Chapter 124 Vitreous, Retinal, and Choroidal Biopsy
Introduction
The diagnosis of posterior segment infectious or inflammatory disease is based primarily on clinical examination. In the majority of cases, the slit-lamp biomicroscopic and ophthalmoscopic appearance of the vitreous, retina, and choroid is diagnostic of a specific disorder, especially when combined with the findings from ancillary tests such as fluorescein angiography, ocular echography, and radiologic and serologic tests.1
However, not all patients have the characteristic clinical features or classic appearance of a specific disease, and the etiology of their disorder can be a diagnostic dilemma. The inability to make a specific diagnosis has been reported in up to 33% of patients who present with intraocular inflammation.2 Moreover, some infectious and malignant processes are manifested first, or only, in the eye, in which case the diagnostic yield from systemic testing is low. This is often the situation in patients with atypical malignancy in the eye, chronic low-grade endophthalmitis after cataract surgery, intraocular inflammation of atypical viral, bacterial, or fungal origin, and some types of progressive retinitis, choroiditis, and pigment epitheliopathies. In these patients, particularly if they are immunocompromised or if one of the possible diagnoses is malignancy, the disease process may threaten not only vision but also life. Intraocular biopsy provides affected ocular tissue for culture, examination, and other tests to help with therapeutic decision-making.3 In most of these cases, therapeutic intervention will be either antimicrobial, antineoplastic, or anti-inflammatory.4,5
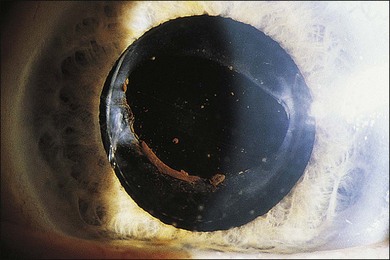
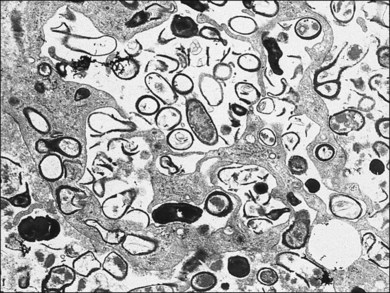
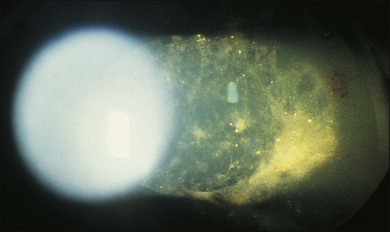
A number of factors in recent years have brought about an increased demand for tissue diagnosis in posterior segment inflammatory disease. One of these is the rise in the number of individuals who have been iatrogenically immunosuppressed as a result of systemic disease, malignancy, or organ transplantation. In addition, the frequent use of intravitreal steroids has also led to the report of opportunistic ocular infections in immunocompetent individuals.6 The clinical presentation of ocular inflammatory disease may be variable and atypical in these immunocompromised patients; the possible etiologic agents are numerous and diverse, and serologic testing is frequently misleading. Another factor increasing the indication for intraocular biopsy is the technologic advance of modern cataract surgery. With phacoemulsification and in-the-bag intraocular lens placement, every year millions of patients are left with most of their capsular bags intact, forming small potential reservoirs in which, rarely, sequestered organisms of normally low-grade pathogenicity can multiply. Clinicians are confronted with a number of patients whose chronic, smoldering, postoperative endophthalmitis is due to organisms such as Candida parapsilosis or Propionibacterium acnes (Figs 124.1–124.5), which cannot be adequately treated until a tissue diagnosis is made. In addition, ongoing advances in laboratory testing continue to improve the diagnostic yield from biopsied material. For example, polymerase chain reaction (PCR) testing, a biochemical technique that allows for the detection of infinitesimal amounts of specific nucleic acids, now permits identification of genetic components of infectious organisms. Finally, advances in vitreoretinal surgical instrumentation and increasing use of 25- and 23-gauge (G) transconjunctival pars plana vitrectomy7 have allowed for possibly safer access and acquisition of ocular tissue for analyses. Table 124.1 lists common indications for ocular biopsy.
Table 124.1 Common indications for biopsy
| Biopsy site | Diagnostic dilemma |
|---|---|
| Vitreous | Endophthalmitis (bacterial or fungal) |
| Intraocular lymphoma | |
| Retinitis with associated vitritis | |
| Retinal | Retinitis (atypical or not responsive to empiric therapy) |
| Choroidal | Tumor |
| Choroiditis (atypical or not responsive to empiric therapy |
The efficacy of ocular biopsy depends partly on the expertise of the surgeon who obtains the specimen and the laboratory personnel who analyze the specimen(s). Before embarking on surgery, harvesting and handling of the specimen should be carefully mapped out in advance. The pathologist should be familiar with ophthalmic disease processes, aware of the diagnoses being considered, and involved at the outset in planning how the specimen will be handled from the moment it is taken – a step that is critical to the success of the procedure.4,8,9
The pathologist’s available investigative methods include, among others, light microscopy, electron microscopy, immunohistochemistry, and PCR. PCR can be used to identify unique DNA and RNA sequences specifically from infectious agents and, occasionally, from other disease processes, such as proliferating monoclonal B lymphocytes in ocular malignant lymphoma.9–13 The laboratories responsible for culture and identification of bacterial, fungal, and viral disease should receive the specimen as soon as possible. It should be plated on a variety of specific media and maintained under the correct (aerobic, anaerobic, temperature) conditions. Plates may need to be specially treated or held for extra time to reveal growth from some of the more fastidious organisms.
Vitreous biopsy
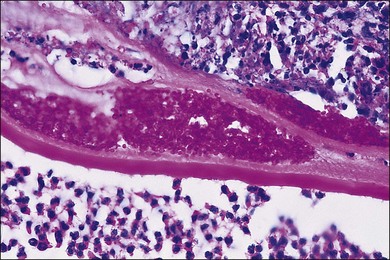
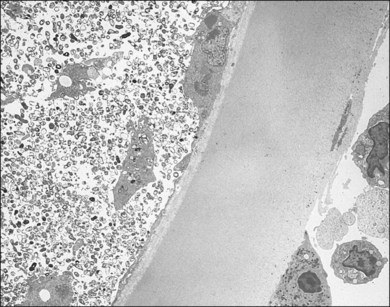
With the evolution of modern vitrectomy techniques, including 23G, 25G, and 27G vitrectomy, removal of vitreous from the eye has become a common and relatively safe procedure.14 A number of different goals are achieved with diagnostic vitrectomy5,15 In vitritis with a possible neoplastic, infectious, or inflammatory cause, diagnostic vitrectomy may provide a definitive diagnosis, thereby allowing appropriate therapy to be instituted.5,8 In some cases of retinitis and choroiditis, a diagnosis can be made from vitreous sampling alone, particularly if marked vitritis is present. The vitrectomy also removes or debulks infectious material within the eye, reduces the intraocular load of inflammatory cells and associated biologically active material and debris, effectively removes material (e.g. retained lens material and blood) that may be part of the pathogenesis of the ocular disorder, allows clinicopathologic correlation in the active stages of disease for some conditions, and helps to define the pathophysiologic basis of many disorders.5 Cytopathologic diagnosis has proved valuable in reported series or case reports of patients with: endogenous and exogenous bacterial and fungal infections (Fig. 124.6); nematode endophthalmitis; inflammatory pseudotumor of the iris and ciliary body; pars planitis; phacoanaphylaxis; intraocular lymphoma; leukemia; metastatic melanoma; metastatic carcinoma; medulloepithelioma of the ciliary body; secondary glaucoma (including blood-induced, phacolytic, and melanomalytic glaucoma); epithelial ingrowth; proliferative vitreoretinopathy, and miscellaneous conditions with vitreous opacities (including chronic inflammation in patients with suspected lymphoma, acute retinal necrosis, birdshot chorioretinopathy, toxoplasmosis, Whipple’s disease, amyloidosis, and asteroid hyalosis).4,5,16,17 Vitrectomy may also help to clear vitreous opacities, remove scaffolding on which postoperative cicatricial proliferation can grow, relieve traction, and allow for improved intraocular penetration of antibiotics.5,15 Table 124.2 lists a sample of vitreous findings in major disease categories.
Surgical technique
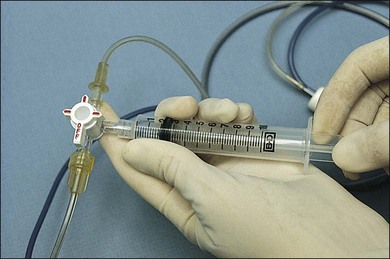
To obtain an undiluted specimen, a variety of vitrectomy techniques (with either 20-, 23-, 25-, or 27G instrumentation) may be used. If a large sample (>0.5 mL) of vitreous is needed, a standard three-port vitrectomy system should be utilized. A 10 mL Luer–Lok syringe is spliced via a three-way stopcock into the aspiration line (Fig. 124.7). Manual aspiration and specimen collection are carried out for retrieval of an undiluted specimen from the most intensely involved area (usually posteriorly, inferiorly, or over the area of retinitis or chorioretinitis).
Alternatively, a vitreous “trap” method may also be performed using two 18G needles, a male adaptor, and a sterile red-topped test tube.18 In the vitreous trap method, the male adaptor is attached to one of the needles, and the needles are attached to the ends of the aspiration tubing. The two needles are then inserted into the test tube. The diagnostic vitrectomy is performed and the pure vitreous sample is directed into the test tube.
After a sufficient amount of sample is obtained, the infusion is turned on and either fluid or air is infused before large choroidals form and the eye begins to collapse.14 The vitrector is withdrawn from the eye, and the vitreous specimen is aspirated into the syringe. Vitrectomy can then be completed. At completion of the surgery, the cassette with the dilute residual vitreous is also sent to the pathology laboratory, where the specimen is concentrated, using a micropore filter, or cytospin smears are made.4,19
If a small sample (≤0.5 mL) of vitreous is needed, a 27G vitrectomy with a single incision may be utilized.20,21 In this technique, the 27G vitrector is inserted perpendicular to the pars plana. Using a cut rate of 800 cuts/minute and an aspiration rate of 600 mmHg, a small amount of undiluted vitreous may be safely removed from the eye without the need of an additional infusion line or endoillumination port. The 27G scleral wound is also self-sealing and does not require sutures.
Histologic technique and preparations
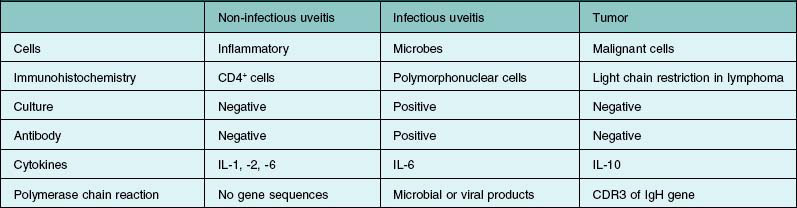
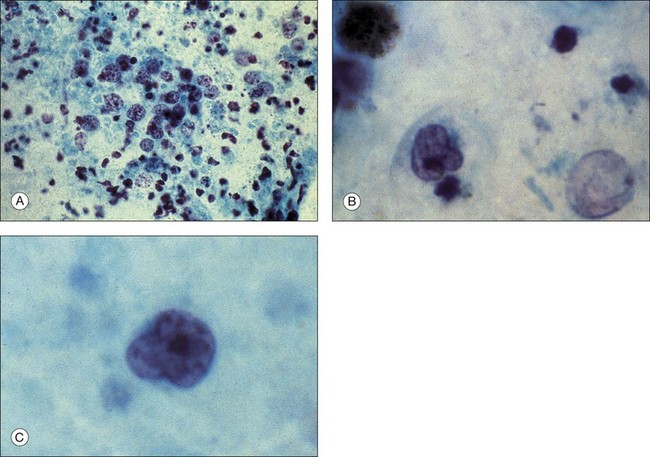
If primary intraocular lymphoma is suspected, obtaining a sufficient vitreous sample for cytologic testing is essential. In addition, consultation with an experienced cytopathologist is recommended because correct identification of lymphoma cells in the biopsy sample is essential for the diagnosis. In addition to characterizing cells from the vitreous biopsy, testing for immunohistochemical markers for leukocytes (e.g., CD45), B cells (e.g., CD 20), and T cells (e.g., CD45RO) can also aid in the diagnosis.14,22,23 Other diagnostic modalities that can be useful include flow cytometry, PCR to detect V–J immunoglobulin gene rearrangements, and measurements of IL-6 and IL-10 in aqueous or vitreous samples.22–24 Table 124.3 summarizes the key laboratory features of several patients who underwent 25G diagnostic vitrectomy for suspected intraocular lymphoma. Additional information on the diagnosis and management of primary intraocular lymphoma is available in Chapter 155, Leukemias and lymphomas.
Results
In cases of infectious endophthalmitis, vitreous samples can often reveal the presence of microbes. Unfortunately, these may not be found in all infectious cases, principally because of suboptimal sampling, inadequate culturing, or pretreatment with antimicrobials that render microbes nonculturable on routine media.10 In the Endophthalmitis Vitrectomy Study, confirmed microbiologic growth was demonstrated in intraocular specimens from 291 of 420 patients (69.3%).25 More recently, PCR testing has been applied to identify bacterial causes of endophthalmitis. Very little tissue is needed for PCR. Usually 50–100 µL samples are sufficient, but even a sample as small as 1 µL can be processed for PCR testing.26 Melo and co-authors evaluated real-time PCR to diagnose bacterial endophthalmitis after cataract surgery and demonstrated that PCR assays were positive in 91% of patients with a clinical diagnosis of endophthalmitis using either aqueous and/or vitreous samples.27 Lohmann and colleagues28 used PCR to analyze 25 patients with late-onset endophthalmitis, and they showed that bacterial DNA (belonging to P. acnes, Staphylococcus epidermis, or Actinomyces israelii) was obtained by PCR from 92% of vitreous samples. By comparison, bacterial culture was positive in only 24% of these cases. PCR can also be helpful for suspected fungal endophthalmitis. Salman and colleagues29 demonstrated that PCR was highly sensitive and specific in the diagnosis of post-traumatic fungal endophthalmitis. In their study of 26 endophthalmitis cases, PCR correctly identified Aspergillus organisms, while culture on blood agar was negative in all cases.
PCR is also a useful tool in the diagnosis of posterior segment infectious uveitis. Harper and colleagues conducted a retrospective review of 133 patients who underwent PCR testing of aqueous or vitreous for possible infectious chorioretinitis.30 PCR tests for herpes virus, varicella zoster virus, and CMV were performed in the majority of cases. The predictive value of positive and negative tests was 98.7% and 67.9% in this population. In addition, an alteration in treatment based on the PCR result led to a resolution in 25 of 26 patients after treatment was changed, suggesting that PCR is a useful test to aid in the diagnosis and management of challenging cases of infectious uveitis.
Several studies have shown that PCR has sensitivities exceeding 90% for varicella-zoster virus (VZV), herpes simplex virus (HSV), and cytomegalovirus (CMV), with specificities in excess of 95% for these organisms.26 Sensitivity approaches 95% in detecting untreated CMV retinitis, dropping to 48% in treated eyes.31–33 Moreover, false-positive rates are low.34 In addition, PCR can aid in detecting antiviral-resistant strains of CMV, which have mutations in the UL97 polymerase gene.26 Information on possible antiviral resistance is essential in the management of viral retinitis and may influence the choice of agent for a particular patient. PCR inhibitor has been detected in normal aqueous and vitreous fluids, but it can be removed by diluting the specimen, by chloroform extraction, or by thermostable DNA polymerases.35
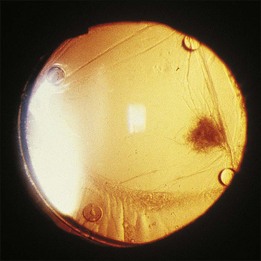
Palexas and colleagues36 reported on 405 consecutive vitreous biopsy cases studied at the Wilmer Eye Pathology laboratory. Vitrectomy had been performed in five categories of eye disease: post-traumatic infections (8.4%); postoperative endophthalmitis (38.5%); endogenous endophthalmitis (6.2%); idiopathic inflammation (25.4%); intraocular neoplasm (14.3%); and miscellaneous (7.2%). Diagnostic vitrectomy was performed in 215 cases (53%) for suspected endophthalmitis. Overall, 60 of these (28.8%) had microbial organisms present, and 36 (16.7%) were culture-positive. In the postoperative endophthalmitis group, microorganisms were observed microscopically in 31 (20%) of 156 eyes, 74% of these Gram-positive. Bacteria were found in six (18%) of 34 post-traumatic cases of endophthalmitis and in 23 (92%) of 25 endogenous cases. Patients with endogenous infections had the most fungal infections. Neoplasms were diagnosed in 58 (14%) of the 405 vitrectomy specimens, the most common being ocular lymphoma in 42 (72%) (Fig. 124.8). However, only 42 (48.3%) of 87 patients clinically suspected of having ocular lymphoma had a vitreous biopsy that was positive for the disease. Other malignancies diagnosed cytopathologically included metastatic squamous cell carcinoma and acute lymphoblastic leukemia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree