Purpose
To assess 3-year incidence and associated factors of posterior capsule opacification (PCO) after phacoemulsification surgery.
Design
Cohort study.
Methods
A total of 1934 consecutive patients aged ≥64 years undergoing phacoemulsification surgery at Westmead Hospital were recruited, of whom 1495 (77.3%) had retroillumination anterior segment images taken of the surgical eyes after 1 month and at a further postoperative visit within 3 years. Severe PCO was defined if the view of the optic disc was obscured, or neodymium–yttrium-aluminum-garnet capsulotomy was performed. Cumulative incidence of PCO was estimated using Kaplan-Meier methods. Associations of PCO with surgeon groups and different types of implanted intraocular lenses (IOLs) were assessed, adjusting for age, sex, diabetes, and ethnicity.
Results
Three-year cumulative incidence of PCO was 38.5% (95% confidence interval [CI] 36.1%-40.9%) among the 1495 patients, including 4.7% (95% CI 3.5%-5.8%) with severe PCO. PCO incidence was higher in: (1) eyes operated on by junior trainees (49%) compared to those by senior ophthalmologist surgeons (36%) (adjusted odds ratio [OR] 1.6, 95% CI 1.2-2.0); and (2) eyes with hydrophobic, 3-piece (either square-edged [51%, OR 2.3, 95% CI 1.4-3.6] or partial-round-edged [39%, OR 1.5, 95% CI 1.1-2.1]), or hydrophilic IOLs (64%, OR 2.9, 95% CI 1.9-4.4) compared to those with single-piece, square-edged, hydrophobic IOLs (34%), after additional adjustment for surgeon group.
Conclusions
Predominantly mild PCO occurred in one-third of eyes after phacoemulsification surgery. Possible predisposing factors associated with PCO development include surgery performed by ophthalmologic trainees and the use of hydrophilic or other hydrophobic IOLs apart from the single-piece, square-edged one.
Posterior capsule opacification (PCO) is a frequent complication after cataract surgery. PCO incidence has been reported in 36%-97% of patients 2-4 years after extracapsular cataract extraction. The corresponding incidence within 1 year after phacoemulsification surgery was 2%-15%, and within 3 years it was found to range between 2% and 63%. Factors associated with PCO remain uncertain.
Many previous cohort or randomized controlled studies that assessed PCO after cataract surgery had small sample sizes (<300 patients) or had assessed postoperative outcomes of a few, mainly senior, surgeons. These studies had not adjusted for different operating surgeons or different surgical experience. It remains unclear whether these previous study findings are applicable to other settings, such as that in a public tertiary hospital, where a large number of cataract procedures are performed by ophthalmologic trainees.
In this report we aimed to assess incidence of PCO within 3 years after phacoemulsification surgery in a large Australian cataract surgical cohort, and to explore preoperative and surgical factors associated with PCO in the study sample.
Methods
The Australian Prospective Cataract Surgery and Age-related Macular Degeneration study is a cohort study of patients recruited for cataract surgery at Westmead Hospital and some private ophthalmology clinics in Western Sydney, Australia. For the purpose of this report, only patients recruited at Westmead Hospital were included for consideration in analysis. The study conduct adhered to the tenets of the Declaration of Helsinki and was approved by the Human Research Ethics Committees of the University of Sydney and the Sydney West Area Health Service. All participants provided written informed consent. Details of the study and characteristics of the study sample are previously described.
Of 1934 patients aged ≥64 years who were recruited for phacoemulsification surgery, 96.5% (n = 1867) subsequently had cataract surgery in 1 eye per person (the surgical eye). All patients in the study attended 1-month and 1 or more annual follow-up visits at the Westmead Eye Clinic over 3 postoperative years. Of the 1867 patients, 2.9% (n = 55) died before their 1-year postoperative visit, and 17.0% (n = 317) did not attend follow-up visits either at 1 month or any time later during the 3-year period. After excluding the above groups of patients, 1495 (80.1% of 1867) patients remained for analysis ( Figure 1 ). Of these 1495 patients, 68.0% (n = 1016) had a single-piece, square-edged, hydrophobic acrylic intraocular lens (IOL) (AcrySof SA60AT; Alcon Laboratories, Fort Worth, Texas, USA); 4.5% (n = 67) had a 3-piece, square-edged, hydrophobic acrylic IOL (MA50BM; Alcon Laboratories); 10.4% (n = 156) had a 3-piece, hydrophobic acrylic IOL with partially round (ie, anterior round and posterior square) edges (Sensar AR40e; Advanced Medical Optics, Santa Ana, California, USA); 6.7% (n = 101) had a square-edged, hydrophilic acrylic IOL (Akreos Adapt; Bausch & Lomb, Rochester, New York, USA; Akreos Adapt AO; Bausch & Lomb; or Quatrix; Corneal/Croma, Leobendorf, Austria); and 10.4% (n = 155) had an unknown type of IOL.
All phacoemulsification surgery was performed by any of 20 ophthalmologist consultants, 6 fellows, or 45 ophthalmologic trainees. Consultants were specialists of the Royal Australian and New Zealand College of Ophthalmologists (RANZCO). Trainees were in their first to fourth year of a 5-year ophthalmologic training program. Fellows were either senior ophthalmologic trainees from overseas or Australian trainees in their final (fifth) year of training who had completed their fellowship examinations. A report by Prakash and associates showed that second-year ophthalmologic trainees had greater subjective difficulty with phacoemulsification surgery than their third-year counterparts. Hence, we have categorized junior surgeons as trainees in the first or second training year and senior surgeons as those who were third- or fourth-year trainees, fellows, or consultants.
Comprehensive eye examinations were conducted preoperatively and at 1, 12, 24, and 36 months postoperatively, and included measurement of presenting visual acuity (VA) (with habitual correction), using a back-illuminated logarithm of the minimal angle of resolution (logMAR) chart (Vectorvision CSV 1000; Vectorvision Inc, Dayton, Ohio, USA), in a room with standardized lighting. Patients found to have cystoid macular edema (CME) at the 1-month postoperative examination received a course of ketorolac trometamol 0.5% (Acular; Allergan Australia, New South Wales, Australia) 4 times a day and topical dexamethasone 0.1% (Maxidex; Alcon Australia, New South Wales, Australia) 4 times a day for 4 weeks. VA was recorded as the number of letters read correctly at 2.44 m (8 ft) and scored in logarithmic scale. The maximum number of letters read correctly was 70, while −15 letters was allocated to those who could not read any logMAR letter correctly at any distance and had vision recorded as counting fingers, hand movements, or perception or no perception of light. The surgical eye of each patient was included in the analysis.
At the preoperative visit, demographic data and questions on glaucoma and diabetes mellitus were collected and checked against patient medical records. Operation details were extracted from surgical audit forms completed by the surgeon immediately following the operation. The audit form asked questions about the main surgeon, capsulorrhexis diameter, type of IOL implanted, and whether any surgical complication occurred.
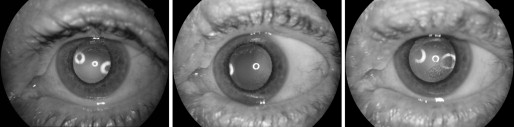
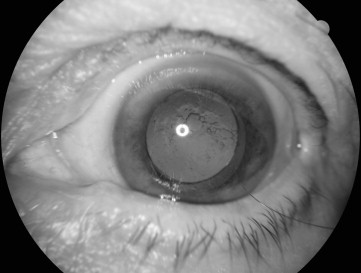
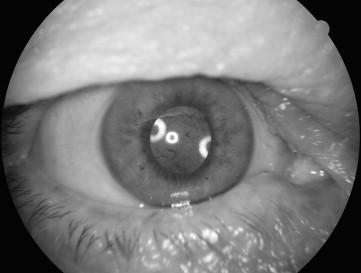
Mydriatic photographs of the anterior segment and retina were taken of both eyes at each visit using either a retinal camera (Topcon TRC 50 IA; Topcon Optical, Tokyo, Japan) with Kodachrome 64 35-mm slide film or a mydriatic digital retinal camera (CF-60DSi; Canon, Tokyo, Japan). The diagnosis of diabetic retinopathy was based on the modified Early Treatment Diabetic Retinopathy Study (ETDRS) classification system. PCO (opacity or wrinkling) was graded by the first author (C.S.F.) and adjudicated by a senior researcher (J.J.W.), according to Congdon and associates, as follows: no grade, PCO absent ( Figure 2 , Left) or only affecting the peripheral capsule; grade 1, any PCO affecting an area 4 mm in diameter around the visual axis, but not obscuring the retina ( Figure 2 , Center); grade 2, central PCO slightly obscuring the view of the macula, but permitting an assessment of the cup-to-disc ratio of the optic disc ( Figure 2 , Right); grade 3, central PCO making the assessment of the optic disc difficult ( Figure 3 ); grade 4, central PCO sufficiently obscuring the view of the entire fundus ( Figure 4 ). PCO was graded without reference to the patient’s VA in that eye. Severe PCO at any visit was defined as PCO sufficiently obscuring the view of either the optic disc or the entire fundus (grades 3-4), or in patients who had undergone neodymium: yttrium-aluminum-garnet (Nd:YAG) capsulotomy. This grading scheme had good internal validity in our study as postoperative PCO of grades 3-4 was associated with significantly worse presenting VA compared to none, mild PCO, or Nd:YAG capsulotomy ( Table 1 ).
| No PCO | PCO of Grade 1-2 | Untreated PCO of Grade 3-4 | Nd:YAG Capsulotomy | |
|---|---|---|---|---|
| 12 months, N = 1448 | ||||
| n | 1253 | 181 | 4 | 10 |
| Mean VA (95% CI) | 42.0 (41.2-42.7) | 38.5 (36.3-40.6) | 23.3 (-23.8-70.3) | 42.4 (33.7-51.1) |
| P a | .29 | .38 | – | .29 |
| 24 months, N = 1282 | ||||
| n | 925 | 321 | 16 | 20 |
| Mean VA (95% CI) | 42.2 (41.3-43.1) | 40.9 (39.2-42.5) | 17.0 (7.6-26.4) | 40.4 (32.4-48.3) |
| P a | <.0001 | <.0001 | – | .0003 |
| 36 months, N = 1132 | ||||
| n | 651 | 419 | 26 | 36 |
| Mean VA (95% CI) | 41.6 (40.5-42.7) | 41.5 (40.1-42.8) | 22.5 (13.2-31.8) | 41.5 (37.0-46.0) |
| P a | .0003 | .0003 | – | .0005 |
a P value for the comparison of mean presenting VA between grade 3-4 PCO and each of the other PCO groups.
PCO progression was defined as PCO grade 1 or 2 detected at the 1-, 12-, or 24-month examination that had progressed to grade 3 or 4, or the presence of Nd:YAG treatment in the following 12 months, assessed at the following annual postoperative visit. Stable PCO was defined as PCO that remained as grade 1 or 2 from the 1-, 12-, or 24-month visit to the following annual visit.
Statistical Analysis
SAS (Version 9.3; SAS Institute Inc, Cary, North Carolina, USA) was used for all analyses. Cumulative 3-year incidence of any PCO was calculated while considering the competing risk of death. This is an adaptation of the Kaplan-Meier method where time to the competing event, either death or the development of PCO, was modeled. Patients eligible to be included in analyses included those with information up to the time of any of the following events occurring: death, development of PCO, or the last visit during the 3-year postoperative period.
The χ 2 test was used to compare proportions between groups with and without PCO. Student t test was used to compare means of VA between different PCO groups. Multivariable-adjusted discrete logistic regression models were used to calculate adjusted odds ratios (ORs) and 95% confidence intervals (CIs) for factors associated with the incidence of PCO, including surgeon group (junior or senior surgeon) and type of IOL implanted (single-piece, square-edged, hydrophobic acrylic; 3-piece, square-edged, hydrophobic acrylic; 3-piece, hydrophobic acrylic with partial-round edges; or square-edged, hydrophilic acrylic). Comparisons were made between patients operated on by junior surgeons and those operated on by senior surgeons; and between the different types of IOLs implanted. Two models were used for the IOL analyses: Model 1 adjusted for baseline age and sex; Model 2 additionally adjusted for diabetes, ethnicity (white, East Asian, Middle Eastern, South Asian, or other ethnic group), and surgeon group. A multivariable model that included both the co-variables in Model 2 and the interaction term (a product term of IOLs and surgeon group) was used to test for interaction between IOL and surgeon groups; No interaction ( P = .44) was found. PCO progression rate was compared between patients implanted with different IOL types, adjusted for baseline age, sex, diabetes, ethnicity, and surgeon group.
Results
Patients included in this report were younger (mean age 74.8 years) and had a lower proportion of cardiovascular disease (339/1494; 22.7%) compared to those who were excluded (mean age 76.8 years, P < .0001; cardiovascular disease 27.8% [103/371], P = .040; Table 2 ). In addition, there was a lower proportion of white and a higher proportion of South Asian patients among the 1495 included patients (white 67.7% [n = 1006], South Asian 5.5% [n = 81]) than among the 372 excluded patients (white 72.9% [n = 266], South Asian 3.0% [n = 11]; P = .06), although the differences were marginal ( Table 2 ).
| Characteristic | N | Group | P a | |
|---|---|---|---|---|
| Included (N = 1495), n (%) | Excluded (N = 372) ∗ , n (%) | |||
| Age, mean ± SD, y | 1867 | 74.8 ± 5.8 | 76.8 ± 6.9 | <.0001 |
| Female, % | 1867 | 846 (56.6) | 226 (60.8) | .15 |
| Ethnicity, b % | 1851 c | |||
| White | 1272 | 1006 (67.7) | 266 (72.9) | .056 |
| East Asian | 211 | 175 (11.8) | 36 (9.9) | .30 |
| Middle Eastern | 210 | 170 (11.4) | 40 (11.0) | .80 |
| South Asian | 92 | 81 (5.5) | 11 (3.0) | .055 |
| Other | 66 | 54 (3.6) | 12 (3.3) | .75 |
| Glaucoma, % | 1861 c | 165 (11.1) | 40 (10.8) | .87 |
| Diabetes mellitus, % | 1867 | 440 (29.4) | 110 (29.6) | .96 |
| Cardiovascular disease, d % | 1865 c | 339 (22.7) | 103 (27.8) | .040 |
| Previous cataract surgery, % | 1861 c | 358 (24.0) | 91 (24.5) | .84 |
∗ Data are n (%) unless indicated .
a P value for the comparison of patients with and without follow-up visits.
b Comparisons of the proportion of each ethnic group between patients with and without PCO.
c Excludes cases with missing data.
d Cardiovascular disease includes either angina or myocardial infarction.
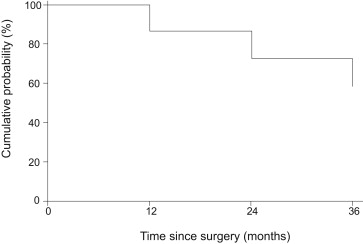
The 3-year cumulative incidence of any PCO and severe PCO was 38.5% (95% CI 36.1%-40.9%) (n = 544) and 4.7% (95% CI 3.5%-5.8%) (n = 62), respectively, among 1495 cataract surgery patients. Of the 62 severe cases, 45% (n = 28) had grade 3 or 4 but were untreated, and 55% (n = 34) had had Nd:YAG capsulotomy. Of 1125 patients with data on postoperative medications at the 1-month postoperative visit, CME was observed in 4.4% (n = 49). Three-year PCO incidence was similar in those who had (42.9%) and those who had not received anti-inflammatory medications for CME (41.1%, P = .80). Over the 3-year period, there was a progressive increase in the cumulative incidence of PCO ( Figure 5 ).
Of included patients, those with PCO were more likely to be female (60.3%; 328/544) and less likely to be South Asian (3.1%; 17/542) compared to those without PCO (female 54.5% [518/951], age-adjusted P = .025; South Asian 6.8% [64/944], age- and sex-adjusted P = .003; Table 3 ). In addition, there was a higher proportion of white patients (70.7%; 383/542) and a lower proportion of patients with diabetes mellitus (26.5%; 144/544) among those with PCO compared to those without PCO (white 66.0% [623/944], age- and sex-adjusted P = .064; diabetes 31.1% [296/951], age- and sex-adjusted P = .067) ( Table 3 ). Of the 544 patients who developed PCO within the 3-year postoperative period, 36.4% (n = 198) developed PCO within the first postoperative year.
| Characteristic | N | Group | P a | |
|---|---|---|---|---|
| With PCO (N = 544) ∗ , n (%) | No PCO (N = 951) ∗ , n (%) | |||
| Preoperative | ||||
| Age, mean ± SD, y | 1495 | 74.7 ± 5.8 | 74.9 ± 5.8 | .43 |
| Female, % | 1495 | 328 (60.3) | 518 (54.5) | .025 |
| Ethnicity, b % | 1486 c | |||
| White | 1006 | 383 (70.7) | 623 (66.0) | .064 |
| East Asian | 175 | 68 (12.6) | 107 (11.3) | .48 |
| Middle Eastern | 170 | 55 (10.2) | 115 (12.2) | .28 |
| South Asian | 81 | 17 (3.1) | 64 (6.8) | .0029 |
| Other | 54 | 19 (3.5) | 35 (3.7) | .81 |
| Glaucoma, % | 1490 c | 53 (9.8) | 106 (11.2) | .40 |
| Diabetes mellitus, % | 1495 | 144 (26.5) | 296 (31.1) | .067 |
| Moderate-severe DR, % | 1246 c | 29 (6.3) | 46 (5.8) | .73 |
| Intraoperative | ||||
| Capsulorhexis diameter, mm | 764 c | 5.2 | 5.2 | .69 |
| Anterior capsular tear | 980 c | 13 (3.7) | 31 (4.9) | .38 |
| Posterior capsular tear | 980 c | 14 (4.0) | 18 (2.9) | .33 |
| Iris tear | 980 c | 8 (2.3) | 11 (1.8) | .56 |
| Wound burn | 980 c | 5 (1.4) | 6 (1.0) | .50 |
| Dropped lens fragment | 980 c | 1 (0.3) | 6 (1.0) | .24 |
| Anterior vitrectomy | 980 c | 8 (2.3) | 14 (2.2) | .95 |
| Pupil manipulation | 980 c | 3 (0.9) | 8 (1.3) | .56 |
| Incomplete capsulorhexis | 980 c | 2 (0.6) | 3 (0.5) | .84 |
| Excess movement | 980 c | 8 (2.3) | 11 (1.8) | .56 |
| Zonular dialysis | 980 c | 5 (1.4) | 5 (0.8) | .34 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


