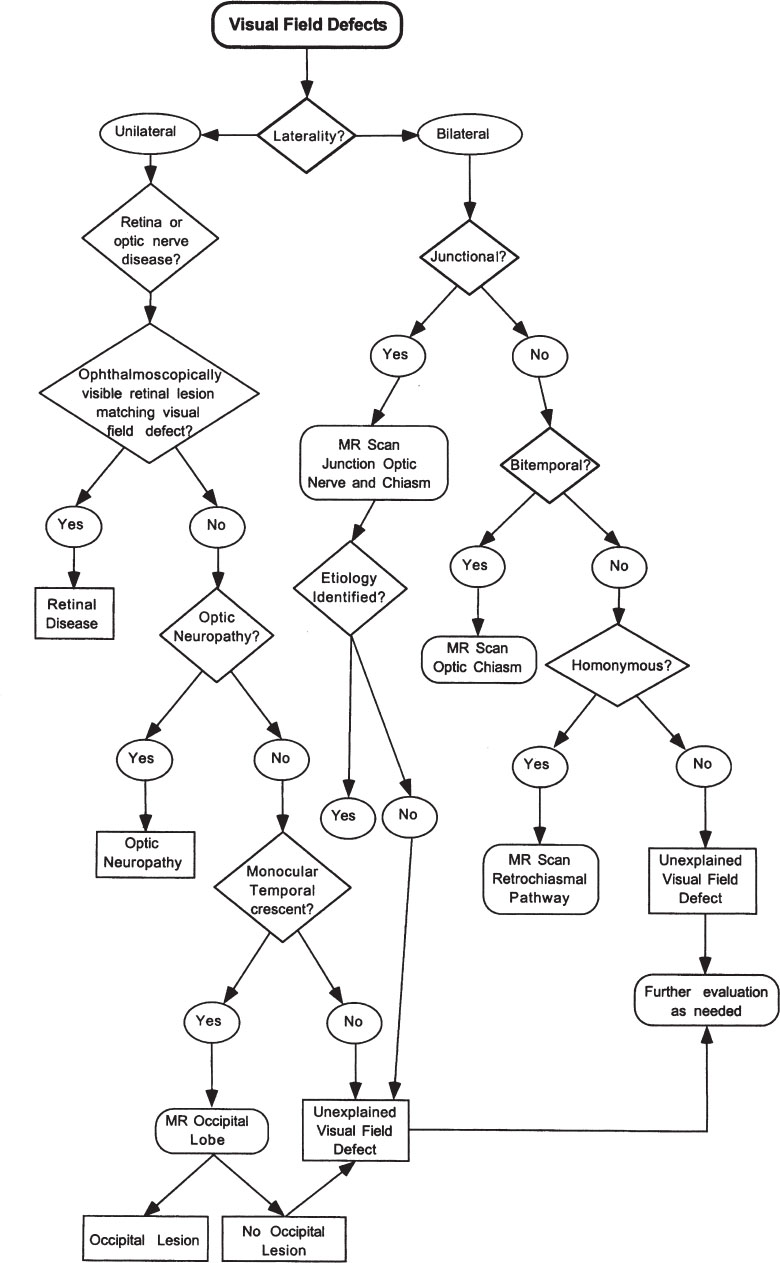
Visual Field Defects What Is the Topographical Diagnosis of Visual Field Defects? The localization of visual field defects is outlined in Figure 9–1. Is the Visual Field Defect Unilateral? Lesions affecting the retina, nerve fiber layer, or optic nerve produce visual field defects in the ipsilateral eye that correspond in position, shape, extent, and intensity to the lesion. The lesion may be inflammatory, ischemic, degenerative, or neoplastic. Because the nerve fiber layer arising from the peripheral retina arches over the fovea, superior or inferior nerve fiber layer damage results in arcuate-shaped visual field defects. Rarely, patients with a lesion of the anterior occipital lobe may have a unilateral, contralateral visual field defect (see monocular temporal crescent). Is a Retinal Lesion Responsible for the Visual Field Defect? Almost all retinal lesions resulting in visual field loss are visible ophthalmoscopically. Careful attention should be directed to the retina and retinal nerve fiber layer corresponding to the visual field defect. Patients with macular disease also may complain of metamorphopsia, micropsia, and positive photopsias (e.g., flashing lights) that are unusual in patients with optic neuropathies. Easily visible retinal lesions are not discussed in detail. Table 9–1 lists some retinal disorders that may be difficult to visualize without careful attention to the macula with high magnification and stereoscopic viewing. In some cases the retina may appear normal or near normal, and ancillary testing may be required to define the etiology as retinal (e.g., fluorescein angiography or focal, multi-focal, or full field electroretinography). Figure 9–1. Evaluation of visual field defects. Cystoid macular edema Epiretinal membrane Outer retinal inflammatory diseases Multiple evanescent white dot syndrome (MEWDS) Acute macular neuroretinitis (AMN) Acute retinal pigment epitheliitis Acute multifocal placoid pigment epitheliopathy (AMPPE) Acute zonal occult outer retinopathy (AZOOR) Serous detachment of the macula Cone-rod dystrophy Retinitis pigmentosa sine pigmento Cancer-associated retinopathy (CAR) Melanoma-associated retinopathy (MAR) Annular or ring scotomas may occur with retinopathies or optic neuropathies. Etiologies of annular or ring scotomas include pigmentary retinopathies, retinitis, choroiditis, blinding diffuse light, retinal migraine, myopia, cancer-associated retinopathy (CAR), open angle glaucoma (from coalescence of upper and lower arcuate scotomas), and optic neuropathies (e.g., anterior ischemic optic neuropathy, AION). Bilateral annular or ring scotomas may be due to bilateral retinal or optic nerve disease but may also occur with bilateral occipital pole damage or occur on a functional (nonorganic) basis. Is There Evidence for an Optic Neuropathy? Central visual field defects (unilateral or bilateral) are the result of damage to the papillomacular bundle or optic nerve. Any visual field defect produced by a retinal lesion may be produced by a lesion of the optic nerve (Miller, 1998) and virtually any etiology may be responsible (e.g., glaucomatous, degenerative, ischemic, traumatic, inflammatory, infiltrative, compressive, vascular optic neuropathy). Patients with a unilateral visual field defect and evidence for an optic neuropathy should undergo evaluation for an optic neuropathy (see Chapter 1). Patients with a unilateral hemianopic visual field defect (junctional scotoma of Traquair) may harbor a lesion of the optic nerve at the junction of the optic nerve and chiasm. In assessing optic nerve–related visual field defects, several anatomic points are worth remembering: 1. Fibers from peripheral ganglion cells occupy a more peripheral position of the optic disc, whereas fibers from ganglion cells located closer to the disc occupy a more central position. 2. Peripheral fibers course peripherally through the entire extent of the optic nerve. 3. The papillomacular bundle occupies a large sector-shaped region of the temporal disc. This bundle of fibers moves centrally in the more distal (posterior) portions of orbital optic nerve. 4. All retinal fibers retain their relative positions throughout the visual pathways except in the optic tract and at the lateral geniculate nucleus, where there is a rotation of 90 degrees that becomes “straightened out” in the optic radiations. Is the Monocular Temporal Crescent Affected in Isolation? Although monocular peripheral temporal visual field defects are most often the result of retinal or optic nerve disease, a lesion of the peripheral nasal fibers in the anterior occipital lobe may also produce a unilateral (monocular) temporal crescent-shaped visual field defect from 60 to 90 degrees (“half-moon syndrome”) (Chavis, 1997). Although retrochiasmal lesions in the visual pathway usually result in homonymous visual field loss, the monocular temporal crescent is the one exception. Homonymous visual field loss with sparing of this temporal crescent or selective involvement of this area localizes the lesion to the occipital lobe, and neuroimaging should be directed to the contralateral calcarine cortex (Landau, 1995; Lepore, 2001). Is the Visual Field Defect Bilateral? Bilateral lesions of the retina or optic nerve may result in bilateral visual field defects. Table 9–2 reviews the clinical features of bilateral superior or inferior altitudinal defects and bilateral central or cecocentral scotomas. In the absence of evidence for bilateral retinal disease or bilateral optic neuropathy, bilateral visual field loss results from disease affecting the optic chiasm or retrochiasmal pathways. Is the Visual Field Defect Junctional in Nature? Nerve fibers originating in the retina follow a specific topographic arrangement in the optic nerve and chiasm. Compressive lesions at the junction of the intracranial optic nerve and optic chiasm may produce characteristic visual field defects. Optic nerve involvement at the junction of the optic chiasm results in unilateral visual field loss (optic neuropathy). If fibers from the inferonasal retina of the contralateral eye (Wilbrand’s knee) are involved, there will also be a superotemporal visual field defect in the contralateral eye. The intracranial optic nerves extend posteriorly from the optic foramen and join at the optic chiasm. Within the chiasm, fibers from the nasal retina of each eye cross into the contralateral optic tract, and fibers from the temporal retina pass uncrossed into the ipsilateral optic tract. Within the intracranial optic nerve, the crossed (nasal retinal) and uncrossed (temporal retinal) fibers are anatomically separated at the junction of the optic nerve and chiasm. In addition, inferior nasal crossing fibers may loop anteriorly for a short distance into the contralateral optic nerve. These fibers are often referred to as the anterior knee or Wilbrand’s knee. Bilateral superior or inferior (altitudinal hemianopia) Most caused by bilateral optic nerve or retinal disease Rarely large prechiasmal lesion compresses both nerves inferiorly to cause bilateral superior hemianopia; compression of nerves from below may also elevate them against the dural shelves extending out from the intracranial end of the optic canals and cause bilateral inferior altitudinal defects Bilateral symmetric damage to postchiasmal pathways may cause bilateral altitudinal defects; bilateral lesions of medial aspect of lateral geniculate body may cause bilateral inferior hemianopias Bilateral occipital lesions may cause bilateral inferior, and less often, superior hemianopias Bilateral “checker board” altitudinal hemianopia—superior defect in one eye and inferior in the other eye Bilateral optic neuropathies May cause binocular diplopia because of vertical hemiheld slide—preexisting minor phoria becomes a tropia because of vertical or horizontal separation or overlap of two nonoverlapping hemifields; this phenomenon may also occur with broad arcuate defects Bilateral cecocentral or central scotomas May be bilateral optic neuropathy of any cause (e.g., compressive) but more common etiologies include: Toxic or nutritional amblyopia Bilateral demyelinating optic neuritis Syphilis Leber’s hereditary optic neuropathy Bilateral macular disease Bilateral occipital lesions involving macular projections Lesions at the junction of the optic nerve and chiasm may produce specific types of visual field defects that allow topographic localization. Selective compression of the crossed or uncrossed visual fibers at the junction may result in a unilateral temporal or nasal hemianopic field defect, respectively. In addition, involvement of the inferonasal fibers of the anterior knee (Wilbrand’s knee) results in a superotemporal visual field defect contralateral to the lesion. In 1927, H. M. Traquair used the term junctional scotoma to refer to a unilateral temporal hemicentral field defect due to compression of the nasal fibers crossing in the intracranial optic nerve at the junction of the optic nerve and chiasm. Miller emphasized that the junctional scotoma described by Traquair refers to a strictly unilateral temporal scotoma that is assumed to arise from a lesion at the junction of the optic nerve and chiasm (Miller, 1998). Unfortunately, some confusion has arisen regarding the use of the term junctional scotoma. As opposed to the defect described by Traquair, some authors have used the term to refer to an ipsilateral optic neuropathy with a contralateral superotemporal visual field defect. This superotemporal defect is caused by compression of the inferonasal fibers from the contralateral eye traveling in Wilbrand’s knee. To clarify this distinction, Miller, citing J. Lawton Smith, recommended that the unilateral temporal visual field defect described by Traquair should be referred to as the “junctional scotoma of Traquair” to differentiate it from the contralateral superotemporal defect more commonly referred to as the “junctional scotoma” (Miller, 1998). Recently, the existence of Wilbrand’s knee has come into question. Wilbrand was restricted to examining human subjects who had undergone enucleation. In the enucleated eye, the nerve fibers atrophied and became distinct from the nerve fibers of the normal eye as seen on myelin staining. Horton, utilizing axon labeling techniques in nonenucleated monkeys, was unable to demonstrate crossing fibers looping into the contralateral optic nerve (Wilbrand’s knee) (Horton, 1997). In one monkey that had undergone enucleation 4 years previously, however, nerve fiber topography similar to that described by Wilbrand was found. Horton hypothesized that Wilbrand’s knee may be an artifact of enucleation caused by atrophy of the optic nerve and not a normal anatomic finding. Nevertheless, whether Wilbrand’s knee exists anatomically, the localizing value of junctional visual field loss to the junction of the optic nerve and chiasm remains undiminished because chiasmal compression alone may result in the contralateral superotemporal visual field defect (junctional scotoma). Karanjia and Jacobson described a junctional scotoma due to a focal lesion (pituitary tumor) of the prechiasmatic segment of the distal optic nerve and stressed the “exquisite localizing value” of a junctional scotoma (Karanjia, 1999). Trobe and Glaser noted that junctional visual field loss was due to a mass lesion in 98 out of 100 cases (Trobe, 1983). The differential diagnosis of a junctional syndrome includes pituitary tumors, suprasellar meningiomas, supraclinoid aneurysms, craniopharyngiomas, and gliomas (Hershenfeld, 1993). Chiasmal neuritis, pachymeningitis, and trauma are rare etiologies of the junctional syndrome. Junctional visual field abnormalities may also occur on a functional (nonorganic) basis. Patients with the junctional scotoma of Traquair or the junctional scotoma should be considered to have a compressive lesion at the junction of the optic nerve and chiasm until proven otherwise. Neuroimaging studies, preferably magnetic resonance imaging (MRI), should be directed to this location. Patients with junctional scotoma may be unaware of a small superotemporal visual field defect, and patients presenting with strictly unilateral visual complaints may be misdiagnosed as having an optic neuritis or other unilateral optic neuropathy. Therefore, in any patient with presumed unilateral visual loss, careful visual field testing should be performed in the contralateral asymptomatic eye. Is a Bitemporal Hemianopsia Present? Bitemporal hemianopsia may be peripheral, paracentral, or central. The visual field defect may “split” or “spare” the macular central field. The bitemporal defect usually is the result of a compressive mass lesion at the level of the optic chiasm (Miller, 1998). Tables 9–3 and 9–4 list the possible etiologies of a chiasmal lesion. Pseudochiasmal visual field defects (i.e., bitemporal defects that do not respect the vertical midline) may be due to tilted discs, colobomas, bilateral nasal retinal disease (e.g., schisis), glaucoma, and bilateral optic neuropathies. Neuroimaging (preferably MRI) should be directed at the optic chiasm in all patients with bitemporal defects that respect the vertical midline. Certain anatomic relationships are important in evaluating chiasmal visual field defects: • The ratio of crossed to uncrossed fibers is 53:47. • Uncrossed fibers, both dorsal and ventral, maintain their relative position at the lateral aspects of the chiasm and pass directly into the ipsilateral optic tract. • Dorsal extramacular crossing fibers from each eye decussate posteriorly in the chiasm and then directly enter the dorsomedial aspect of contralateral optic tract. • Macular fibers that cross do so in the central and posterior portions of chiasm. • Some inferonasal retina fibers, primarily peripheral fibers, may loop in Wilbrand’s loop (although anatomic existence of this structure is questioned). Most common Pituitary apoplexy (Bills, 1993; Biousse, 2001; Embil, 1997) Pituitary tumor (especially pituitary adenoma) (Abe, 1998; Ikeda, 1995; Kerrison, 2000; Kupersmith, 1994; Lee, 1998; Peter, 1995; Petruson, 1995) Optic chiasm diastasis from pituitary tumor (Duru, 1999) Meningioma (Kinjo, 1995) Craniopharyngioma (Fahlbusch, 1999; Honegger, 1999; Mikelberg, 1993) Dysgerminoma Suprasellar aneurysm Chiasmal glioma (Cirak, 2000; Miyairi, 2000; Rossi, 1994) Less common Abscess Anaplastic astrocytoma (Miyairi, 2000) Arachnoid cyst Aspergillosis Cavernous hemangioma (Cobbs, 2001; Hwang, 1993) Chiasmal hematoma (chiasmal apoplexy) (Pakzaban, 2000) Chondroma Chordoma Choristomas Colloid cyst of the third ventricle Dermoid Dolichoectatic internal carotid arteries (Jacobson, 1999; Slavin, 1990b) Ependymoma Epidermoid Esthesioneuroblastoma Extramedullary hematopoiesis (Aarabi, 1998) Fibrous dysplasia Gangliocytoma (McCowen, 1999) Ganglioglioma (Liu, 1996) Giant cell tumor of bone Glioma Granular cell myoblastoma Hemangioblastoma (Sawin, 1996) Hemangioma (Bourekas, 2000) Hemangiopericytoma Histiocytosis X Hydrocephalus and distention of the third ventricle Intrasellar Langerhans’ cell histiocytoma (Job, 1999) Leukemia and lymphoma (Lee, 2001; McFadzean, 1990) Lipoma Lymphocytic hypophysitis (Abe, 1995; Beressi, 1994; Honegger, 1997; Jabre, 1997; Kerrison, 1997; Kristof, 1999; Lee, 1994; Naik, 1994; Nishioka, 1997; Stelmach, 1991; Thodou, 1995; Tubridy, 2001) Lymphohistiocytosis (Galetta, 1991) Melanoma (Aubin, 1997) Meningeal carcinomatosis Metastatic disease to brain or pituitary gland (Baeesa, 1999) Mucocele or mucopyocele Multiple myeloma Nasopharyngeal cancer Nonneoplastic pituitary gland enlargement Paraganglioma Plasmacytoma Rathke cleft cyst (El-Mahdy, 1998; Fischer, 1994; Rao, 1995; Voelker, 1991; Yamamoto, 1993) Rhabdomyosarcoma (Arita, 2001) Sarcoid granuloma Sarcoma Schwannoma Septum pellucidum cyst Sinus tumors Sphenoid sinus Syphilitic granuloma Teratoma Vascular malformation Venous aneurysm arising from carotid-cavernous sinus fistula (Wolansky, 1997) Venous angioma Is a Binasal Hemianopsia Present? Most organic nasal visual field defects are actually arcuate in nature. Bilateral irregular nasal defects may be associated with optic disc drusen, but such defects do not obey the vertical midline and in fact are generally arcuate in nature. Binasal defects are usually due to bilateral intraocular disease of the retina or optic nerve (e.g., chronic papilledema, ischemic optic neuropathy, glaucoma, optic nerve drusen, or retinal disease such as sector retinitis pigmentosa or retinoschisis). Rarely compression of the lateral chiasm may result in a binasal defect. Bilateral nasal defect may occur with hydrocephalus with third ventricle enlargement causing lateral displacement of optic nerves against the supraclinoid portion of the internal carotid arteries. Binasal defects have also been described in patients with primary empty sella syndrome and with other suprasellar lesions (Charteris, 1996). Hydrocephalus (Bogdanovic, 2000) Cobalamin deficiency (Wilhelm, 1993) Demyelinating disease (Newman, 1991; Wilhelm, 1993) Empty sella syndrome (primary or secondary) (Kosmorsky, 1997) Chiasmal ischemia Optochiasmatic arachnoiditis Foreign body–induced granuloma (e.g., muslin) Idiopathic Infection Chronic fungal infection Cryptococcus Cysticercosis (Chang, 2001) Encephalitis Epstein-Barr virus (Beiran, 2000) Meningitis Mucormycosis (Lee, 1996) Nasopharyngeal and sinus infections Syphilis Tuberculosis Inflammatory Collagen vascular disease (e.g., systemic lupus erythematosus) (Frohman, 2001; Siatkowski, 2001) Rheumatoid pachymeningitis Sarcoid Multiple sclerosis Posthemorrhagic Posttraumatic Radiation necrosis Shunt catheter Toxic (see toxic optic neuropathies) Tobacco-alcohol toxicity (Danesh-Meyer, 2000) Ethchlorvynol (Placidyl) Pheniprazine (Catron) Trauma, including postsurgical (Carter, 1998; Domingo, 1993; Heinz, 1994) Fat packing after transsphenoidal hypophysectomy (McHenry, 1993) Tethering scar tissue causing delayed visual deterioration after pituitary surgery (Czech, 1999) Vascular occlusion Vasculitis (Wilhelm, 1993) Hereditary (probably autosomal recessive) chiasmal optic neuropathy (Pomeranz, 1999) Nonorganic (functional) (Miele, 2000) An unusual binasal visual field impairment has been noted with spontaneous intracranial hypotension from a dural cerebrospinal fluid leak (Horton, 1994). Some of these patients have a binasal defect with peripheral depressions most severe in the upper nasal quadrants but also involving the lower nasal and upper temporal quadrants. Is a Homonymous Hemianopsia Present? Homonymous visual field impairments appear with lesions of the retrochiasmal pathways. Those affecting the optic tract and lateral geniculate body tend to be incongruous, but the more posteriorly the lesion is located in the optic radiation, the greater the congruity of the defects. In general, tumors produce sloping field defects, whereas vascular lesions produce sharp field defects. The localization of homonymous field defects depends on the nature of the field defect and associated neuro-ophthalmologic and neurologic findings. Homonymous field defects may be caused by lesions affecting the optic tract, lateral geniculate body, optic radiations, or occipital lobe. Rarely, an occipital lesion may cause a monocular field defect (see monocular crescent, above). In general, complete homonymous hemianopias are nonlocalizing and may be seen with any lesions of the retrochiasmal pathway, including lesions of the lateral geniculate body, optic radiations, and striate cortex. Is the Homonymous Hemianopia Caused by an Optic Tract Lesion? In the optic tract, macular fibers lie dorsolaterally, peripheral fibers from the upper retina are situated dorsomedially, and peripheral fibers from the lower retina run ventrolaterally. Complete unilateral optic tract lesions cause a complete macular splitting homonymous hemianopia, usually without impaired visual acuity, unless the lesion extends to involve the optic chiasm or nerve. Partial optic tract lesions are more common than complete lesions and result in an incongruous field defect that may be scotomatous. (The only other postchiasmatic location for a lesion causing a scotomatous hemianopic visual field defect is the occipital lobe.) Optic tract lesions are often associated with a relative afferent pupillary defect (RAPD) in the eye with the temporal field loss (contralateral to the side of the lesion). An afferent pupillary defect in the contralateral eye in a patient with normal visual acuity bilaterally and a complete homonymous hemianopia is usually indicative of optic tract involvement (Miller, 1998). Wilhelm et al described a possible exception to this clinical rule. These authors described a RAPD contralateral to the lesions in 16 of 43 patients with congruous homonymous hemianopias (optic tract lesions excluded) (Wilhelm, 1996). Responsible lesions were postgeniculate and closer than 10 mm to the lateral geniculate nucleus. A RAPD did not occur in lesions farther than 18 mm from the lateral geniculate nucleus. The authors postulated that the RAPD was probably not caused by a lesion of the visual pathway per se, but by a lesion of intercalated neurons between the visual pathways and the pupillomotor centers in the pretectal area of the midbrain. Another abnormality of the pupil that may occur with optic tract lesions is due to concurrent third nerve involvement by the pathologic process causing the tract damage. In these cases, the pupil ipsilateral to the lesion may be large and poorly reactive. Finally, many patients with chronic optic tract lesions develop bilateral optic atrophy with a characteristic “wedge,” “band,” or “bow-tie” pallor in the contralateral eye (identical to that seen in some patients with bitemporal visual field loss from chiasmal lesions), and a more generalized pallor in the ipsilateral optic nerve associated with loss of nerve fiber layer in the superior and inferior arcuate regions corresponding to the bulk of temporal fibers subserving the nasal visual fields (hemianopic optic atrophy) (Miller, 1998). Hemianopic optic atrophy indicates postchiasmal, preoptic radiation involvement (i.e., optic tract or lateral geniculate body damage), but has also been rarely described in congenital retrogeniculate lesions (Miller, 1998). Etiologies of optic tract lesions include space-occupying lesions (e.g., glioma, meningioma, craniopharyngioma, metastasis, pituitary adenoma, ectopic pinealoma, abscess, sella arachnoidal cyst), aneurysms, arteriovenous malformations, dolichoectatic basilar arteries, demyelinating disease, and trauma, including neurosurgical procedures (e.g., temporal lobectomy, insertion of intraventricular shunt) (Chun, 1998; Freitag, 2000; Groomm, 1997; Guirgis, 2001; Liu, 1997; Miller, 1998; Molia, 1996; Shults, 1993; Slavin, 1990a; Vargas, 1994). Patients undergoing posterior pallidotomy for parkinsonism may develop mild to moderate contralateral homonymous superior quadrantanopias associated with small paracentral scotomas likely due to optic tract damage (Biousse, 1998). A congenital optic tract syndrome has also been described (Murphy, 1997). A complete neurologic examination and MRI, with specific attention to the optic tract region, are warranted in all patients suspected of having an optic tract lesion. If MRI fails to reveal the responsible lesion, then MR angiography or cerebral angiography may be warranted in nontraumatic cases to investigate the presence of vascular lesions (e.g., aneurysm). Is the Homonymous Hemianopia Caused by a Lesion of the Lateral Geniculate Body? In the lateral geniculate body, axons from ganglion cells superior to fovea are located medially, axons originating from ganglion cells inferior to fovea are located laterally, and macular fibers terminate in a large central area. As axons leave the lateral geniculate body they re-rotate back to their original positions so that within the optic radiations and the striate cortex, fibers that have synapsed with axons from superior retinas are located in superior radiations and above the calcarine fissure in the striate cortex, whereas fibers that have synapsed with axons from the inferior retinas are located in the inferior optic radiations and below the calcarine fissure. Upper field fibers originate in the medial aspect of lateral geniculate nucleus and travel through the parietal lobes, whereas lower fields originate from the lateral aspect of the lateral geniculate body and make a loop in the temporal lobe (Meyer’s loop or the Meyer-Archambault loop). Lateral geniculate body lesions may also cause a complete macular splitting homonymous hemianopia (Miller, 1998). Partial lesions result in an incongruous homonymous field defect. Hemianopic optic atrophy may develop and no RAPD is usually evident. Although the study of Wilhelm et al suggests that a RAPD may occasionally be present with lateral geniculate body or parageniculate optic radiation lesions (Wilhelm, 1996), this observation has not been confirmed by other investigators.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


