared in the former eyes in a week and in the latter in 2–5 weeks. The occurrence of hyphema with occlusion of all the vortex veins was also seen in studies with vortex vein occlusion in rabbits by others [2, 3, 9], but not observed in dogs [12].
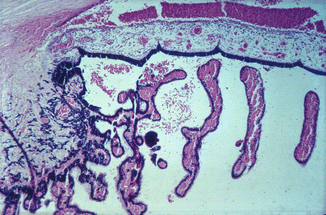
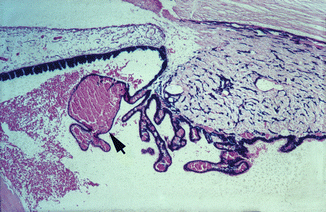
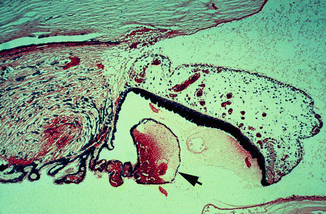
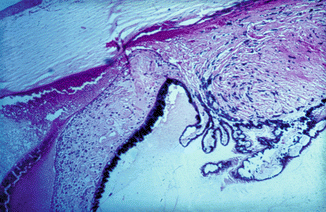
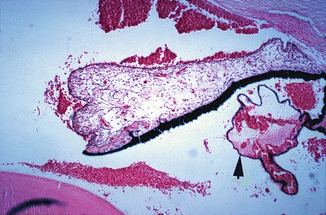
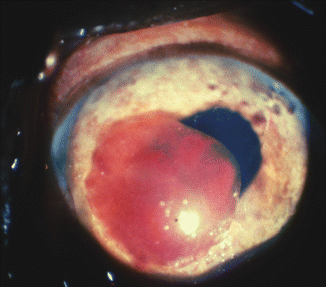

Fig. 27.1
Photomicrograph of anterior segment of rhesus monkey eye, 1–2 h after occlusion of all vortex veins. Hematoxylin and eosin staining. Markedly engorged vessels in ciliary processes, ciliary body, and iris, with hemorrhages in the posterior and anterior chambers (Reproduced from Hayreh and Baines [16])

Fig. 27.2
Photomicrograph of anterior segment of rhesus monkey eye, 1–2 h after occlusion of all vortex veins. Hematoxylin and eosin staining. Markedly engorged vessels in ciliary processes, a blood cyst (arrow), and hemorrhages in the posterior chamber and proteinous exudate in the posterior and anterior chambers (Reproduced from Hayreh and Baines [16])

Fig. 27.3
Photomicrograph of anterior segment of rhesus monkey eye, 1–2 h after occlusion of all vortex veins. Hematoxylin and eosin staining. Markedly engorged vessels in ciliary processes and iris, a blood cyst (arrow), and proteinous exudate in the posterior and anterior chambers (Reproduced from Hayreh and Baines [16])

Fig. 27.4
Photomicrograph of anterior segment of rhesus monkey eye, 1–2 h after occlusion of all vortex veins. Hematoxylin and eosin staining. Markedly distended ciliary processes, hemorrhage in the anterior chamber, and proteinous exudate in the posterior and anterior chambers (Reproduced from Hayreh and Baines [16])

Fig. 27.5
Photomicrograph of anterior segment of rhesus monkey eye, 1–2 h after occlusion of all vortex veins. Hematoxylin and eosin staining. Markedly engorged vessels in ciliary processes; a blood cyst (arrow); hemorrhages in the posterior chamber, pupil, and anterior chamber; and proteinous exudate in the posterior and anterior chambers and pupil (Reproduced from Hayreh and Baines [16])

Fig. 27.6
Photograph of the anterior segment shows a large blood clot in the anterior chamber and engorged vessels in the iris
In all the eyes of this series, immediately after occlusion, and before the appearance of any hyphema, thick protein-rich fluid extended from the ciliary processes in the sector drained by the occluded vortex vein (Figs. 27.2, 27.3, and 27.4); this exudate coated the anterior surface of the lens. In the anterior chamber, the protein-rich aqueous resulted in the formation of a gelatinous cobweb-like deposit in its dependent part (Fig. 27.7). It would thus appear that a marked rise in blood pressure in the capillaries of the ciliary processes results in the outpouring of protein-rich aqueous. This was due to the breakdown of the blood-aqueous barrier. Soon after the injection of intravenous fluorescein, profuse leakage of fluorescein, from the involved sector only, was seen; this was at first localized to the involved sector of the pupil and anterior chamber but progressively became more prominent and diffuse (Fig. 27.7). The blood, protein-rich aqueous, and fluorescein from the posterior chamber passed through the pupil on to the anterior lens surface and anterior chamber, but none was seen in the vitreous or on the posterior lens surface. The vitreous in all eyes was clear. On follow-up, the protein-rich aqueous cleared with the return to normal of the uveal circulation.
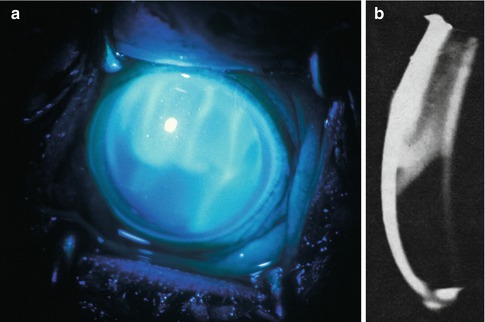

Fig. 27.7
Fluorescein photographs of the anterior segment of an eye 21 h after occlusion of superior temporal and nasal vortex veins in a rhesus monkey (about 10–15 min after intravenous injection of fluorescein) (Reproduced from Hayreh and Baines [16]). (a) Ordinary view shows fluorescein-stained proteinous leakage in the upper part of anterior chamber, as well as seepage of fluorescein from the posterior chamber into the anterior chamber. (b) Slit lamp photograph shows proteinous exudate in the upper part of the anterior chamber, corresponding to that seen in (a)
Depth of Anterior Chamber Findings
The iris-lens diaphragm moved forward significantly in eyes with occlusion of all or three vortex veins, because there was an increase in the volume of the posterior compartment of the eye, due to marked venous congestion in the choroidal vascular bed. This resulted in a shallow anterior chamber – shallower after occlusion of all the vortex veins than after three veins (Fig. 27.8). On follow-up, in three of six eyes with all the vortex veins occluded, the anterior chamber was found to be absent after the hyphema had cleared and it never reformed, while in the remaining three eyes, the depth of the anterior chamber became nearly normal after the hyphema had cleared. In eyes with occlusion of one or two vortex veins, no significant change in the depth of the anterior chamber was noticed. The findings of Koster [9] were similar.


Fig. 27.8
Slit lamp photograph about 15 min after occlusion of all vortex veins shows a very shallow anterior chamber (present only in the pupillary region) (Reproduced from Hayreh and Baines [16])
Intraocular Pressure Findings
As mentioned above, vortex vein occlusion was originally designed to produce raised intraocular pressure and glaucoma [1–11]. In my series [16], eyes with occlusion of all or three vortex veins showed a marked rise in the intraocular pressure immediately after occlusion – about 40–60 mmHg after occlusion of all the vortex veins and about 30–40 mmHg after occlusion of three vortex veins – but no significant rise in intraocular pressure was recorded after occlusion of two or one. This rise in pressure was not related to hyphema, because the intraocular pressure rose as much in eyes without as those with hyphema. Similarly, the protein-rich aqueous seemed to play no role in the rise in intraocular pressure, because the eyes with occlusion of one or two vortex veins had a large amount of gelatinous deposit in the anterior chamber without any rise in the intraocular pressure. In the eyes with elevated intraocular pressure, the angle of the anterior chamber was very narrow, and it could have been blocked in most of them by a forward shift in the iris-lens diaphragm and engorgement of the ciliary body and iris, because of marked venous congestion in the entire uveal tissue.
On the day after the occlusion, the intraocular pressure was either the same as or even higher than the immediate post-occlusion pressure. After 1 week a fall in the intraocular pressure had set in, so that it returned to near normal in eyes with occlusion of all the vortex veins and to unrecordable low levels in eyes with occlusion of three vortex veins; in the eyes with occlusion of all the veins, intraocular pressure then dropped to an unrecordable level in 2 weeks. Having reached the lowest level, the intraocular pressure started to recover; this occurred within 2 weeks after occlusion of three vortex veins. In eyes with occlusion of all the vortex veins, however, the recovery of the intraocular pressure depended upon the state of the anterior chamber, i.e., if the anterior chamber was absent, the eye had permanent marked hypotony, but if the anterior chamber was present, the recovery started after an interval of more than 2 weeks. The intraocular pressure in these eyes did not recover completely but remained subnormal during the period of follow-up. Histological studies showed evidence of atrophy of the ciliary processes and ciliary body in these eyes (Figs. 27.9 and 27.10).
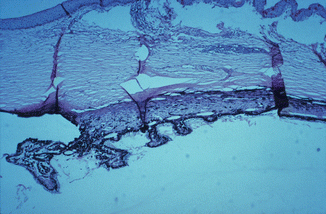
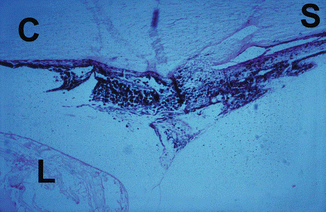

Fig. 27.9
Photomicrograph of the anterior segment of a rhesus monkey eye shows degenerative changes in the ciliary body and processes after occlusion of all the vortex veins. Hematoxylin and eosin staining

Fig. 27.10
Photomicrograph of the anterior segment of a rhesus monkey eye shows marked degenerative changes in the ciliary body and processes after occlusion of all the vortex veins. C cornea, L lens, S sclera; hematoxylin and eosin staining (Reproduced from Hayreh and Baines [16])
Iris Findings
The iris was markedly hyperemic soon after the occlusion of all or three vortex veins (Fig. 27.6). With occlusion of one to three vortex veins, there was sectoral hyperemia of the iris corresponding to the occluded vortex veins. Other authors reported similar findings [2, 9, 12].
On follow-up, three eyes with all the veins occluded and an absent anterior chamber had anterior and posterior synechiae with iris atrophy and iris neovascularization (Fig. 27.11), the clinical appearance resembling that of an eye with long-standing chronic anterior uveitis or anterior segment ischemia. In one of the eyes with three vortex veins occluded, posterior synechiae and sectoral iris atrophy were present. Koster [9] also found these changes in eyes after occlusion of all the vortex veins in rabbits.
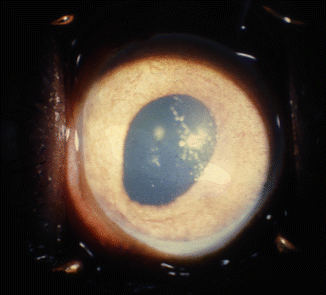

Fig. 27.11
Photograph of the anterior segment shows dilated pear-shaped pupil, iris, and angle neovascularization, with peripheral anterior synechia after occlusion of all vortex veins
Lens Findings
In all the eyes, the lens, soon after occlusion of the various vortex veins, had a proteinous deposit on its anterior surface (Fig. 27.7). During a follow-up period of up to 4 weeks, the deposit cleared without leaving any lens abnormalities in any eye except those with all the vortex veins occluded. During follow-up, all the eyes with all the vortex veins occluded showed pigment deposit on the anterior lens surface; in three of these eyes with an absent anterior chamber and permanent ocular hypotony, the lens became completely cataractous within 5 weeks (Fig. 27.12). The changes appeared at first in the anterior cortex of the lens and later resulted in a complete cataract 14, 19, 38, and 90 days after the occlusion. The cataractous change is evidently secondary to impaired nutrition due to degeneration of the ciliary epithelium and ciliary body, as shown by histological studies.
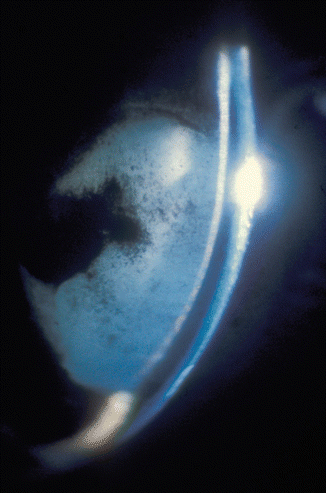

Fig. 27.12
Slit lamp photograph 91 days after occlusion of all vortex veins shows a very shallow anterior chamber (present only in the pupillary region) and cataractous lens. Figure 8 shows the same eye 15 min after the occlusion (Reproduced from Hayreh and Baines [16])
Corneal Findings
Epithelial edema was seen when the intraocular pressure was very high, in eyes with all or three vortex veins occluded. On follow-up, stromal clouding and vascularization by radial vessels from the limbus were seen in three eyes with all the vortex veins occluded. Two of these had no anterior chamber. Koster [9] also noticed a similar corneal vascularization after occlusion of all the vortex veins and likened it to that of interstitial keratitis, with a gradually progressive pannus from the limbus toward the center. I feel that the corneal changes are secondary to the changes in the intraocular pressure and anterior chamber.
The degenerative changes seen in the lens and anterior segment of the eye in these monkeys (particularly in those with occlusion of all the vortex veins) very much resemble those seen in patients with long-standing anterior uveitis or anterior segment ischemia. Almost all the pathologic changes in the anterior segment of the monkey eyes depend upon the changes in the anterior chamber (e.g., protein-rich aqueous, hyphema, shallow or absent anterior chamber), which in turn are secondary to marked disturbances of the uveal circulation associated with the changes in the ciliary processes and the forward displacement of the iris-lens diaphragm, by engorgement of the posterior uveal vascular bed increasing the volume of the posterior segment.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


