Although various treatments for tympanic membrane (TM) perforation have been used, almost all of them, especially in cases of larger TM perforations, are surgical treatments, such as myringoplasty or tympanoplasty. Previous studies have demonstrated the ability to repair small, chronic TM perforations using collagen sheets or other types of patches.1,2 For larger perforations spanning greater than one-third of the TM, however, conventional surgical treatment is usually still required. These surgeries require a skin incision and harvesting of autologous tissue, which is then used as repair material for the TM. Traditional otosurgical treatments may demand that patients endure some combination of anesthesia, hospitalization and/or bed rest, the high price of a surgical operation, and restrictions on daily life. In addition, these surgical techniques often pose other risks, including the development of iatrogenic cholesteatoma, otitis media, sensorineural hearing loss, and dysgeusia.
Given the disadvantages associated with conventional surgical treatments, research has been performed to investigate the causes of healing arrest in chronic TM perforations and the mechanisms involved in this process.3,4,5 Acute small perforations of the TM usually heal spontaneously while chronic perforations and large perforations are less likely to do so. However, if given an adequate arrangement of scaffolding and growth factors to support the healing process, large chronic TM perforations may regenerate. Recent progress in regenerative medicine has provided us with various kinds of scaffolds and growth factors that make it possible to accelerate this type of tissue repair.6,7
Because the TM generally demonstrates good regenerative capabilities, we hypothesized that a better selection of scaffolds and other extrinsic factors, applied under the appropriate conditions, could bring out its maximum regenerative potential. According to the principles of in situ tissue engineering, regeneration of tissues/organs requires the introduction of three elements: cells, scaffolds, and regulatory factors, under suitable conditions.6,7 In the TM regenerative scheme described here, scaffolding (in the form of a gelatin sponge) and a regulatory factor (basic fibroblast growth factor [bFGF]) are provided, while cells are endogenously supplied from the mechanically disrupted edge of the perforation. Mechanical disruption of the perforation edge may be the trigger to stimulate progenitor/tissue stem cells in this region of the TM.8 This is another significant advantage of this novel therapy, as cell transplantation would make it difficult to widely and easily apply this therapy as an outpatient treatment.
By applying the principles of modern tissue engineering, we developed a novel, minimally invasive therapy for regenerative repair of large or total TM perforations (▶ Fig. 46.1) that may represent a new stage in the advancement of otologic surgery. In what follows, a description of TM regeneration is provided in the framework of our recently published prospective clinical trial.9
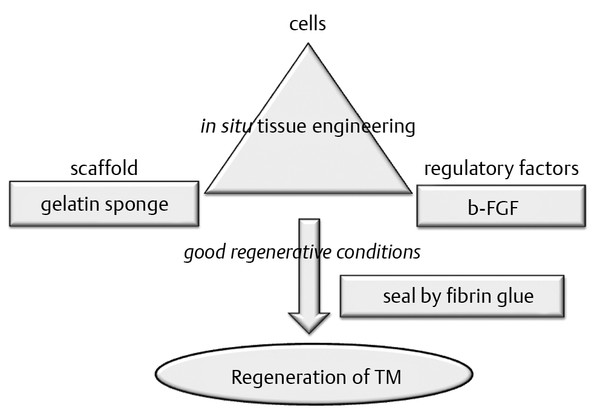
Fig. 46.1 Strategy for regeneration of the TM. Based on the concept of in situ tissue engineering, we introduce just two of these elements, gelatin sponge for scaffolding and bFGF for a regulatory factor, as cells are expected to be endogenously supplied from the mechanically disrupted edge of the perforation. For creating optimal regenerative conditions, we seal the gelatin sponge with fibrin glue.
46.2 Patient Selection
The criteria used to screen patients for TM regeneration are:
Dry TM and tympanic cavity without active inflammation during the previous 6 months.
Proper aeration and no regions of soft tissue density in the mastoid and tympanic cavities based on temporal bone CTs.
Intact ossicular chain.
No cholesteatoma and no invasion of epithelia to tympanic cavity.
During our study we selected 140 patients with 158 chronic TM perforations from outpatients with a dry TM and tympanic cavity without active inflammation during the previous 6 months. The diseases affecting these patients included chronic otitis media (n=95), postoperative re-perforated TM (n=16), traumatic perforation (n=21), and iatrogenic TM perforation following insertion of a ventilation tube (n=26). Eighteen patients had bilateral TM perforations.
Patients ranged in age from 10 to 91 years, with an average age of 59 years. The patients consisted of 59 males and 81 females. The TM perforations were classified into three groups based on the size of the perforation, as follows: <1/3 as Grade I (n=39), 1/3 to 2/3 as Grade II (n=69), and >2/3 as Grade III (n=50).
46.3 Materials
Materials used for TM regeneration include gelatin sponge (Spongel, Astellas Pharma Inc., Tokyo, Japan) (▶ Fig. 46.2), basic fibroblast growth factor (bFGF, Fibrast, Kaken Pharma Co., Ltd, Tokyo, Japan), and fibrin glue (Bolheal, Kaketsuken, Kumamoto, Japan; Beriplast, CSL Behring K.K., Tokyo, Japan).

Fig. 46.2 Gelatin sponge. Left side: Trimmed gelatin sponge; right side: Trimmed gelatin sponge with bFGF.
Basic-FGF is one of the few commercially produced growth factors available for clinical applications. It is usually used for the treatment of skin ulcers or decubitus ulcers because it has strong inductive effects on both fibroblasts (from which the name comes) and blood capillaries.10,11 This former property is particularly suitable for TM regeneration because the pars tensa, which represents the largest part of the TM, consists of an intermediate layer of fibrous tissue produced by fibroblasts.11 The capillary-inducing ability of bFGF may also foster improvements in local regenerative conditions. Although several reports have tested the use of growth factors and repair materials for improving TM regeneration in animal models,2,12 an ideal combination of factors and materials has yet to be determined. The results of these controlled studies, however, suggest that bFGF may be suitable to act as the regulatory factor for optimal TM regeneration.
Given the limitations of sheet-form scaffolds, we use gelatin sponge as our scaffold material of choice. Gelatin sponge is made of a protein extracted from the collagen found inside connective tissues of animals and is often used for its hemostatic properties in medical procedures. Gelatin sponge has a high affinity for living tissues and is absorbed in vivo within a few months.13,14 Another benefit of gelatin sponge is that it can fit into any size/shape of TM perforation and is easy to handle because of its gel form. Observations of the regenerative processes occurring in the TM reveal that cells will grow into a gelatin sponge and can occupy a large space. Therefore, with this technique a portion of the gelatin sponge extends into the TM, whereas another portion remains outside of the regenerating TM, providing a large surface area for cell growth. Gelatin sponge that remains in the tympanic cavity following TM regeneration is gradually absorbed and disappears within 3 months. A gelatin sponge works not only as a scaffold for the regeneration of the TM, but also as a sustained release substrate for bFGF.14,15 These characteristics make it particularly advantageous for use in regeneration of the TM.
In the final step of our procedure, we seal the gelatin sponge with fibrin glue to insulate it from the outside and create optimal regenerative conditions. Not only does the fibrin glue help to keep the sponge in place, it also prevents cells from drying out and provides a barrier to infection, thus providing ideal culture conditions in vivo.
46.4 Surgical Technique
The tympanic region is fully anesthetized by applying a cotton ball soaked in 4% lidocaine to the perforation, in contact with the residual TM, for 15 minutes. Mechanical disruption of the perforation edge is then created under the microscope with a myringotomy knife. A gelatin sponge that is larger than the perforation is immersed in bFGF (5–30 μg of trafermin, a genetic recombinant, used at 100 μg/mL) and inserted into the perforation in contact with the edge of the TM. In our study, this was applied to 148 ears and saline, inserted in a similar fashion, was used as a control in 10 ears. Fibrin glue is then dripped over the sponge. ▶ Fig. 46.3 illustrates this process.
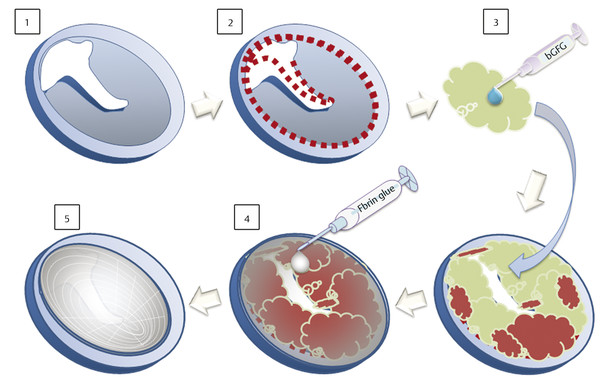
Fig. 46.3 Schematic diagram showing the method of TM regeneration. (1) TM perforation. (2) A mechanical disruption of the TM perforation edge is created under the microscope following local anesthesia with 4% lidocaine. (3) A gelatin sponge immersed in bFGF is placed over the perforation, in contact with the residual TM. (4) A gelatin sponge that has absorbed blood and changed color. Fibrin glue is dripped over the sponge. (5) At 3 weeks after treatment, residual crust is removed. In cases of incomplete closure of the TM perforation, the treatment is performed repeatedly.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


