Purpose
To compare mortality risk between cataract surgical patients with corrected and persistent visual impairment.
Design
Cohort study.
Methods
A total of 1864 consecutive patients, aged ≥64 years, undergoing phacoemulsification surgery at Westmead Hospital were followed annually for 5 years postoperatively. Visual impairment status in the surgical eye was categorized as none (presenting visual acuity [VA], ≥20/40), mild (VA <20/40-20/60), or moderate-severe (VA <20/60). All-cause mortality was obtained from the Australian National Death Index.
Results
Of 901 patients with moderate-severe visual impairment before surgery, 60.4% (n = 544), 15.5% (n = 140), and 24.1% (n = 217) had no, mild, or moderate-severe visual impairment in the surgical eye, respectively, 1 month postoperatively. Age-standardized 5-year mortality rates were nonsignificantly lower in patients with either mild (24.7%, 95% confidence interval [CI] 16.5%-32.9%) or no visual impairment (24.1%, 95% CI 19.9%-28.4%) post surgery compared to that in patients whose moderate-severe visual impairment persisted (30.6%, 95% CI 23.3%-37.9%). After adjusting for age, sex, smoking, body mass index, and individual comorbid conditions, such as hypertension, diabetes, angina, myocardial infarction, stroke, transient ischemic attack, and kidney disease, patients with no visual impairment 1 month postoperatively had a lower mortality risk (HR 0.73, 95% CI 0.52-1.01) compared to those with persistent moderate-severe visual impairment after surgery. This finding was significant (HR 0.71, 95% CI 0.51-0.99) after additional adjustment for number of medications taken (continuous variable) and number (≥3 vs <3) of comorbid conditions.
Conclusion
Correcting moderate-severe visual impairment in older patients with phacoemulsification surgery was associated with a lower mortality risk, compared to surgical patients whose visual impairment persisted postoperatively.
Previous studies have reported that objectively measured visual impairment (visual acuity [VA] ≤20/40) was associated with a 20%-90% increased mortality risk. The Salisbury Eye Evaluation (SEE) project reported a 53% reduction in 6-year mortality risk in participants who had a gain of ≥2 lines in binocular presenting VA 2 years after baseline examinations compared to those whose presenting VA was unchanged, after multivariable adjustment. Approximately half of those with a presenting VA gain had undergone cataract surgery during the follow-up period while the rest were likely to have had refractive correction.
The Blue Mountains Eye Study (BMES) found that older (≥49 years) persons with cataract-related visual impairment corrected by cataract surgery had a 40% reduction in 15-year mortality risk after multivariable adjustment, compared to those who remained visually impaired because of cataract and did not have surgery. We aimed, using an independent, older cataract surgical cohort, to assess a similar question to the BMES report, that correcting moderate-severe visual impairment using phacoemulsification surgery is associated with better survival among older people with visual impairment attributable to cataract, compared to that in patients who remained visually impaired after their cataract surgery.
Methods
The Australian Prospective Cataract Surgery and Age-related Macular Degeneration study is a prospective cohort study of patients, aged ≥64 years, recruited for cataract surgery at Westmead Hospital, Western Sydney, Australia. Westmead Hospital is a major public, tertiary hospital that provides public services to the Western Sydney region. All patients provided written informed consent. The study adhered to the tenets of the Declaration of Helsinki and was approved by the Human Research Ethics Committees of the University of Sydney, the Sydney West Area Health Service, and the Australian Institute of Health and Welfare. Details of the Australian Prospective Cataract Surgery and Age-related Macular Degeneration study are described elsewhere.
Participants
During 2004-2007, 1864 consecutive patients had phacoemulsification surgery at Westmead Hospital. They were recruited into the study either preoperatively or shortly after surgery. The eye that was to be operated on during the study recruitment period was defined as the surgical eye of the patient. The better eye was defined as either the surgical eye, if fellow eye data were not available, or the better of the 2 eyes, irrespective of surgical eye status, with regard to presenting VA levels. Study participants had VA assessment (with and without pinhole) and retinal photography at preoperative and 1-, 6-, and 12-month postoperative visits.
Measurements and Definitions
In brief, presenting VA in both eyes was assessed using a retroilluminated logarithm of the minimal angle of resolution (logMAR) chart read at 2.4 m with current distance glasses, if worn. Presenting VA was defined as the number of letters read correctly from zero to 70 and converted to a Snellen fraction. Presenting visual impairment was defined using either the surgical eye or the better of the 2 eyes and categorized as none (presenting VA ≥20/40), mild (presenting VA <20/40-20/60), or moderate-severe (presenting VA <20/60). For eyes reading fewer than 54 letters (<20/40) using current distance correction, VA was further assessed with a pinhole aperture. Under-corrected VA was defined as presenting VA that was corrected with a pinhole aperture by ≥3 lines. Pseudophakic status was confirmed from photographs of the anterior segment of the eye, taken using a Canon CF-60DSi digital fundus camera (Canon Inc, Tokyo, Japan). Retinal photographs, obtained with the digital camera, were assessed by a trained grader for the diagnosis of retinal conditions that could have contributed to poor vision postoperatively. Adjudication of the photographs was obtained by a senior researcher (J.J.W.) and a retinal specialist (P.M.). The International Classification and Grading System for Age-related Maculopathy and Age-related Macular Degeneration and the modified Early Treatment Diabetic Retinopathy Study classification system were closely followed for the diagnosis of these 2 conditions.
Demographic and anthropomorphic data were collected at baseline visits. Regular medications and history of eye disease and comorbid conditions, including hypertension, diabetes mellitus, angina, myocardial infarction, transient ischemic attack, stroke, and kidney disease, were recorded during interview and confirmed from hospital patient medical records. Categorized number of comorbid conditions (≥3 vs <3) included any of the following medical conditions: hypertension, diabetes mellitus, angina, myocardial infarction, transient ischemic attack, stroke, and kidney disease. Body mass index (BMI) at baseline was calculated as weight in kilograms divided by the square of height in meters, with BMI less than 20 kg/m 2 defined as low. Current smokers included persons who had stopped smoking within the past year.
The main outcome was death since the preoperative or 1-month-postoperative examinations (if preoperative data were not available), up to October 13, 2011 (mean follow-up period 5.7 years, range 4-8 years), information that was obtained via data linkage to the Australian National Death Index (NDI) dataset. A probabilistic record linkage package was used to cross-match the 2 datasets, based on characteristics that included given and family names, sex, date of birth, and the name of the suburb of residence. Causes of death were provided by the NDI, which records causes documented on death certificates, based on the International Classification of Diseases, Ninth Revision, and the International Statistical Classification of Diseases, Tenth Revision. Australian NDI data have been validated previously, with an estimated sensitivity of 93.7%-95.0% and specificity of 100% for all-cause deaths. Seven patients identified as having nonexact matching could not be ascertained from either their medical records or family members, and thus were excluded, leaving 1857 patients for study analyses.
Statistical Analysis
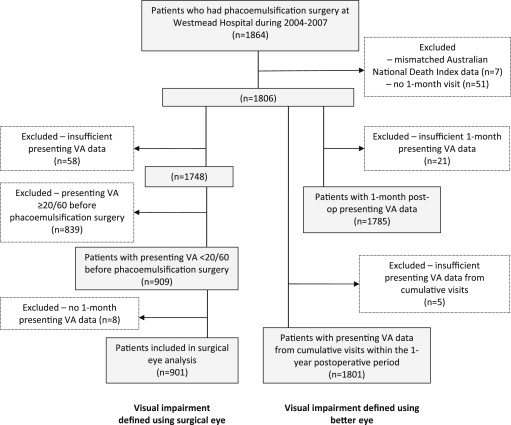
After excluding 839 participants (45.2% of 1857) with preoperative presenting VA ≥20/60 and 117 (6.3% of 1857) with VA data missing at either the preoperative or 1-month-postoperative visits, 901 participants with preoperative presenting VA <20/60 were included in the primary analysis of this report, for the association between visual impairment correction and improved survival among cataract surgical patients ( Figure , left panel). Presenting VA, or VA using the participants’ current distance correction, was used in all analyses as this represents the daily vision that these persons used in their everyday activities.
Statistical software (SAS version 9.2; SAS, Cary, North Carolina, USA) was used to perform survival analyses. Five-year mortality rates by visual impairment status at 1-month-postoperative visits, defined using the surgical eye, were calculated after direct age standardization to the Australian Prospective Cataract Surgery and Age-related Macular Degeneration study population (n = 1857).
These primary analyses were performed to assess the association between visual impairment correction via cataract surgery and 5-year survival. Mortality risk was assessed and compared between patients with mild or no visual impairment in the surgical eye 1 month postoperatively and those remaining with moderate-severe visual impairment in the operated eye at the same visit, using Cox proportional hazard regression models. Patients in these visual impairment groups were censored to either the census date (October 13, 2011) or date of death. Three Cox regression models were used: Model 1 adjusted for baseline age and sex; Model 2 additionally adjusted for smoking status: current (including persons who had stopped within the past year) vs past or never smokers, low BMI, and history of hypertension, diabetes, angina, myocardial infarction, stroke (including transient ischemic attack), and kidney disease; Model 3 adjusted for all variables contained in the second model plus indicators of poor general health and frailty: the number of medications taken (as a continuous variable) and the number (≥3 vs <3) of comorbid conditions. Co-variables were checked for violation of the proportional hazard assumption using Schoenfeld residuals. As age was found to violate this assumption, we defined 3 age-group strata (64-69, 70-79, and 80+ years) in the Cox models. Mortality hazard ratios (HRs) are presented with 95% confidence intervals (CIs).
Supplementary analyses were performed comparing mortality risk between patients with no or mild visual impairment after cataract surgery to those with persisting moderate-severe visual impairment, defined using the better of the 2 eyes, irrespective of whether the assessed eye was the surgical eye or not. Two different VA criteria were used in the supplementary analyses; first, using VA that was measured 1 month postoperatively, and second, using VA that was measured from the latest visit within the 12-month-postoperative period. These supplementary analyses aimed to assess the visual impairment–mortality association in a larger sample of participants who had VA data out of the 1857 surgical patients, using the better of the 2 eyes regardless of whether it was the surgical eye or not. Analyses using both 1-month- and cumulative 12-month-postoperative visual impairment status are to confirm the robustness of the findings. Of the 1857 participants, 1785 (96.1%) had VA data for the better eye 1 month postoperatively and 1801 (97.0%) had VA data from the latest visit within the 12-month follow-up period ( Figure , right panel).
Results
Of the 901 patients with VA data in the surgical eye 1 month postoperatively, 60.4% (n = 544), 15.5% (n = 140), and 24.1% (n = 217) had no visual impairment, mild visual impairment, and moderate-severe visual impairment, respectively. Patients with no visual impairment were younger, had a higher proportion of women, and had a lower prevalence of diabetes than the group with moderate-severe visual impairment ( Table 1 ). There were no other significant differences in the proportions with chronic conditions, with ≥3 comorbid conditions, or taking ≥6 medications ( Table 1 ). Table 2 shows the known causes of visual impairment among the 217 cases with persistent moderate-severe visual impairment shortly after surgery. Of the 901 surgical eyes, 64.9% (n = 585) and 33.5% (n = 302) were the better and worse of the paired eyes, respectively, and 1.6% (n = 14) were the only eye with data available.
| Characteristics | Visual Impairment Status in Surgical Eye at the 1-Month-Postoperative Visit | |||||
|---|---|---|---|---|---|---|
| N a | Presenting VA <20/60 N = 217 | Presenting VA <20/40 to ≥20/60 N = 140 | Presenting VA ≥20/40 N = 544 | |||
| n (%) | n (%) | P Value b | n (%) | P Value c | ||
| Age, mean (SD), y | 901 | 76.1 (6.7) | 75.2 (6.9) | .21 g | 74.4 (5.8) | .0001 g |
| Female | 901 | 111 (51.2) | 72 (51.4) | .87 h | 320 (58.8) | .02 h |
| Previous cataract surgery | 901 | 53 (24.4) | 38 (27.1) | .56 | 150 (27.6) | .38 |
| Current smoker d | 895 | 23 (10.7) | 22 (15.8) | .15 | 84 (15.6) | .08 |
| Body mass index <20 kg/m 2 | 777 | 17 (9.1) | 7 (6.3) | .44 | 30 (6.3) | .20 |
| Hypertension | 901 | 134 (61.8) | 92 (65.7) | .45 | 322 (59.2) | .52 |
| Diabetes mellitus | 901 | 82 (37.8) | 44 (31.4) | .22 | 144 (26.5) | .002 |
| Cardiovascular disease e | 901 | 41 (18.9) | 39 (27.9) | .05 | 126 (23.2) | .20 |
| Stroke f | 901 | 36 (16.6) | 19 (13.6) | .44 | 65 (12.0) | .09 |
| Kidney disease | 901 | 21 (9.7) | 18 (12.9) | .35 | 55 (10.1) | .86 |
| ≥3 comorbid conditions | 901 | 98 (45.2) | 62 (44.3) | .87 | 224 (41.2) | .32 |
| ≥6 regular medications | 901 | 71 (32.7) | 53 (37.9) | .32 | 172 (31.6) | .77 |
b Age-sex-adjusted P value for comparison between “VA <20/60” and “20/60≤ VA <20/40.”
c Age-sex-adjusted P value for comparison between “VA <20/60” and “VA ≥20/40.”
d Includes participants who gave up smoking less than a year prior to the baseline examinations.
e Includes either angina or myocardial infarction.
f Includes either stroke or transient ischemic attack.
| Characteristic | Frequency, % (N) (N = 217) |
|---|---|
| Nonoperative | |
| Under-corrected VA a | 33.2 (72) |
| AMD | 21.2 (46) |
| Glaucoma | 16.1 (35) |
| Diabetic retinopathy | 13.4 (29) |
| Epiretinal membranes | 8.3 (18) |
| Myopic retinopathy | 1.4 (3) |
| Optic disc atrophy | 1.4 (3) |
| Previous retinal tear | 0.9 (2) |
| Corneal lesion | 0.9 (2) |
| Intraoperative or postoperative | |
| Posterior capsular tear | 5.5 (12) |
| Anterior vitrectomy | 6.0 (13) |
| Iris trauma | 3.2 (7) |
| Any intraoperative complication b | 9.7 (21) |
| Untreated posterior capsule opacification | 6.5 (14) |
| Cystoid macular edema (not associated with diabetic retinopathy) | 3.2 (7) |
| Other c | 1.4 (3) |
| No known nonrefractive cause d | 36.4 (79) |
a Cases with VA that was corrected with a pinhole aperture by ≥3 lines.
b Includes posterior capsular tear, anterior vitrectomy, or iris trauma.
c One case each of previous penetrating eye trauma, branch retinal vein occlusion, or amblyopia.
d Nonrefractive causes include AMD, glaucoma, diabetic retinopathy, epiretinal membranes, myopic retinopathy, optic disc atrophy, previous retinal tear, previous penetrating eye trauma, corneal lesion, branch retinal vein occlusion, amblyopia, intraoperative complication, posterior capsule opacification, or cystoid macular edema.
By October 2011, 232 of the 901 patients (26%) had died. The age-standardized mortality rates were marginally lower in patients with either no visual impairment (24.1%, 95% CI 19.9%-28.4%) or mild visual impairment (24.7%, 95% CI 16.5%-32.9%) at the 1-month-postoperative visits, compared to those with persistent moderate-severe visual impairment (30.6%, 95% CI 23.3%-37.9%) after surgery. However, these differences were not significant ( P = .12 and P = .056, respectively). Of the 232 patients who had died in the 5-year follow-up period, only 24% (n = 55) had data on cause of death. In this small number of cases, there was a higher incidence of cardiovascular death in the group with visual impairment remaining after surgery (69.6%, 16/23) compared to those with visual impairment corrected by cataract surgery (40.6%, 13/32; P = .034). Age, low BMI, and history of angina, myocardial infarction, stroke, and kidney disease were significantly associated with increased mortality ( P < .05) in Model 2, and the remaining co-variables were not significant in the model but were forced into the model. After adjusting for all the variables in Model 2 (age, sex, smoking, BMI, hypertension, diabetes, angina, myocardial infarction, transient ischemic attack, stroke, and kidney disease) and grouped indicators of general health and frailty (number of medications used and number of comorbid conditions), patients with no visual impairment postoperatively had a significantly lower mortality risk (HR 0.71, 95% CI 0.51-0.99, P = .044) compared to those with persistent moderate-severe visual impairment at 1-month-postoperative visits. Patients with mild visual impairment postoperatively had nonsignificantly lower mortality risks compared to those with persistent moderate-severe visual impairment postoperatively ( P > .06, Table 3 ).



