Tumors of the Conjunctiva and Cornea
Carol L. Shields
Jerry A. Shields
Supported by the Eye Tumor Research Foundation, Inc., Philadelphia, PA.
GENERAL CONSIDERATIONS
Tumors of the conjunctiva and cornea occupy a large spectrum of conditions ranging from benign inflammatory lesions such as pingueculitis or episcleritis to aggressive, life-threatening malignancies such as melanoma or Kaposi sarcoma.1,2,3,4,5,6 The clinical differentiation of these tumors is based on the patient’s medical background as well as recognizable clinical features of the tumor. The identification and proper management of such tumors requires an understanding of the anatomy of the conjunctiva and cornea and knowledge of general principles of tumor management, both of which are described in this chapter. The specific clinical and histopathologic features as well as the management of each tumor are discussed, based on the authors’ personal experience with more than 2,000 patients with conjunctival tumors during a three-decade period.3
Anatomy
The conjunctiva is a continuous mucous membrane that covers the anterior portion of the globe. It extends from the eyelid margin onto the back surface of the eyelid (palpebral portion), into the fornix (forniceal portion), onto the surface of the globe (bulbar portion), and up to the corneoscleral limbus (limbal portion). The conjunctiva is composed of epithelium and stroma. The epithelium consists of both stratified squamous and columnar epithelium. The squamous pattern is found near the limbus, and the columnar pattern is found near the fornix. The stroma is composed of fibrovascular connective tissue that thickens in the fornix and thins at the limbus.
Special regions of the conjunctiva include the plica semilunaris and caruncle. The plica semilunaris is a vertically oriented fold of conjunctiva, located in the medial portion of the bulbar conjunctiva near the medial eyelid canthus. It is speculated that the plica semilunaris represents a remnant of the nictitating membrane found in certain animals. The caruncle is located in the medial canthus between the upper and lower punctum. It contains both conjunctival and cutaneous structures such as nonkeratinized stratified squamous epithelium overlying the stroma of fibroblasts, melanocytes, sebaceous glands, hair follicles, accessory lacrimal tissue, and striated muscle fibers.
The conjunctiva can spawn neoplasms from both its epithelial and stromal structures. These are similar clinically and histopathologically to tumors that arise from other mucous membranes in the body. Similarly, the cornea can develop epithelial tumors, but corneal stromal tumors are extremely rare. The caruncle, with its unique composition of both mucous membrane and cutaneous structures, can generate tumors found in both mucosa and skin.
Diagnostic Approaches
Unlike many other mucous membranes in the body, the conjunctiva is readily visible. Thus, tumors and related lesions that occur in the conjunctiva are generally recognized at a relatively early stage. Since many of these tumors have typical clinical features, an accurate diagnosis can often be made by external ocular examination and slitlamp biomicroscopy, provided that the clinician is familiar with their clinical characteristics. A diagnostic biopsy is not usually necessary in cases of smaller tumors that appear benign. If a smaller tumor does require a biopsy, it is often better to completely remove the lesion in one operation (excisional biopsy). In cases of larger lesions, however, it may be appropriate to remove a portion of the tumor (incisional biopsy) to obtain a histopathologic diagnosis prior to embarking on more extensive therapy. In some cases, exfoliative cytology or fine-needle aspiration biopsy can be performed to spare the patient surgery. This is especially useful for surface neoplasia and sometimes helpful for melanocytic conjunctival tumors.7
In addition to evaluation of the conjunctival lesion, meticulous slitlamp examination of the cornea is essential in patients with suspected conjunctival tumors. Invasion of squamous cell carcinoma and melanoma into the peripheral cornea may appear as a subtle, gray surface opacity. It is important to completely outline such corneal involvement prior to surgery, since it is often less visible through the operating microscope than it is with slitlamp biomicroscopy in the office.
Management
Depending on the presumptive diagnosis and the size and extent of the lesion, the management of a conjunctival tumor can be complex and involve numerous solitary or combined therapies. These consist of serial observation; incisional biopsy; excisional biopsy; cryotherapy; antibiotics; topical or systemic chemotherapy; local injection or systemic monoclonal antibody; topical, injection, or systemic interferon; teleradiotherapy or brachyradiotherapy; modified enucleation; or orbital exenteration.8,9,10 If large areas of conjunctiva are removed, mucous membrane grafts from the conjunctiva of the opposite eye, buccal mucosa, or amniotic membrane may be necessary.11
Observation
Observation is generally the management of choice for most benign, asymptomatic tumors of the conjunctiva. Selected examples of lesions that can be observed without interventional treatment include pingueculum, dermolipoma, and nevus. External or slit lamp photographs are advisable to document all lesions and are critical to follow-up of the more suspicious lesions. Most patients are examined every 6 to 12 months looking for evidence of growth, malignant change, or secondary effects on normal surrounding tissues.
Incisional Biopsy
Incisional biopsy is reserved for extensive suspicious tumors that are symptomatic or suspected to be malignant. Examples include large squamous cell carcinoma, primary acquired melanosis, melanoma, and conjunctival invasion by sebaceous gland carcinoma. It should be understood that if such tumors occupy four to six clock hours or less on the bulbar conjunctiva, excisional biopsy is generally preferable to incisional biopsy. However, larger lesions can be approached by incisional wedge biopsy or punch biopsy. Further definitive therapy would then be planned based on the results of biopsy. Incisional biopsy is also appropriate for conditions that are ideally treated with topical or systemic chemotherapy, interferon, or monoclonal antibody (rituximab), or those treatable with radiotherapy. Such lesions include squamous cell carcinoma, conjunctival intraepithelial neoplasia, primary acquired melanosis, papillomatosis, lymphoid tumors, and metastatic tumors.
Excisional Biopsy
Primary excisional biopsy is appropriate for intermediate and small tumors that are symptomatic or suspected to be malignant. In such situations, excisional biopsy is preferred over incisional biopsy to avoid inadvertent tumor seeding. Examples of benign and malignant lesions that are ideally managed by excisional biopsy include symptomatic limbal dermoid, epibulbar osseous choristoma, steroid-resistant pyogenic granuloma, squamous cell carcinoma, and melanoma. When such lesions are located in the conjunctival fornix they can be completely excised and the conjunctiva reconstructed primarily with absorbable sutures, sometimes with fornix deepening sutures or symblepharon ring to prevent adhesions. If the defect cannot be closed primarily, then a transpositional flap or graft with mucous membrane or amnion can be inserted.
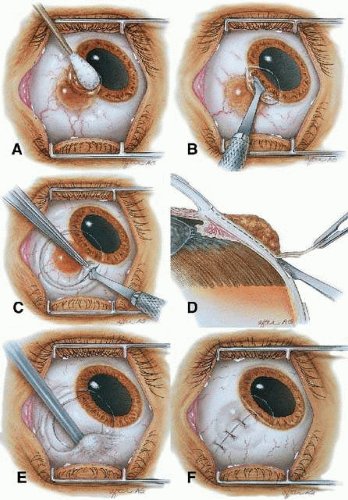
Most primary malignant tumors of the conjunctiva, such as squamous cell carcinoma and melanoma, arise in the interpalpebral area near the limbus, and the surgical technique for limbal tumors is different than that for forniceal tumors.1,8 Limbal neoplasms have a propensity to invade through the corneal epithelium and sclera into the anterior chamber and also through the soft tissues into the orbit. Thus, it is often necessary to remove a thin lamella of underlying sclera to achieve tumor-free margins and to decrease the chance for tumor recurrence. In this regard, we use a partial lamellar sclerokeratoconjunctivectomy with primary closure for such tumors (Fig. 10-1). Because cells from these friable tumors can seed into adjacent tissues, a precise technique without touching the tumor (no-touch technique) is mandatory. Additionally, the surgery should be performed using microscopic technique and the operative field should be left dry so that cells adhere to the resected tissue. It is best to avoid wetting the field with balanced salt solution until after the tumor is completely removed to minimize seeding of cells.
The technique for resection of limbal tumors is shown in Figure 10-1. Using retrobulbar anesthesia and the operating microscope, the corneal epithelial component is approached first and the conjunctival component is dissected second, with the goal of excising the entire specimen completely in one piece. Absolute alcohol soaked on a Weck-cel sponge is gently applied to the corneal component. This causes epithelial cellular devitalization, drying of the tissue, and allows easier release of the tumor cells from the Bowman layer. A Beaver blade is used to microscopically scroll off the involved epithelium starting at the centralmost portion and working toward the limbus. An epitheliorhexis technique about 2 mm outside the corneal component is the preferred technique. Next, a pentagonal incision based at the limbus is made 3 to 4 mm outside the tumor margin. Radial incisions on either side of the limbal mass in an anteroposterior direction followed by posterior connector incision complete the perilesional cuts. The incision is carried through the underlying Tenon fascia until the sclera is exposed so that full-thickness conjunctiva and Tenon fascia are incorporated into the excisional biopsy. Cautery is applied to control bleeding. Using a 57 Beaver blade, the limbal portion is removed with the blade flat and dissecting the tumor plus 1 mm of underlying thin scleral bed. This is necessary only at the limbus directly underneath the tumor to ensure that the entire tumor with tumor-free margins is removed in one piece. The tumor itself is never handled as only normal surrounding tissue is manipulated (no-touch technique). The removed specimen is then placed flatly on a piece of thin cardboard from the surgical tray and then placed in fixative and submitted for histopathologic studies. This step prevents the specimen from folding and allows better assessment of the tumor margins histopathologically. The used instruments are then replaced with fresh instruments for subsequent steps, to avoid contamination of healthy tissue with possible tumor cells.
After excision of the specimen, cryotherapy is applied to the margins of the remaining bulbar conjunctiva. This is performed by freezing the surrounding bulbar conjunctiva as it is lifted away from the sclera using the cryoprobe. A double freeze-thaw technique is used to treat all conjunctival margins, but not the corneal margin. The corneal margin and tumor bed are treated with absolute alcohol wash on Weck-cel sponge with removal of residual episcleral and corneal debris using a blunt sweeping technique of the cells onto dry sponge. Bipolar cautery allows for later hemostasis. Avoid direct cryotherapy to the sclera.
Using clean instruments, the conjunctiva is mobilized for closure of the defect by loosening the intermuscular septum with Steven scissor spreading and creation of sliding or transpositional conjunctival flaps. Larger defects are closed with mucosal grafts. Closure is completed with interrupted absorbable 6-0 or 7-0 sutures. In some cases, grafts and flaps are closed with tissue glue. If the surgeon prefers, an area of bare sclera can be left near the limbus, but we prefer complete closure as this promotes better healing and allows for facility of further surgery if the patient should develop recurrence. The patient is treated with topical antibiotics and corticosteroids for 2 weeks and then followed at 3- to 6-month intervals.
Cryotherapy
In the management of conjunctival tumors, cryotherapy can be used as a supplemental treatment to excisional biopsy as described previously. In such cases it can eliminate microscopic tumor cells and prevent recurrence of malignant tumors such as squamous cell carcinoma and melanoma. It can also be used as a principal treatment for papilloma, primary acquired melanosis, and pagetoid invasion of sebaceous gland carcinoma. If cryotherapy can devitalize the malignant or potentially malignant cells in such instances, radical surgery such as orbital exenteration can often be delayed or avoided.
Chemotherapy
Recent evidence has revealed that topical eyedrops composed of mitomycin C, 5-fluorouracil, or interferon are effective in treating epithelial malignancies such as squamous cell carcinoma, primary acquired melanosis, and pagetoid invasion of sebaceous gland carcinoma.10,12,13,14,15,16,17,18,19,20,21,22 These agents are used most successfully for squamous cell carcinoma, especially after tumor recurrence following previous surgery. Mitomycin C is prescribed topically for four times daily for a 1-week period followed by a 1-week hiatus to allow the ocular surface to recover (Table 10-1). This cycle is repeated once again so that most patients receive a total of 2 weeks of the chemotherapy topically over a 1-month period. Both mitomycin C and 5-fluorouracil are more effective for squamous cell carcinoma than for primary acquired melanosis and pagetoid invasion of sebaceous gland carcinoma. Toxicities include dry eye findings, superficial punctate epitheliopathy, and punctal stenosis. Corneal melt, scleral melt, and cataract can develop if these agents are used with open conjunctival wounds or used excessively. Topical interferon can be effective for squamous epithelial malignancies and is less toxic to the surface epithelium. Interferon can be delivered in a subconjunctival injection and is likewise well tolerated but perhaps with more systemic malaise. Topical interferon is delivered as 1 million units per cc four times daily for several months (Table 10-1).
Table 10-1 Protocol for use of topical chemotherapy or interferon for conjunctival neoplasia | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
Radiotherapy
Two forms of radiotherapy are used for conjunctival tumors, namely external beam radiotherapy and custom-designed plaque radiotherapy. External beam radiotherapy to a total dose of 3,000 to 4,000 cGy is used to treat conjunctival lymphoma and metastatic carcinoma when such lesions are too large or diffuse to excise locally. Side effects of dry eye, punctate epithelial abnormalities, and cataract should be anticipated.
Custom-designed plaque radiotherapy23,24,25 to a dose of 3,000 to 4,000 cGy can be used to treat conjunctival lymphoma or metastasis. A higher dose of 6,000 to 8,000 cGy can be used to treat the more radiation-resistant melanoma and squamous cell carcinoma. In general, plaque radiotherapy is reserved for those patients who have diffuse tumors that are incompletely resected and for those who display multiple recurrences. The two designs for conjunctival custom plaque radiotherapy include a conformer plaque technique placed on the eye for a 1-week period as an outpatient or a nonshielded plaque technique with the device sutured onto the episcleral tissue as an outpatient. Such treatment should be performed by experienced radiation oncologists and ocular oncologists.
Modified Enucleation
Modified enucleation is a treatment option for primary malignant tumors of the conjunctiva that have invaded through the limbal tissues into the globe, producing secondary glaucoma. Such an occurrence is quite rare but can occasionally occur with squamous cell carcinoma and melanoma. The uncommon mucoepidermoid variant of squamous cell carcinoma of the conjunctiva has a greater tendency for such invasion. At the time of enucleation, it is necessary to remove the involved conjunctiva intact with the globe so as to avoid spreading tumor cells. Thus, the initial peritomy should begin at the limbus, but when the tumor is approached, the incision should proceed posteriorly from the limbus to surround the tumor-affected tissue by at least 3 to 4 mm. The tumor will remain adherent to the globe at the limbus. Occasionally, a suture is used through the surrounding conjunctiva into the episclera to secure the tumor to the globe so that it will not be displaced during subsequent manipulation. The remaining steps of enucleation are gently performed and the globe is removed with tumor adherent after cutting the optic nerve from the nasal side. The margins of the remaining, presumed unaffected conjunctiva are treated with double freeze-thaw cryotherapy. Often this surgical technique leaves the patient with a limited amount of residual unaffected conjunctiva for closure. In these instances, a mucous membrane graft, amniotic membrane graft, dermis fat graft, or dermis graft may be necessary for adequate closure and to provide fornices for a prosthesis. In some instances, a simple horizontal inferior forniceal conjunctival incision from canthus to canthus may suffice, as long as the conformer is constantly worn as a template so the new conjunctival fornix grows deep and around this structure.
Orbital Exenteration
Orbital exenteration is probably the treatment of choice for primary malignant conjunctival tumors that have invaded the orbit or that exhibit complete involvement of the conjunctiva.1,9,25,26 Either an eyelid-removing or eyelid-sparing exenteration is used, depending on the extent of eyelid involvement. The eyelid-sparing technique is preferred in that the patients have better cosmetic appearance and heal within 2 or 3 weeks. Specifically, if the anterior lamella of the eyelid is uninvolved with tumor, an eyelid-sparing (eyelid-splitting) exenteration may be accomplished.
Mucous Membrane Graft
Mucous membrane grafts are occasionally necessary to replace vital conjunctival tissue after removal of extensive conjunctival tumors. The best donor sites include the forniceal conjunctiva of the ipsilateral or contralateral eye and buccal mucosa from the posterior aspect of the lower lip or lateral aspect of the mouth. Such grafts are usually removed by a freehand technique, fashioned to fit the defect, and secured into place with cardinal and running absorbable 6-0 or 7-0 sutures. Currently, in most instances, we use a donor amniotic membrane graft to replace lost conjunctiva.10,11 The tissue is delivered frozen and must be defrosted for 20 minutes. The fine, transparent material is carefully peeled off its cardboard surface, laid basement membrane side up, and sutured into place with absorbable sutures or glued with tissue glue. Topical antibiotic and steroid ointments are applied following all conjunctival grafting procedures.
It is important that the surgeon use a minimal manipulation technique for tumor resection. For graft harvest and placement, the surgeon should always use clean, sterile instruments at both the donor and the recipient sites. Free tumor cells can rest on instrument tips and later implant and grow in previously uninvolved areas if such precautions are not taken.
CONGENITAL LESIONS
Various tumors and related conditions may be present at birth or become clinically apparent shortly after birth.4,25 Most of the lesions to be considered here are choristomas, consisting of tissue elements that are not normally present at the involved site. Despite their presence at a young age, all of the conjunctival choristomas discussed herein are sporadic, without hereditary tendency.
Dermoid
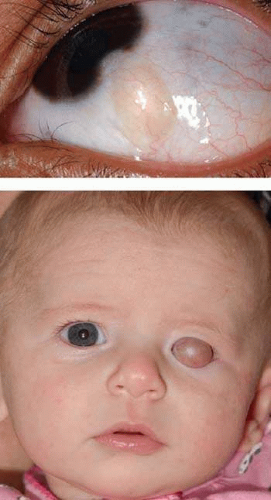
Conjunctival dermoid is a congenital well-circumscribed yellow-white solid mass that involves the bulbar conjunctiva or the corneoscleral limbus.4,25,26,27 It characteristically occurs near the limbus inferotemporally, and often this tumor has fine white hairs, best seen with slitlamp biomicroscopy (Fig. 10-2). In rare cases, it can extend to the central cornea or be located in other quadrants on the bulbar surface. It may occur as an isolated lesion, or it can be associated with Goldenhar syndrome. Hence, the patient should be evaluated for ipsilateral or bilateral preauricular skin appendages, hearing loss, eyelid coloboma, orbitoconjunctival dermolipoma, and cervical vertebral anomalies that constitute this nonheritable syndrome. Histopathologically, the conjunctival dermoid is a simple choristomatous malformation that consists of dense fibrous tissue lined by conjunctival epithelium with deeper dermal elements such as hair follicles and sebaceous glands.
The management of an epibulbar dermoid includes simple observation if the lesion is small and visually asymptomatic. It is possible to excise the lesion for cosmetic reasons, but the remaining corneal scar is sometimes cosmetically unacceptable. Larger or symptomatic dermoids can produce visual loss from astigmatism. These can be approached by lamellar keratosclerectomy with primary closure of overlying tissue if the defect is superficial or closure using corneal graft if the defect is deep or full thickness. It has been reported that the cosmetic appearance may improve, but the refractive and astigmatic error and visual acuity may not change.27 When the lesion involves the central cornea, a lamellar or penetrating keratoplasty may be necessary and long-term amblyopia can be a problem.
Dermolipoma
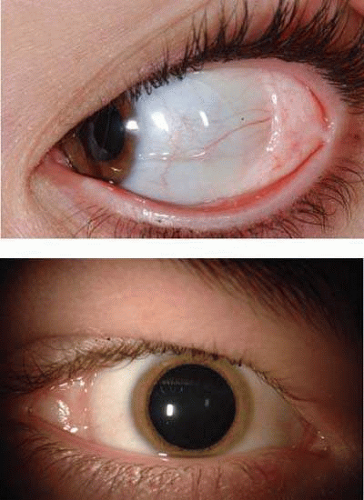
Dermolipoma is believed to be congenital and present at birth, but it typically remains asymptomatic for years and may not be detected until adulthood when it protrudes from the orbit through the conjunctival fornix superotemporally (Fig. 10-3). It appears as a pale yellow, soft, fluctuant, fusiform mass below the palpebral lobe of the lacrimal gland, best visualized with the eye in inferonasal gaze. It usually extends for a variable distance into the orbital fat and onto the bulbar conjunctiva, and occasionally it can extend anteriorly to reach the limbus. Unlike herniated orbital fat, dermolipoma can contain fine white hairs on its surface and it cannot be reduced with digital pressure into the orbit.
With computed tomography (CT) or magnetic resonance imaging (MRI), dermolipoma has features similar to orbital fat from which it may be indistinguishable. Histopathologically, it is lined by conjunctival epithelium on its surface and the subepithelial tissue has variable quantities of collagenous connective tissue. Pilosebaceous units and lacrimal gland tissue may occasionally be present. Most dermolipomas require no treatment, but larger symptomatic ones or those that are cosmetically unappealing can be managed by excision of the entire orbitoconjunctival lesion through a conjunctival forniceal approach or by simply removing the anterior portion of the lesion in a manner similar to that used to remove prolapsed orbital fat.
Epibulbar Osseous Choristoma
Epibulbar osseous choristoma is a rigid deposit of bone generally located in the bulbar conjunctiva superotemporally28 (Fig. 10-4). It is believed to be congenital and typically remains undetected until personally palpated by the patient in the preteen years. It is clinically suspected due to its rock hard consistency on palpation, although fibrous tissue tumors can feel similar. The diagnosis can be confirmed with ultrasonography or computed tomography to illustrate the calcium component. This tumor is generally best managed by periodic observation. Occasionally patients report a foreign body sensation, and such symptomatic lesions can be excised with a circum-tumoral conjunctival incision followed by dissection to bare sclera for full-thickness conjunctival resection. For those tumors that might be adherent to the sclera, a superficial sclerectomy might be warranted.
Lacrimal Gland Choristoma
Lacrimal gland choristoma is a congenital lesion often discovered in young children as an asymptomatic pink stromal mass, most often in the inferior bulbar or forniceal conjunctiva. It is speculated that this lesion presents in such location due to the pathway that the lacrimal gland takes during embryogenesis from the inferior to the superotemporal region. The lacrimal gland choristoma can masquerade as a focus of inflammation due to its pink color. Rarely, a cystic appearance ensues from this secretory mass if there is no connection to the conjunctival surface. Excisional biopsy is usually performed to confirm the diagnosis.
Respiratory Choristoma
In unique instances, a cystic choristoma, appearing as congenital sclerocorneal ectasia, is found. In one report, such a case was found to manifest respiratory mucosa.29
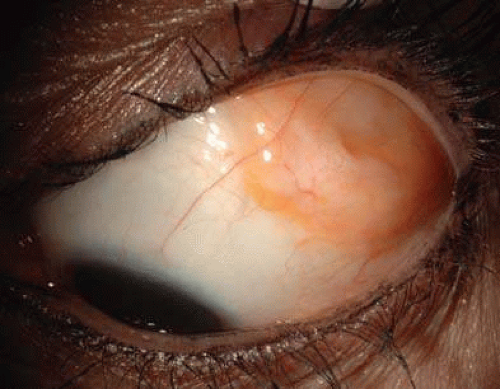
Complex Choristoma
The conjunctival dermoid and epibulbar osseous choristoma are termed simple choristomas as they contain one tissue type such as skin or bone. A complex choristoma contains a greater variety of tissue such as dermal appendages, lacrimal gland tissue, cartilage, bone, and occasionally other elements. It is quite variable in its clinical appearance and may cover much of the epibulbar surface or it may form a circumferential growth pattern around the limbus (Fig. 10-5). For example, such tumor with extensive lacrimal tissue appears as a lobular pink mass whereas one with dermal tissue appears yellow and thick and one with cartilage displays a smooth blue-gray hue. The complex choristoma has an association with the linear nevus sebaceous of Jadassohn.30,31 The nevus sebaceous of Jadassohn includes cutaneous features such as sebaceous nevus in the facial region and neurologic features such as seizures, mental retardation, arachnoid cyst, and cerebral atrophy. The ophthalmic features of this syndrome include epibulbar complex choristoma and posterior scleral cartilage.
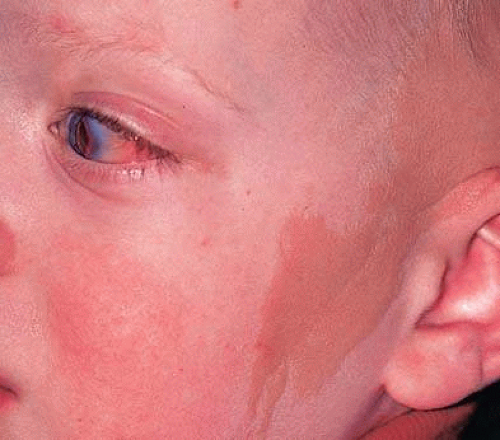 Figure 10-5. Cutaneous nevus sebaceous of Jadassohn with temporal epibulbar complex choristoma as features of organoid nevus syndrome. |
The management of the complex choristoma depends on the extent of the lesion. Observation or wide local excision with mucous membrane graft reconstruction are options. In the rare case of a very extensive lesion, in which the lesion causes dense amblyopia with no hope for visual acuity, modified enucleation with ocular surface reconstruction may be necessary.
BENIGN TUMORS OF SURFACE EPITHELIUM
Several benign tumors and related conditions can arise from the squamous epithelium of the conjunctiva.
Papilloma
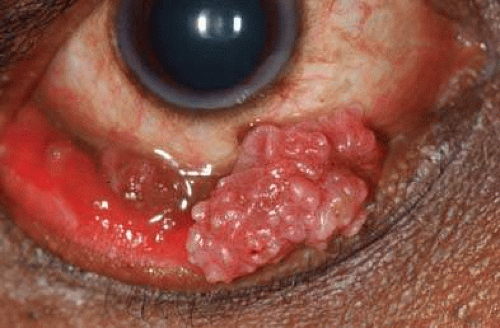
Squamous papilloma is a benign tumor, documented to originate from human papillomavirus infection of the conjunctiva.32,33 This tumor can occur in both children and adults. It is speculated that the virus is acquired through transfer from the mother’s vagina to the newborn’s conjunctiva as the child passes through the mother’s birth canal. Papilloma appears as a pink fibrovascular frond of tissue arranged in a sessile or pedunculated configuration. The numerous fine vascular channels ramify through the stroma beneath the epithelial surface of the lesion. In children, the lesion is usually small, multiple, and located in the inferior fornix (Fig. 10-6). In adults, it is usually solitary, more extensive, and can often extend to cover the entire corneal surface simulating malignant squamous cell carcinoma. Histopathologically, the lesion shows numerous vascularized papillary fronds lined by acanthotic epithelium.
In the case of a small sessile papilloma in a child, a trial of topical corticosteroids might bring about resolution of the lesion. Sometimes, periodic observation allows for slow spontaneous resolution of the viral-produced tumor. Larger or more pedunculated lesions are generally symptomatic with foreign body sensation, chronic mucus production, hemorrhagic tears, incomplete eyelid closure, and poor cosmetic appearance. Such lesions are best managed by surgical excision. Complete removal of the mass without direction manipulation of the tumor (no-touch technique) is generally advisable to avoid spreading of the tumor-related virus. Double freeze-thaw cryotherapy is applied to the remaining conjunctiva around the excised lesion to help prevent tumor recurrence. In some instances, the pedunculated tumor is frozen alone and allowed to slough off the conjunctival surface later. For some large, unwieldy pedunculated tumors, complete cryotherapy of the mass down its stalk to its base is performed and excision while the mass is in the frozen state is achieved. This is especially important for large lesions to allow for traction on the tumor without forceps manipulation. Closure is completed with absorbable sutures. For those lesions that show recurrence, oral cimetidine or topical interferon for several months can resolve the papilloma virus-related tumor by boosting the patient’s immune system and stimulating regression of the mass.33
Keratoacanthoma
The conjunctiva can give rise to benign reactive inflammatory lesions that simulate carcinoma such as pseudocarcinomatous hyperplasia and its variant, keratoacanthoma.34 In some instances a distinct nodule is found. This lesion appears gelatinous or leukoplakic, similar to squamous cell carcinoma of the conjunctiva, but its onset may be more rapid. Massive acanthosis, hyperkeratosis, and parakeratosis are found histopathologically.
Hereditary Benign Intraepithelial Dyskeratosis
Hereditary benign intraepithelial dyskeratosis (HBID) is a peculiar condition seen in an inbred isolate of Caucasians, African Americans, and Native Americans (Haliwa Indians). This group resided initially in North Carolina. HBID has subsequently been detected in several other parts of the United States. It is an autosomal dominant disorder characterized by bilateral elevated fleshy plaques on the nasal or temporal perilimbal conjunctiva.35 Similar plaques can occur on the buccal mucosa. It can remain relatively asymptomatic or it can cause severe redness and foreign body sensation. In some instances it can extend onto the cornea. It has no known malignant potential. It is characterized histopathologically by acanthosis, dyskeratosis, and prominent rete pegs.
HBID is a benign condition that does not usually require aggressive treatment. Smaller, less symptomatic lesions can be treated with ocular lubricants and judicious use of topical corticosteroids. Larger symptomatic lesions can be managed by local resection with mucous membrane grafting if necessary.
Epithelial Inclusion Cyst
Conjunctival cysts can occur spontaneously or following inflammation, surgery, or nonsurgical trauma. Histopathologically, they are lined by conjunctival epithelium and are filled with clear fluid that often contains desquamated cellular debris (Fig. 10-7). Such cysts can be simply observed or they can be excised completely with primary closure of the conjunctiva.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree