Upper Airway Stenosis: Evaluation and Management
Philip A. Weissbrod
Albert L. Merati
Upper airway stenosis is an umbrella term for scarring or cicatrix of the trachea, subglottis, glottis, or supraglottis that can lead to airway obstruction. Stenosis can be limited to soft tissue scarring and fibrosis, involve cartilaginous remodeling from chondritis, or be a combination of the two. On a patient level, symptomatic stenosis is debilitating leading to significant reduction of quality of life due to obstructed breathing patterns and voice alterations (1). For many patients, especially those with postintubation-related stenosis, the difficulty breathing that comes with an airway lesion can potentiate their medical condition or significantly hinder their recovery. For all patients with airway stenosis at any level, their road to a diagnosis can often be laborious, time-consuming, and at the expense of considerable personal and societal resources. Many of them have seen numerous physicians and traveled a road fraught with misdiagnosis and unclear answers. Often patients present having been misdiagnosed with asthma, vocal fold dysfunction, or other respiratory conditions (2,3). During this period of delay between onset of symptoms and diagnosis, especially if there is significant symptomatology, their function can be limited and both their physical and mental well-being impaired limiting capacity for work and normal daily living.
In this chapter, subsites of airway stenosis including supraglottic, glottic, subglottic, and tracheal are independently addressed. Common causes, staging systems, treatment methods, and outcomes are discussed in an effort to provide overview of the current state of medical and surgical management. Etiology and evaluation of airway stenosis are discussed in general terms for all subsites, as there is significant overlap between subsites. When appropriate, specifics for each subsite are also discussed. Discussion of pediatric and congenital stenosis is not included as it is covered elsewhere in the text.
ETIOLOGY
The etiology of airway stenosis is varied (Table 62.1). While certain sites are more prone to specific disease processes, many can cause scarring anywhere between the supraglottis and trachea.
The origin of airway stenosis encompasses a broad spectrum of disease processes. Inflammation is the common link between all the disease processes. Injury to the airway can occur acutely from trauma, intubation, infection, endoscopy, or any form of surgery to the intrinsic airway that can cause mucosal or cartilaginous injury. Systemic processes such as laryngopharyngeal reflux, autoimmune disease, and neoplasm can create an environment of acute and/or chronic insult, which ultimately leads to scar and stenosis. Injury that is circumferential, in particular, can lead to greater likelihood of airway stenosis as noted by Andrews and Pearson (4) in their 2-year prospective study of intubated patients.
A number of canine models of airway stenosis have been created using drills, surgical dissection, or endotracheal tubes as the mechanism of injury. Regardless of the origin of trauma, any mucosal disruption, ulceration, or chondritis can lead to the formation of intraluminal granulation tissue and subsequently damage to the underlying cartilaginous framework, mucosal thickening with submucosal fibrosis, and ultimately, scar formation (5,6,7,8).
Trauma
The most common inciting factor for airway stenosis is trauma, most frequently in the form of postintubation injury. An endotracheal tube can create injury at two levels: the glottis due to the rigid tube’s wall and the trachea due to cuff pressure (9). An endotracheal or tracheotomy tube has the potential to create a circumferential injury within the
trachea (7). Injury can come from direct mucosal abrasion or from vascular compromise as a result of sustained cuff pressure (10). The vascular supply to the tracheal cartilage is derived from small intercartilaginous vessels that create a submucosal plexus (11). A number of these vessels originate from the inferior thyroid artery. Other vascular contributions to the cervical and thoracic trachea include the supreme intercostal, subclavian, right internal thoracic, and innominate artery (12,13). Increased cuff pressures, drops in systemic perfusion, cardiac bypass, and systemic vasculopathy, such as diabetes, can all contribute to local deprivation of blood supply. With ischemia come congestion, edema, and eventually ulceration (14,15). Ulceration can lead to chondritis and necrosis. Healing of necrotic compromised regions occurs by secondary intention leading to scar formation (16).
trachea (7). Injury can come from direct mucosal abrasion or from vascular compromise as a result of sustained cuff pressure (10). The vascular supply to the tracheal cartilage is derived from small intercartilaginous vessels that create a submucosal plexus (11). A number of these vessels originate from the inferior thyroid artery. Other vascular contributions to the cervical and thoracic trachea include the supreme intercostal, subclavian, right internal thoracic, and innominate artery (12,13). Increased cuff pressures, drops in systemic perfusion, cardiac bypass, and systemic vasculopathy, such as diabetes, can all contribute to local deprivation of blood supply. With ischemia come congestion, edema, and eventually ulceration (14,15). Ulceration can lead to chondritis and necrosis. Healing of necrotic compromised regions occurs by secondary intention leading to scar formation (16).
TABLE 62.1 ETIOLOGY OF UPPER AIRWAY STENOSIS | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Size of endotracheal tube, length of intubation, cuff pressure, and traumatic intubation all potentially can increase the likelihood of airway stenosis (17). Postintubation stenosis arose as a frequent disease process in the mid-20th century after an increase in long-term intubation injury as related to poliomyelitis (18). It became clear that the increases in incidence of stenosis was due to injury at the tracheostoma site from surgical manipulation, the presence of a pressurized balloon, and irritation of the tip of the respiration device (19). Localized infection of the stoma and trachea also was found to be a significant risk factor in early animal studies (20).
Over time, a number of mechanisms to reduce injury of the tracheal wall were identified. Early cuff pressures were high: the balloons were initially small in size and required intraluminal cuff pressures of greater than 180 mm Hg to allow for adequate positive pressure ventilation at 20 to 25 mm Hg (21,22). By increasing the size and compliance of the endotracheal and tracheotomy tube balloon subsequently lowering the intracuff pressures, there are fewer tracheal lesions from postintubation trauma seen in the later half of the last century (23,24).
Stenosis, however, can occur with a brief intubation and small endotracheal tube. In addition to tracheal and subglottic stenosis, intubation injury can lead to trauma and scarring of the posterior glottis in the intra-arytenoid region and cricoarytenoid joints, the location within the glottis which receives the most pressure from the presence of an endotracheal tube (25).
Tracheotomy or tracheostomy is the most common form of surgical trauma causing airway stenosis. Stenosis can be at any level along the tracheotomy tube or above the stoma. Postintubation or posttracheotomy stenosis is not uncommon, ranging from 10% to 19%, but typically only is symptomatic in less than 1% of cases (26,27,28). It is unclear in the current literature if there are any differences in rates of stenosis between open and percutaneous dilational tracheotomy (29,30), however, conceptually, percutaneous tracheotomy may cause greater cartilaginous injury from less controlled entry into the airway leading to destabilization of the laryngotracheal framework (31,32).
Other forms of injury can include laryngotracheal trauma secondary to blunt or penetrating neck trauma, which can lead to cartilaginous framework disruption, mucosal lacerations, and tears, both of which can potentially lead to scarring at any level. Other forms of surgically induced injury along the course of the upper airway can come in the form of thyroidectomy, partial pharyngectomy, partial laryngectomy, tracheal and bronchial surgery, and any endoluminal airway procedure that can lead to inflammation and scarring.
Iatrogenic glottic stenosis can occur with bilateral deepithelialization of the anterior commissure and anterior true vocal folds leading to anterior glottic web. Once formed, this can be difficult to treat, often requiring multiple procedures and stent or keel placement. Similarly, posterior glottic stenosis (PGS) can occur with excess manipulation of the posterior vocal folds and posterior glottis leading to scar formation and cricoarytenoid joint fixation; however, it should be restated that the most common cause is from endotracheal tubes. It has also been hypothesized that severe coughing can lead to telescoping of the cricoid cartilage over the first tracheal ring leading to vascular insult, which in the setting of poor wound healing can lead to airway stenosis (33).
Systemic Disease
Wegener’s granulomatosis is a disease of unknown etiology that can involve the kidney, lungs, and upper respiratory tract. The frequency of airway stenosis in Wegener’s, in particular subglottic stenosis varies depending on the series, but can
range from 8.5% to 50% (34,35,36). While the subglottis is the most commonly involved portion of the upper airway, glottic and supraglottic involvement has also been noted. Biopsy of the region can show noncaseating granulomas; however, biopsy is not always diagnostic. Antineutrophil cytoplasmic antibodies (c-ANCA) are greater than 90% positive during the active vasculitic phase of the disease and can be a useful adjunct in diagnosis (37). Adequate systemic control of disease is recommended prior to entertaining definitive treatment for stenosis. Systemic control can include medications such as methotrexate, cyclophosphamide, and in refractory cases rituximab (38).
range from 8.5% to 50% (34,35,36). While the subglottis is the most commonly involved portion of the upper airway, glottic and supraglottic involvement has also been noted. Biopsy of the region can show noncaseating granulomas; however, biopsy is not always diagnostic. Antineutrophil cytoplasmic antibodies (c-ANCA) are greater than 90% positive during the active vasculitic phase of the disease and can be a useful adjunct in diagnosis (37). Adequate systemic control of disease is recommended prior to entertaining definitive treatment for stenosis. Systemic control can include medications such as methotrexate, cyclophosphamide, and in refractory cases rituximab (38).
Relapsing polychondritis is another systemic disease process that has consequences in the airway. Most commonly associated with inflammation of the cartilaginous structures of the ear, it can also involve the nose, joints, larynx, and tracheobronchial tree. In a large cohort series of patients, 31/145 patients (21%) with polychondritis had airway manifestations. Of the 31 patients with airway symptoms, 8 (26%) had subglottic stenosis, 8 (26%) had other foci of stenosis within the tracheobronchial tree, and the remainder had diffuse malacia. Forty percent required surgical intervention including tracheotomy and dilation (39).
Laryngopharyngeal Reflux
Laryngopharyngeal reflux was first suggested as a culprit of airway stenosis in the early 1980s (40,41). In recent years, there has been increasing evidence that chronic acid injury to the lining of the airway can be caustic and lead to, among other things, stenosis. There have been numerous studies aimed at teasing apart the causative role of reflux in subglottic and tracheal stenosis. Koufman (42), in 1991, studied 32 patients with laryngotracheal stenosis and noted laryngopharyngeal reflux in 78%. Others have identified similar trends in 24-hour pH probe results establishing laryngopharyngeal reflux as one of the many factors that play a role in the development of airway stenosis (43,44,45).
The intubated patient is at particular risk of acid-related injury given the presence of an endotracheal tube and its associated trauma. Supine position and the use of a nasogastric or orogastric feeding tube has been suggested to increase rates of reflux events, aspiration, and ventilatorassociated pneumonia in the intubated patient (46,47). Currently, most ICUs place patients in a semirecumbent position (45 degrees), which has been shown to reduce ventilator-associated pneumonia and presumably gastric reflux events (48). Additionally, the widespread use of acid suppression medication in ICUs has likely further reduced acid-related injury in this patient population.
Idiopathic Stenosis
Idiopathic laryngotracheal stenosis occurs predominantly in childbearing aged women, ages 30 to 50. It is often a diagnosis of exclusion; patients usually have no history of airway trauma, systemic disease, or other identifiable etiology of stenosis. Typically, idiopathic stenosis involves a short segment of the subglottis and/or upper trachea; it manifests as a circumferential fibrous ring. These are usually isolated single level lesions as apposed to intubation injury-related stenosis, which often can have multilevel involvement (49). Interestingly, symptomatically, idiopathic stenosis patients tend to have less symptoms and individuals maintain normal respiratory effort until a critical stenosis has formed leading to relatively sudden onset of symptoms. Pathologically, idiopathic stenosis also behaves differently than stenosis from intubation trauma. Lacking is chondromalacia, a common factor seen in other forms of stenosis, and instead, there is a dense fibrotic process of the lamina propria (50).
Given the age and sex predilection, there is question as to the role of hormones in the development of idiopathic stenosis. Subglottic tissue has been evaluated for the presence of estrogen receptors; however, initial studies have failed to identify a link leaving the association still unfounded (51,52).
There is considerable debate of the role of laryngopharyngeal reflux as the underlying cause of idiopathic stenosis; however, to date there has been no definitive evidence to support this. Blumin and Johnston recently evaluated for the presence of pepsin in patients with idiopathic glottic stenosis. Pepsin serves as a highly sensitive and specific marker for the exposure of mucosal tissue to gastric contents (53). They noted 13/22 patients with idiopathic stenosis (59%) had evidence of pepsin in the stenotic tissue as apposed to controls that had no pepsin noted in biopsy specimens (P = 0.041) (54). Despite the relatively high rate of pepsin identified, there was not a statistically significant correlation between 24-hour pH probe and the presence of pepsin, which could relate to the deficiencies in acid testing alone or temporal limitations of testing methods.
It seems plausible that a certain percentage of patients with idiopathic stenosis may have stenosis from laryngopharyngeal reflux, but not all of this population tests positive leaving the underlying process, in many patients, still unknown. Grillo et al. (55) commented in their series of 72 patients with idiopathic stenosis, that only 5 (7%) required additional dilations despite cessation of antireflux medication further strengthening the argument that idiopathic stenosis and reflux-related stenosis are distinct entities.
Laryngitis and Tracheitis
Infection of the upper airway is a consideration in the development of tracheal stenosis. As mentioned previously, inflammatory lesions and ulcerations can lead to chondritis, granulation tissue, and scar formation. Theoretically, any intraluminal airway infection, whether viral or bacterial,
can incite an inflammatory reaction that can ultimately contribute to the development of airway stenosis, especially in cases where endotracheal intubation is necessary, as the results of multiple insults to the area could be additive. In a small but telling animal study, Squire et al. traumatized rabbit tracheas and inoculated them with Staphylococcus aureus. Animals were sacrificed at postoperative day 10. Those treated with antibiotics had significantly lower mean stenosis percentages as compared to those infected tracheas (50.8% vs. 85%) suggesting that infection, in the setting of airway trauma, can significantly worsen the rate and severity of stenosis (8).
can incite an inflammatory reaction that can ultimately contribute to the development of airway stenosis, especially in cases where endotracheal intubation is necessary, as the results of multiple insults to the area could be additive. In a small but telling animal study, Squire et al. traumatized rabbit tracheas and inoculated them with Staphylococcus aureus. Animals were sacrificed at postoperative day 10. Those treated with antibiotics had significantly lower mean stenosis percentages as compared to those infected tracheas (50.8% vs. 85%) suggesting that infection, in the setting of airway trauma, can significantly worsen the rate and severity of stenosis (8).
Other specific infective processes have no associations with stenosis. In some tuberculosis (TB) endemic regions, TB-related airway stenosis is a common, if not the most common etiology of tracheobronchial stenosis (56). Tracheal lesions in TB can develop from lymphatic spread, direct infection of local tissue via infected sputum, or local extension from paratracheal or mediastinal lymph nodes (57).
EVALUATION
The most important component in understanding stenosis is a history that accounts for all associated risk factors and conditions. An accurate understanding of intubation history and sources of trauma can point toward a culprit etiology. In cases where there is no prior history of trauma, consideration of appropriate infectious, laryngopharyngeal reflux, autoimmune, and systemic diseases should be entertained. Other considerations should include details about the rapidity of symptom development, exercise intolerance, and the effects of airway inflammation such as upper respiratory infections. In patients who are operative candidates, special consideration should be paid to processes that could effect wound healing such as diabetes mellitus, collagen disorders, vascular disorders, history of smoking, and alcohol consumption. Patients can be followed over the time by their subjective respiratory symptoms. Self-assessment of dyspnea at rest or on exertion can be valuable clinical indicators of the need for intervention. The rapidity of symptom return can also help determine the urgency of intervention.
Physical Examination and Office Endoscopy
Physical examination should start with careful observation of both shallow and deep respiration. The presence of stridor indicates obstruction and can point toward the level of obstruction. Low pitch inspiratory stridor indicates supraglottic obstruction. High pitched inspiratory or biphasic stridor suggests laryngeal, subglottic, or tracheal obstruction. The utilization of accessory muscles, suprasternal retractions, or rapid breathing can suggest impending airway compromise and the necessity for intervention either with intubation or tracheotomy.
Examination of the airway is important for characterization as it can dictate treatment options. Flexible or rigid laryngoscopy should be an integral part of airway evaluation. For evaluation of the supraglottis, glottis, and immediate subglottis, transnasal flexible endoscopy with topical nasal anesthetic with 4% lidocaine should be sufficient. In most cases, with proper patient positioning, detailed evaluation as far distal as the subglottis is possible. For patients with heightened laryngeal sensitivity or suspected mid or distal tracheal stenosis, direct application of lidocaine to the glottis allows for transglottic views of the trachea with flexible transnasal laryngotracheobronchoscopy with relatively low risk (58). When assessing stenotic patients, all areas of stenosis and scar should be noted. Dynamic examination of the larynx is important to ensure there is no paralysis or paresis of the vocal folds, a potential source of additional obstruction. Evaluation for mucosal inflammation, edema, erythema, and other signs of laryngopharyngeal reflux should also be noted (59).
If a tracheotomy is present, careful study of the stoma in relation to the cricoid should be performed as this can have bearing on the available surgical options. Examination via trans-stomal flexible tracheoscopy is easily accomplished. If the patient is stable and has a well-established tracheotomy track, the tracheotomy should be removed and the scope retroflexed, to view the subglottis from below.
Imaging
If access to an otolaryngologist or flexible laryngoscope may be limited, plain radiograph can be helpful in outlining the silhouette of the airway. The level and length of the stenosis usually can be visualized with plain radiography, but more detailed information such as thickness of the tracheal wall, further characterization of the lumen, and presence of wall calcifications can only be identified with computer tomography (CT) (60). The presence of calcifications is often seen in cases of postintubation-related trauma; in idiopathic stenosis often there is no calcification evident (61). Noncontrast thin cut CT is a more revealing study and can be helpful in visualizing stenotic airway segments or extraluminal compression from tumors or aberrant blood vessels (62,63). In addition, in situations where there is glottic compromise, CT scan may represent a means of airway evaluation while avoiding the risk associated with transglottic endoscopy.
Despite its many benefits, CT does have limitations in airway evaluation. If the stenotic segment is thin and CT scan slice is thick, a thin stenotic segment can be missed on imaging. In addition, when compared to endoscopy, CT if significantly less accurate in predicting stenotic segment length, an important negative predictive factor in airway surgery (64).
A relatively new imaging technology, virtual tracheobron-choscopy, is gaining popularity. This technology takes advantage of CT spiral data acquisition and utilizes the contrast between airway and surrounding tissue to allow for virtual endoluminal navigation through the airway (65,66). In a review of 37 patients, flexible tracheobronchoscopy,
intraoperative endoscopy, CT, and virtual endoscopy all correctly estimated the Cotton-Meyer stenosis grade of stenosis in greater than 94% of cases (67). Considering this, virtual endoscopy may be of particular benefit in patients who have tenuous airways where endoscopic evaluation may be risky or in patients with severe stenosis where the airway distal to the obstruction may not be easily visualized.
intraoperative endoscopy, CT, and virtual endoscopy all correctly estimated the Cotton-Meyer stenosis grade of stenosis in greater than 94% of cases (67). Considering this, virtual endoscopy may be of particular benefit in patients who have tenuous airways where endoscopic evaluation may be risky or in patients with severe stenosis where the airway distal to the obstruction may not be easily visualized.
Pulmonary Physiologic Testing
The use of physiologic objective testing of the airway can also be helpful. Flow volume loops (FVLs) are the standard method for assessing flow and volume characteristics of the upper airway by plotting the forced vital capacity against the rate of flow. Based on FVLs, airway obstruction can be differentiated into intrathoracic versus extrathoracic and fixed versus mobile obstruction. With a fixed obstruction, there is plateau of flow rates on both inhalation and exhalation as apposed to mobile extrathoracic obstruction which has a plateau on inspiration, and mobile intrathoracic obstruction with a plateau of flow on exhalation (68).
Despite the utility of FVLs, there are some limitations. It appears that they are a good qualitative instrument in determining obstruction but do not necessarily show the correlation between the degree of airway obstruction and severity of breathing symptom (69).
Reflux Testing
Objective reflux testing in patients with symptomatic reflux or idiopathic stenosis is also used by many practitioners (70). The gold standard for testing is dual probe 24-hour pH monitoring (71). More recently, esophageal manometry and impedance testing have been included in the diagnostic armamentarium to provide additional detail about the origin of reflux and the presence of both acidic and nonacidic reflux events. There is considerable debate regarding what represents positive testing. This discussion is outside of the scope of this chapter; however, many believe that any acidic (and even nonacidic) reflux event into the pharynx to be abnormal, especially when considering a patient with airway stenosis.
Direct Microlaryngoscopy and Operative Endoscopy
Operating room-based direct laryngoscopy is an integral part in evaluating all levels of airway stenosis. Careful preoperative planning with the anesthesia team is essential for safe administration of anesthesia and airway control. Aside from standard direct laryngoscopy techniques, advanced intubation techniques such as video-assisted endotracheal intubation and awake fiberoptic nasal intubation can be helpful in the appropriate patient. In cases of advanced glottic and supraglottic stenosis, intubation may be difficult. Rigid ventilating bronchoscopes for emergent intubation are a valuable tool in many cases. The surgical team should also be prepared for the possibility of an emergent tracheotomy if needed, preferentially at or just below the level of stenosis to limit the extent of additional airway injury. Ventilation in stenosis cases can be achieved via endotracheal tube, rigid bronchoscope, or supralesional jet.
Direct laryngoscopy and tracheobronchoscopy can serve as both a diagnostic and, when performed in conjunction with dilation, a therapeutic procedure. Diagnostic information attained from endoscopy should include the level, length, and severity of stenosis, as well as the mobility of the cricoarytenoid joints. Characterization of the stenosis is important. Multisubsite involvement complicates management considerably. Length of stenosis, granulation tissue, and thickness of stenosis are important to note as they can alter therapeutic decision making. In subglottic stenosis and tracheal stenosis, distance from the inferior margin of the vocal fold is important in ensuring that there is sufficient soft tissue proximally to perform a reanastomosis.
NONOPERATIVE MANAGEMENT
Nonoperative management is an important component of the long-term management of stenosis patients. This can include measures to prevent recurrence of stenosis or delay intervention with medical management.
Preventative measures include control of reflux, management of rheumatologic disease, and avoidance of chronic irritants that can cause an inflammatory response. For patients undergoing surgical intervention requiring intubation, the use of small caliber tubes reduces the incidence of intubation-related stenosis (14). For patients who require endotracheal intubation or tracheotomy, using low-pressure cuffs and keeping intracuff pressure less than 30 mm Hg greatly reduces airway injury (72,73). In addition, the use of lightweight swivel tracheal connectors and endotracheal tube support devices has been proven to be an effective resource in reducing incidence of intubation and tracheotomy-related stenosis in cases that require mechanical ventilation (4).
In symptomatic stenosis patients, oxygen supplementation, air humidification, systemic steroid application, and antibiotics when appropriate can all provide symptomatic relief in those who are acutely symptomatic. In patients with severe obstruction who are not candidates for dilation or resection, or who are symptomatic from an inflammatory exacerbation such as an upper respiratory infection, the use of heliox can be valuable in avoiding intubation, which could theoretically potentiate additional inflammation and worsen stenosis (74).
SUPRAGLOTTIC STENOSIS
Supraglottic stenosis leading to obstruction is most often in the region of the laryngeal inlet (Fig. 62.1). Postsurgical scarring, infection, caustic ingestion, and autoimmune diseases can lead to contractile scarring of the aryepiglottic folds, arytenoids, epiglottis, and intra-arytenoid region and
subsequent narrowing of the laryngeal inlet. Anterior cervical trauma that leads to injury of the thyroid cartilage and/or hyoid can displace the epiglottis posteriorly and narrow the supraglottic airway. Depending on the extent of involvement, symptoms can vary from stridor to severe airway obstruction.
subsequent narrowing of the laryngeal inlet. Anterior cervical trauma that leads to injury of the thyroid cartilage and/or hyoid can displace the epiglottis posteriorly and narrow the supraglottic airway. Depending on the extent of involvement, symptoms can vary from stridor to severe airway obstruction.
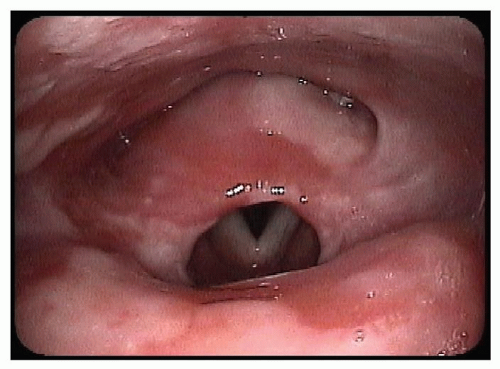 Figure 62.1 Flexible fiberoptic laryngoscopy of supraglottic stenosis in a patient with bullous pemphigus. |
Correction of supraglottic stenosis can be performed both endoscopically and via external neck incision with laryngofissure, and in extreme cases, in the form of supraglottic or suprahyoid laryngectomy (75,76). Endoscopic techniques focus on conservation of mucosa and lysis of scar via releasing procedures.
GLOTTIC STENOSIS
Glottic stenosis can be divided into three subtypes: anterior, posterior, and complete glottic stenosis. Anterior glottic stenosis, which includes laryngeal webs, can be congenital as seen in children and neonates, but also can form from bilateral anterior vocal fold trauma from surgery, caustic injury, infection, or malignancy (Fig. 62.2




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


