10 Vitreous
INTRODUCTION
The transparent gelatinous vitreous humor fills most of the interior of the eye. The most delicate connective tissue in the body, the vitreous humor is composed of a framework of thin, randomly oriented, unbranched fibrils of type II collagen and is rich in hyaluronic acid, an extremely large, negatively charged, hydrophilic polysaccharide. Hyaluronic acid is named after the hyaloid body, an older term for the vitreous humor (both hyaloid and vitreous mean glassy).
The collagenous framework of the vitreous humor adheres to the internal limiting membrane (ILM) of the retina, the periphery of the optic disc, and most firmly to the 2-mm wide vitreous base that straddles the ora serrata. The anterior attachment of the vitreous is particularly firm. Relatively severe trauma that typically disrupts the ciliary epithelium is required to detach or avulse the vitreous humor from the vitreous base. In contrast, the posterior attachments of the vitreous are tenuous. Posterior vitreous detachment affects many individuals after age 55 years and is found in more than 60% of the population in the eighth decade (Fig. 10-1). Patients usually complain of the abrupt onset of floaters and occasionally light flashes that signify vitreoretinal traction. The peripapillary condensation of the vitreous framework may be evident clinically or macroscopically after detachment as a Weiss ring (Fig.10-1B).
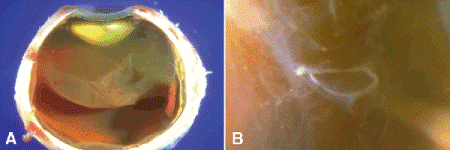
Fig. 10-1. Posterior vitreous detachment. A. Mild opacification by protein accentuates the vitreous. The vitreous framework has detached from the inner retina posteriorly. It remains attached to the anterior vitreous base that straddles the ora serrata. The attachment there is very strong. The anterior chamber is flat, and part of the retina is shallowly detached by a gelatinous exudate rich in lipid. B. Weiss ring, posterior vitreous detachment. The white ring represents the peripapillary condensation of the vitreous framework, which has separated from the optic disc during a posterior vitreous detachment.
The vitreous humor contributes to visual loss in two basic ways. First, the vitreous framework serves as a growth scaffold for cellular proliferation, and resultant vitreoretinal traction plays an important role in the pathogenesis of many retinal detachments. Second, under pathologic conditions, the transparent medium may be opacified by the accumulation of a variety of materials including blood, acute or chronic inflammatory cells, tumor cells, iridescent particles, and amyloid.
Vitreoretinal adhesions and traction cause retinal holes or breaks that predispose to rhegmatogenous retinal detachment. New blood vessels, cells, and fibrous tissue also proliferate on exposed surfaces of the vitreous framework. Tractional retinal detachment is caused by subsequent organization and contraction of the vitreous. The organization of vitreous hemorrhage or inflammation also stimulates vitreous traction.
Detachment of the vitreous and retina exposes new surfaces that can serve as a substrate for cellular growth. The contraction of delicate glial membranes on the inner retinal surface causes sinuous folds in the ILM (surface wrinkling or cellophane retinopathy) that distort vision (Fig. 10-2). Proliferation of myofibroblasts, retinal pigment epithelial (RPE) cells, and glial cells forms fibrocellular membranes on retinal surfaces exposed by retinal detachment. These membranes can bind the detached retina into a folded mass that may be impossible to reattach surgically. Analogous cellular proliferation can also occur on the detached posterior face of the vitreous, inducing fibrosis. Somewhat analogous to “in vivo tissue culture,” this process is called proliferative vitreoretinopathy (PVR) or massive periretinal proliferation (MPP). PVR is an important cause of inoperable retinal detachment, or recurrent detachment after reattachment surgery (Fig.10-3).
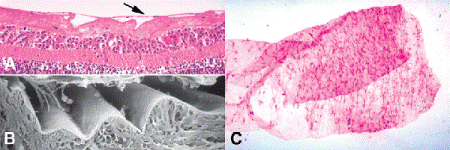
Fig. 10-2. Epiretinal gliosis (surface wrinkling retinopathy). A. Arrow denotes delicate membrane of glial cells on inner retinal surface after posterior vitreous detachment. Contraction of the membrane has caused folds in the internal limiting membrane (ILM). B. SEM shows folds in ILM. Glial membrane has partially detached. C. Vitreoretinal membrane in vitrectomy specimen. Presumed glial cells form a subconfluent membrane on the surface of a condensed sheet of vitreous. The cells have bland spindled nuclei. The indication for vitrectomy was macular pucker. (A. H&E ×100, B. SEM ×640, C. Millipore filter preparation, H&E ×50)
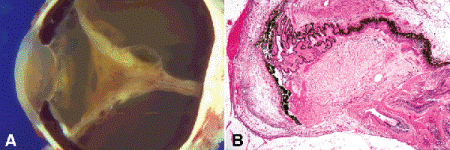
Fig. 10-3. Anterior variant of proliferative vitreoretinopathy (PVR). A. Chronic retinal detachment with macrocysts persists after vitrectomy. PVR membrane extends anteriorly from inner retinal surface to cover ciliary body and iris. Traction draws iris posteriorly. B. Fibrosis of residual vitreous overlying ciliary body exerts traction on retina and produces anterior loop retinal detachment. Iris is drawn posteriorly. (B. H&E ×10)
Blood is the substance that opacifies the vitreous most frequently. Common sources of vitreous hemorrhage include trauma, proliferative diabetic retinopathy, and other disorders with vitreoretinal neovascularization, tractional retinal tears, and posterior vitreous detachment. Rarer causes include intraocular tumors, exudative age-related maculopathy, and subarachnoid hemorrhage (Terson syndrome).
Persistent vitreous hemorrhage is a major indication for vitreous surgery. Blood and other opacities in the vitreous are removed by a surgical procedure called vitrectomy that employs miniaturized cutting and aspiration instruments, intraocular illuminators, and laser photocoagulators, which are inserted through small incisions in the pars plana. The excised vitreous is replaced with saline, which is introduced through an infusion port.
Vitrectomy specimens can be processed using a variety of cytologic techniques including cytocentrifugation, liquid-based monolayer cytology, or Millipore filtration. If particulates are numerous, the fluid can be centrifuged and the pellet embedded in paraffin as a cell block for histology. The latter is particularly helpful if specimen evaluation requires special stains for microorganisms or immunohistochemistry.
Microscopic examination of vitrectomy specimens from patients with chronic vitreous hemorrhage discloses blood and blood breakdown products including erythroclasts or erythrocyte ghost cells, hemoglobin spherules, and macrophages laden with ghost cells and golden-brown granules of the blood pigment hemosiderin (Fig. 10-4). In very chronic cases, the blood breakdown products occasionally stimulate a granulomatous inflammatory foreign body response. Hemoglobin spherules may be quite large if the vitreous blood originates from the choroid. Clinically and macroscopically, chronic vitreous hemorrhage typically is yellow-ochre in color and may appear as an “ochre membrane” (Fig. 4-1A). If the anterior face of the vitreous is ruptured, ghost cells and hemosiderin-laden macrophages may enter the anterior chamber and block the trabecular meshwork, causing secondary open-angle glaucoma (ghost cell or hemolytic glaucoma) (Fig. 8-14B).
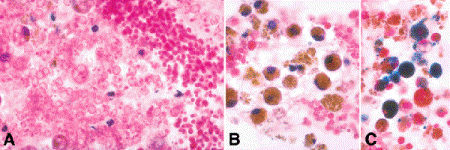
Fig. 10-4. Chronic vitreous hemorrhage. A. Numerous erythrocyte ghost cells or erythroclasts are seen below. Ghost cells are the empty cell membranes of erythrocytes that have lost their hemoglobin-rich cytoplasm. The eosinophilic bodies above are hemoglobin spherules. Macrophages that have ingested ghost cells and other blood breakdown products also are present. B. Hemosiderin-laden macrophages, chronic vitreous hemorrhage. The yellow-brown pigment in the macrophages is the blood breakdown product hemosiderin. C. The pigment in some of the macrophages in this chronic vitreous hemorrhage stains positively (blue) for iron. Many large hemoglobin spherules are present. (A. H&E ×250, B. H&E ×250, C. Perl stain for iron, ×250)
The vitreous is rapidly opacified by an intense influx of polymorphonuclear leukocytes and macrophages in acute purulent endophthalmitis (Fig. 3-12). A vitreous abscess composed of polys forms in neglected cases or virulent infections. Some of the inflammatory cells in a vitreous abscess may be arranged in a linear fashion, reflecting the alignment of the cells along the fibrils of the collagenous vitreous framework. A large solitary vitreous abscess characterizes bacterial endophthalmitis. Fungal endophthalmitis, which generally is a more indolent infection, typically is marked by the presence of multiple smaller vitreous microabscesses (Fig. 3-13). Digestive enzymes released from degenerating polys in a vitreous abscess can cause extensive necrosis of intraocular tissues including retinal destruction (Fig. 3-12B). Vitrectomy is used tosurgically “drain” the vitreous abscess in some patients with endophthalmitis. If vitrectomy is not performed, the vitreous abscess may be organized by an ingrowth of granulation tissue from the ciliary body and choroid. Eventually, a dense collagenous scar elaborated by fibroblasts and metaplastic RPE cells fills the vitreous cavity.
Remnants of the embryonic hyaloid vascular system are seen by patients as innocuous vitreous opacities called floaters or muscae volitantes (“flying flies”). Patients frequently complain that they see a spot like a moving insect in their peripheral visual field that darts away when they move their eyes. Floaters are especially prevalent in myopes and are caused by syneresis or degeneration of the vitreous framework in the enlarging myopic eye. When light flashes accompany floaters, vitreoretinal traction is present and retinal holes must be excluded by a careful and expedient ophthalmoscopic examination.
Iridescent particles accumulate in the vitreous humor in two disorders: asteroid hyalosis (AH) and synchysis scintillans. Asteroid hyalosis (Benson disease) is relatively common, occurring in about 2% of the population (Fig. 10-5). AH was once called asteroid hyalitis, but the name was changed when it became apparent that the disorder is degenerative and not inflammatory in nature. Ironically, histopathologic examination occasionally discloses asteroid bodies enveloped by foreign body giant cells.
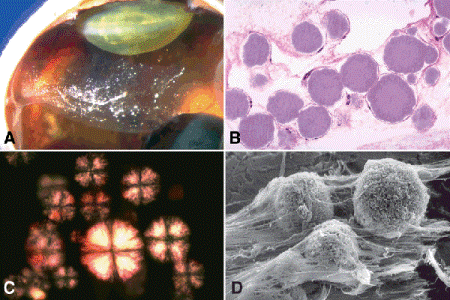
Fig. 10-5. Asteroid hyalosis. A. Numerous calcified spherules impart a starry sky appearance to posteriorly detached vitreous. The asteroid hyalosis was an incidental finding in an eye enucleated for uveal melanoma. B. Asteroid bodies appear as grayish blue spherules in routine H&E sections. They are attached to the vitreous framework. C. Intact asteroid bodies in vitrectomy specimen show characteristic Maltese cross pattern of birefringence during polarization microscopy. D. Scanning electron microscopy (SEM) shows attachment of asteroid bodies to vitreous fibrils. (B. H&E ×250, C. Millipore filter preparation, H&E with crossed polarizers ×250, D. SEM ×640)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


