Ultrasound Biomicroscopy of the Anterior Ocular Segment
Zélia M. Corrêa
James J. Augsburger
Ultrasound biomicroscopy (UBM) is a high-frequency ultrasound technology that allows imaging of structural details of the anterior ocular segment at near-microscopic resolution in living patients.1,2,3 UBM provides exceptionally detailed two-dimensional grayscale images of the epibulbar conjunctiva, cornea and anterior sclera, aqueous chambers, anterior chamber angle structures, uveal and ectodermal components of the ciliary body, lens, zonule, and anterior vitreous.
Image resolution is the principal feature that distinguishes UBM from other diagnostic ultrasonographic technologies. For the purposes of this chapter, UBM is defined as an ultrasonographic cross-sectional imaging method that provides axial and lateral image resolution of approximately 50 to 60 μ or less. To achieve such image resolution, UBM needs to have a scan transducer of 35 to 50 MHz or higher. In general, the higher the transducer frequency, the higher the potential resolution of the images it can produce. Unfortunately, higher transducer frequency comes at the expense of reduced tissue penetration by the ultrasonic beam (approximately 8 mm for a 35-MHz unit, about 5 mm for a 50-MHz unit, and about 3.5 mm for an 80-MHz unit). With early generation instruments, the width and depth of the obtainable image was limited to about 5 mm. Some newer generation instruments now incorporate proprietary linear scan technology that permits unreconstructed images up to 16.0 to 18.5 mm wide (i.e., sufficient to image the entire anterior segment from limbus to limbus, sulcus to sulcus, or pars plana to pars plana in an individual cross-sectional image).
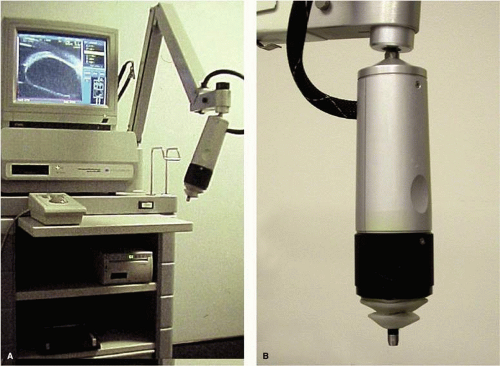
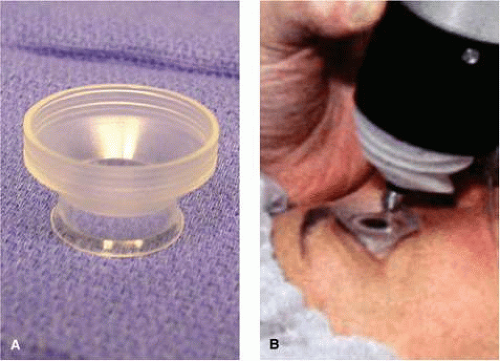
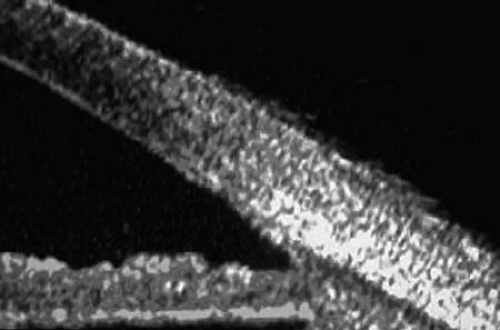
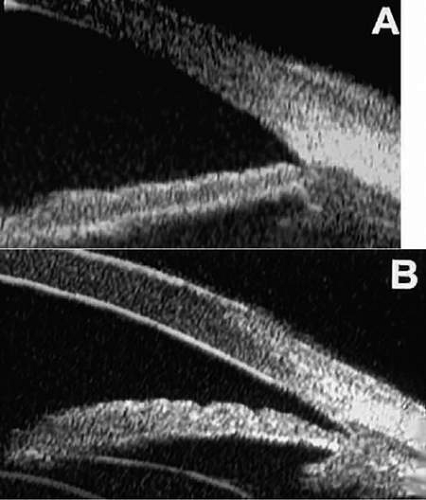
The first commercially available UBM instrument was the Humphrey Ultrasound Biomicroscope Model 840 (Humphrey Instruments Inc., San Leandro, CA). The patent for this technology was eventually acquired by Paradigm Medical Industries Inc., Salt Lake City, UT, and this company subsequently marketed similar instruments as the Paradigm models P40 and P45. Both of these instruments have a rather heavy, bulky probe that requires a counterbalanced arm to facilitate its clinical use (Fig. 1). Attached to the probe there is an exposed oscillating tip that must be submerged in a viscous coupling fluid or balanced salt solution in an eye cup lid speculum water bath (Fig. 2) to produce clinical images. Because of the need for an eye speculum water bath, this instrument can only be used on recumbent patients. Individual cross-sectional tissue slices produced by this instrument are only 5 mm long by 5 mm high. Consequently, a single image from this instrument shows a limited region of the anterior segment and cannot normally show the posterior aspect of the crystalline lens or central anterior vitreous. Several individual images must be pasted together physically or via computer graphics methods to produce wide field images.
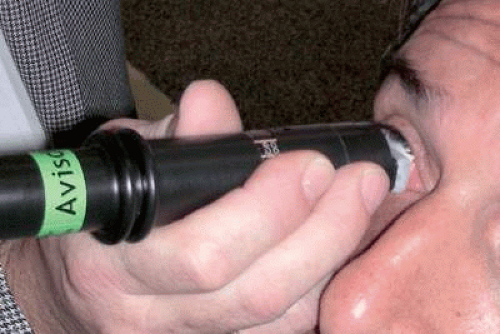
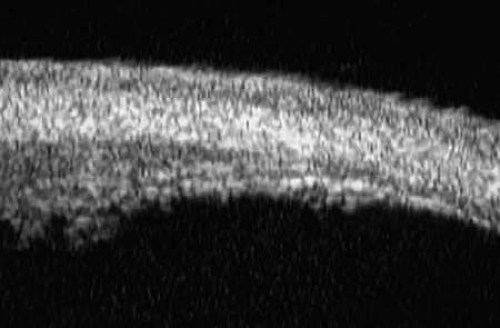
Several advances in UBM technology have occurred in recent years, including development of proprietary water bath attachments that cover the oscillating tip of the probe and permit contact imaging on seated or semirecumbent patients (Fig. 3), development of wide field image technology that permits individual cross-sectional images up to 18.5 mm long and 14 mm depth, and production of commercial instruments having interchangeable probes with transducer frequencies of 35 MHz to 80 MHz. A lower-frequency unit (e.g., 35 to 40 MHz) that produces wide field images will be particularly useful to ophthalmologists who wish to determine the precise size, shape, and location of the intact crystalline lens preoperatively, perform studies of phakic and pseudophakic accommodation, measure the sulcus-to-sulcus distance preoperatively, determine the position and relationships of the haptics of an implanted intraocular lens (IOL), or assess the dimensions and entire extent of ciliary body mass lesions and the full extent of serous or serosanguinous ciliochoroidal effusions. In contrast, a higher-frequency unit (e.g., 50 to 80 MHz) that images a smaller cross-sectional area will probably be preferred by ophthalmologists who wish to identify details of corneal or anterior scleral structure; determine the configuration of the anterior chamber angle; identify the location of minute retained foreign bodies in the cornea, sclera, anterior chamber, or iris; or evaluate the size and internal sonoreflectivity of cysts and solid tumors of the iris and ciliary body. Table 1 summarizes the pertinent features of the several UBM instruments that are commercially available as of January 2008. Most of the images shown in this chapter were obtained with either the Humphrey or Paradigm P40 50 MHz instrument.
Table 1. Commercially Available Ultrasound Biomicroscopy Instrumentsa | ||
|---|---|---|
|
One other advance that has been employed in research settings but is not yet marketed commercially is three-dimensional imaging technology.4,5 This technology has seen its greatest application in studies of accommodation in phakic eyes and pseudoaccommodation in eyes containing an implanted accommodative IOL.6,7
A complete UBM examination usually begins with imaging through the central cornea and pupil. It continues with radially oriented slices through the limbal region for 360° beginning at the 12 o’clock meridian and concludes with additional slices in various planes or regions of interest. When the examination is finished, the water bath lid speculum (if required) is removed and the residual viscous fluid is rinsed from the eye with a sterile irrigating solution. A complete study of one eye usually takes approximately 10 minutes.
Ultrasound Biomicroscopic Images of Normal Eyes
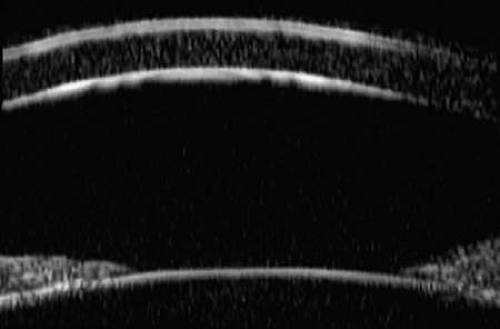
UBM images through the midcornea and pupil (Fig. 4) show the (a) cornea, (b) anterior chamber, (c) lens, and (d) pupillary zone of the iris. The cornea appears to have four well-differentiated layers. The epithelium is a thin, relatively bright (sonoreflective) layer. Just below that is Bowman’s membrane, which appears as a highly reflective, very bright line. The stroma is the thickest layer and shows relatively homogeneous low-amplitude reflectivity. The endothelium and Descemet’s membrane cannot be differentiated; together they form a single, highly reflective line at the posterior corneal surface. The aqueous in the normal anterior chamber appears completely sonolucent. The pupillary zone of the iris appears as two fingerlike structures consisting of a relatively sonolucent stroma and a bright posterior pigment epithelium. The anterior lens capsule appears as a bright retropupillary line. The normal anterior lens cortex appears nearly sonolucent.1,2,3
Radially oriented slices through the corneoscleral limbus (Fig. 5) show the (a) transition from cornea to sclera, (b) anterior chamber angle structures, (c) intermediate and peripheral zones of the iris, (d) anterior ciliary body, (e) equator of the lens, and (f) zonule. The corneoscleral junction is seen as an abrupt transition between the more sonolucent corneal stroma and the more sonoreflective sclera. The scleral spur can usually be identified as the most anterior extension of the deep sclera fibers at the limbus. The iris appears to consist of a relatively sonolucent stroma and a highly reflective pigment epithelium. The plane of curvature of the normal iris is smoothly bowed anteriorly. The insertion site of the iris relative to the scleral spur and anterior face of the ciliary body can be assessed. The pars plicata appears as fingerlike projections with acoustic features similar to those of the iris stroma. The ciliary sulcus is imaged between the peripheral iris and ciliary processes. The lens equator appears as a curved sonoreflective line (capsule) and underlying sonolucent stroma. Zonular filaments can be seen extending from the ciliary processes to the equatorial region of the lens in some eyes.8 Radial, cross-sectional slices through the posterior ciliary body (Fig. 6) show the sclera as a highly reflective structure that can be easily differentiated from the underlying uveal and neuroepithelial layers of the pars plana. Unlike a decade ago, the imaging of the ora serrata, peripheral choroid, and peripheral retina currently depends on the UBM instrument used, exam technique, and individual ocular condition.9
Additional slices through regions of interest can be obtained to evaluate specific features. For example, slices can be made through the pars plicata with the scanning plane parallel to the corneoscleral limbus (Fig. 7). Such images show the ciliary processes as a series of fingerlike projections.
Special Features
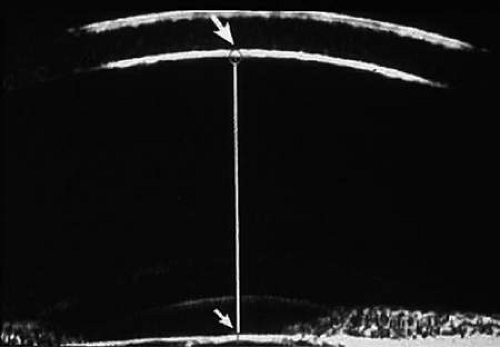
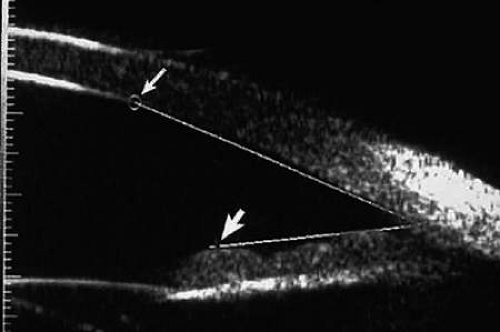
Virtually all of the currently available UBM instruments (Table 1) incorporate software that allows direct measurement of linear distances (e.g., corneal thickness, anterior chamber depth [Fig. 8], and iris thickness) on the images. The measuring cursor is set at one point on the image and then a trackball, knob on the instrument panel, or mouse is used to drag a line to the second point. The length of the line appears on the screen. Direct angular measurement of the anterior chamber angle in degrees (Fig. 9) is also possible with these instruments.
Principal Clinical Uses of Ultrasound Biomicroscopy
UBM can be used to image various structural abnormalities and lesions of the anterior segment.2,3 It has been used most extensively in glaucoma but has also contributed greatly to our current understanding of the various disorders and lesions of the cornea, iris, lens, zonule, and ciliary body.
Normal Anterior Segment Anatomy and Accommodation
UBM has been used by a variety of examiners to study the spectrum of normal findings in healthy volunteer subjects, including central corneal thickness, anterior chamber depth, and width and configuration of the anterior chamber angle and to determine the intraobserver and interobserver reproducibility of the findings.10,11,12,13 UBM has also been used extensively to study changes in lens shape and position that occur during normal accommodation.14,15
Congenital Anomalies of the Anterior Ocular Segment
UBM can show the nature and extent of congenital anterior segment abnormalities that occur in eyes with disorders such as Peter’s anomaly16 (see section on Corneal and Anterior Segment Disorders) and congenital aniridia.17 UBM has also been used in some centers to determine morphologic congenital anomalies of size and shape of the crystalline lens.18
Glaucoma and Hypotony
UBM is usually able to determine the mechanism of elevated intraocular pressure (angle closure vs. open angle) by showing the relationship between the peripheral iris and the trabecular meshwork.2,3 A strong correlation between gonioscopic and UBM estimates of angle configuration has been established.10,19 In addition, imaging of the anterior segment structures is possible even in eyes with profound corneal edema or scarring that precludes gonioscopic assessment of the angle.
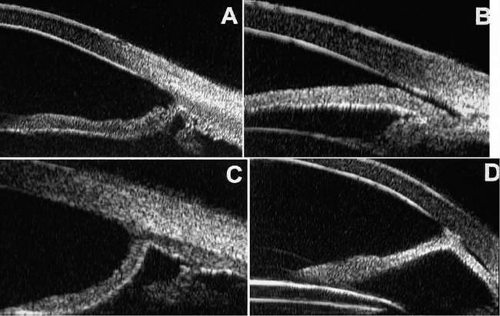
In open-angle glaucoma, UBM can be used to measure the anterior chamber angle in degrees, assess the configuration of the peripheral iris, and evaluate the trabecular meshwork2,3 (Fig. 10). The angle configuration can be graded and compared with gonioscopic findings.10,19 In certain patients with open-angle glaucoma, UBM can provide information that may be of some diagnostic value (Fig. 11). For example, in pigment dispersion syndrome20 (Fig. 11A), UBM typically reveals posterior bowing of the peripheral iris (“q” configuration of peripheral iris by Spaeth classification19). In plateau iris syndrome21 (Fig. 11B), UBM usually reveals abnormally steep anterior angulation of the peripheral iris (“s” configuration of peripheral iris by Spaeth classification19), insertion of the iris from the anterior ciliary body, and retroiridic projection of the ciliary processes. In eyes with peripheral anterior synechiae (Figs. 11C–D), UBM can reveal the extent of iridocorneal adhesion even if the cornea is hazy or opaque.
In eyes with a narrow angle, UBM shows the extent of angle closure, reveals the depth of the anterior and posterior chambers, and identifies pathologic processes pushing the lens and iris forward2,3,22 (Fig. 12). UBM has been able to differentiate between primary angle closure (i.e., cases of angle closure without additional pathology responsible for the anterior lens–iris displacement [Fig. 12A]) and secondary angle closure due to processes such as lens swelling and dislocation (Fig. 12B), massive hemorrhagic retinal detachment pushing the lens and iris anteriorly (Fig. 12C), and multiple neuroepithelial cysts of the iridociliary sulcus (Fig. 12D).
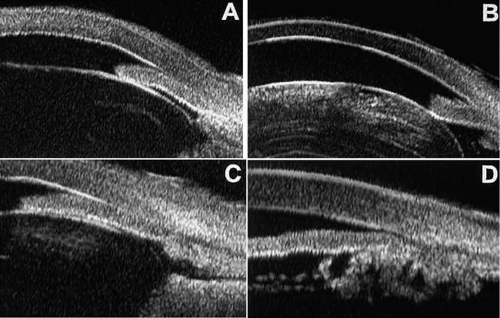 Figure 12. Angle configuration in eyes with angle-closure glaucoma. A: Primary angle-closure glaucoma with anterior displacement of the lens and iris. B: Angle closure secondary to a swollen cataractous lens (phakomorphic angle closure). C: Angle closure secondary to massive hemorrhagic retinal detachment; the subretinal blood is evident in the lower right corner of the photograph. D: Angle closure secondary to multiple peripheral iris cysts.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|