Tube Shunts for Refractory Glaucomas
Anna K. Junk
L. Jay Katz
When standard filtering surgery, such as a trabeculectomy with adjunctive antimetabolite therapy (5-fluorouracil or mitomycin C), has failed or is unlikely to succeed (as with active neovascular glaucoma), more viable methods for aqueous shunting should be considered. Implantation of alloplastic tubes leading to equatorial reservoirs has led to remarkably good results in eyes with refractory glaucoma. The success rate for tube shunts controlling various types of refractive glaucomas is more than 60% to 85% in most series, but it is a complex procedure, with numerous potential intraoperative and postoperative complications. With increasing use, modifications in technique have lessened but not eliminated many of the problems encountered with tube shunts. An alternative therapy is one of the ciliodestructive procedures, such as cyclocryotherapy or diode laser cyclophotocoagulation. The relatively high rates of visual loss, cataract progression, and phthisis make the ciliodestructive procedures less desirable for eyes with useful vision1,2,3.
Historical Perspective
The evolution of aqueous-diversion devices began with the use of translimbal setons, such as a silk thread. These setons provided a passageway from the anterior chamber to the limbal subconjunctival space (Table 6-17.1). In 1906, Rollet4 used a horse hair and, in 1912, Zorab5 used a silk thread to lower intraocular pressure by allowing aqueous flow around the translimbal wick seton and into the limbal subconjunctival space. Long-term success with setons remained elusive, however, despite the exhaustive search for a material that could help the drainage site to remain patent. Materials, such as gold,6,7 platinum,8 tantalum,9 glass rods,10 and polymethylmethacrylate (PMMA)11 were tried as setons for the management of glaucoma. Problems, such as fibrosis within the channel, seton migration, and conjunctival erosion, could not be overcome.
TABLE 6-17.1. Evolution of aqueous shunting | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
The second phase was the development of shunt devices, tubes made of silicone12,13,14,15 or polytetrafluoroethylene (Teflon),16,17 that were used as translimbal conduits from the anterior chamber to the limbal subconjunctival space. The development of synthetic plastic materials thought to be biologically inert promised less tissue reaction and theoretically less subconjunctival fibrosis.18 Despite the placement of a scleral flap over the tube to cover these implants, tube erosion through the conjunctiva still occurred. Fibrosis over the distal tube opening,19,20 with elevation of the intraocular pressure limited their success. The initial Krupin valve shunt was placed subconjunctivally at the limbus, with the distal tube end only several millimeters from the limbus.21 Although there was brief enthusiasm for the shunt, disappointing results were later reported,22 and persistent problems of conjunctival erosion and distal opening scarring became evident.23 The Molteno tube prototype, unveiled in 1969, had the distal tube end attached to an acrylic plate reservoir that originally was placed near the limbus. The plate elevated the limbal conjunctiva considerably, which led to many complications, including conjunctival perforation and corneal dellen.24
In the third and current phase, work is based on the principle of connecting the intracameral tube to a posteriorly placed reservoir at or behind the globe equator. Molteno et al.25 were the first to capitalize on this concept. Instead of using a plate as a reservoir, Schocket et al.26 modified a no. 20 encircling silicone band to house the polymeric silicone (Silastic) tube shunt that was placed under the recti muscles. The Baerveldt tube shunt provides a large, soft, pliable surface area for easy placement in a single quadrant.27,28 The Joseph,29 OptiMed,30 (Joseph, OptiMed, and Krupin devices are no longer commercially available) Krupin,31 and Ahmed32 shunts have a design similar to the Molteno shunt, with the addition of a unidirectional valve to close the tube if the intraocular pressure drops too much (e.g., <2 mm Hg). The White pump shunt (no longer commercially available) also had a unidirectional valve in addition to a pump mechanism that was activated by blinking or touching to help aqueous egress.33,34 There was no reservoir, the distal tube end was posteriorly placed in the equatorial subconjunctival region.
Mechanism of Intraocular Pressure Reduction and Histopathologic Features
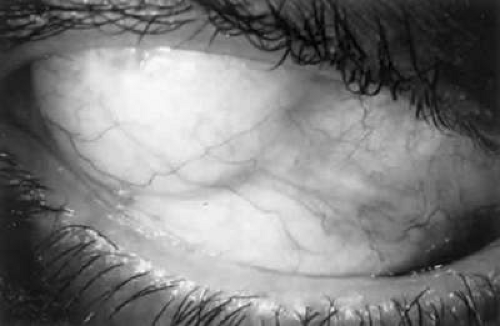
The filtration site is placed posteriorly near the equator in the latest generation of shunts. The morphologic features of these blebs are different from those of functional blebs at the limbus, as seen with a trabeculectomy. They are typically thick-walled, with prominent conjunctival vascularity, and are elevated over the reservoir (Fig. 6-17.1). Although the silicone tube and PMMA plates of the Molteno shunt are biologically inert and do not produce tissue necrosis, a thick fibrous capsule envelops the reservoir and tube (Fig. 6-17.2).35,36
These histopathologic findings are similar to those reported for the Schocket tube,37 Baerveldt tube,36,38 and White pump shunt.39 The shunt apparatus is not degraded; it remains intact, with little or no adjacent inflammatory response and without fibrous adhesion to the implant. In contrast, a case report of an original Krupin valve composed of Supramid describes significant biodegradation, with loss of tensile strength, attenuation, and fracture.23
The fibrous capsule that may originate from the episclera39 is relatively acellular and composed of scattered fibroblasts, collagen, and glycosaminoglycans.35,37 Aqueous is thought to pass through the fibrous capsule and into the orbital circulation through capillaries and lymphatics in addition to conjunctival circulation.35,37 Schocket used horseradish-peroxidase as a tracer and showed that aqueous could pass through the orbital vessels.37 In that study, the reservoirs preserved an area for bleb formation that had a relatively large surface area from which aqueous diffused. Orbital outflow, mechanical maintenance of a bleb zone, and a large surface area distinguish posterior tube shunts from limbal filtering operations and help to explain their success in the treatment of difficult cases of glaucoma.
Shunt Devices
Currently, the most popular shunts include the Baerveldt and Ahmed devices. The Baerveldt shunt (Fig. 6-17.3) was developed to provide easy placement in a single quadrant and also to provide a large surface area. It has a soft, pliable reservoir of silicone and a large surface area (200, 350, or 425 mm2) for aqueous pooling.27,28 Placement of the reservoir underneath the rectus muscle insertion, however, promotes fibrous encapsulation of the reservoir to the muscles, which leads to disturbing diplopia.40,41 The design has been modified, with fenestrations in the reservoir that may allow tissue tacks to limit bleb elevation and may eliminate rectus muscle imbalance and diplopia.
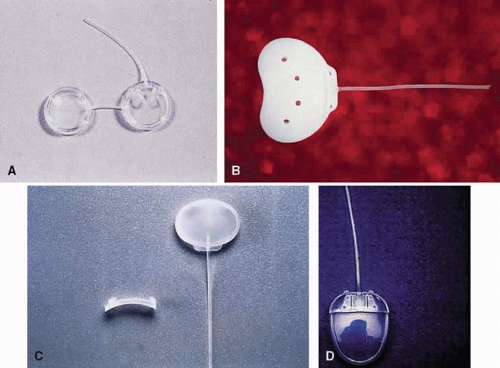 Figure 6-17.3: Shunt devices. A: Double-plate Molteno shunt. B: 350-mm Baerveldt shunt. C: Krupin valve shunt. D: Ahmed valve shunt. |
The Ahmed valve shunt (Fig. 6-17.3) is also a single reservoir system.42,43 The valve is composed of two thin folded-over silicone membrane leaflets, using the Bernoulli principle. The inner outlet is wider than the outlet, which allows patency even at low pressure differentials. The tapering shape of the silicone chamber is claimed to improve flow regulation by Venturi action. Both, a rigid polypropylene plate or pliable silicone plate are marketed. The Ahmed valve is also available as a double plate in both materials offering a greater surface area (364 mm2). For pars plana insertion a “pars plana clip” was developed to avoid tube occlusion from kinking.
Clinical studies suggest that the Molteno implant is more likely to result in intraocular pressures in the lower teens than the Ahmed Glaucoma Valve. The Ahmed implant is less likely to create problems leading to reoperation or visual acuity loss than the Molteno or Krupin implants.44 Long-term results of single- or double-plate Molteno implant and Ahmed Glaucoma Valve are similar.45,46
The Molteno shunt (Fig. 6-17.3) has been investigated clinically by numerous surgeons.45,47,48,49,50,51,52,53,54,55,56,57,58,59,60 The double-plate Molteno shunt is recommended in most cases because the surface area is twice as large as that of the single-plate version (Table 6-17.2). A clinical study comparing single-plate versus double-plate Molteno shunts suggests that surface area is a crucial factor for surgical success.61 Although the Schocket tube has a slightly larger surface area than a double-plate Molteno shunt, two comparative randomized trials showed that a double-plate Molteno tube shunt fared as well.56,60
TABLE 6-17.2. Shunt dimensions and material | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Placement of the Schocket tube1,25,62 is a more lengthy and cumbersome procedure, and its only advantages are lower cost and greater availability of materials. The Schocket implant is not commercially available. The tube is assembled by the surgeon from materials commonly used for retinal buckling procedures (no. 20 band) and a silicone tube used for nasolacrimal intubation. It may be the preferred implant in eyes with a scleral buckle already present because of prior retinal detachment repair.
The Krupin valve is currently not available (Fig. 6-17.3). The patents and rights were purchased by EagleVision, an ophthalmic device company. Two modified versions of the Krupin shunt, a valved (flow-restrictive mechanism) and a non-valved implant were approved by the US Food and Drug Administration (FDA) for the treatment of refractory glaucoma. Both, the smaller, valved version (209 mm2) and the larger, non-valved implant (365 mm2) are currently being tested for safety and efficacy in pilot studies. Historically, the Krupin shunt is a two-piece device with a pressure-regulated slit valve that was modified from the original translimbal tube shunt21 to a tube to a 180 degree band (no. 220 Silastic implant reservoir),63 and a tube to an oval reservoir silicone plate.31 The opening pressure of the valve was designed to be 11 to 14 mm Hg and the closing pressure was 2 mm Hg. The opening and closing pressures could vary significantly,30 however, and this variability may not be discovered until the procedure is completed.
The Joseph valve shunt was a one-piece design that is similar to the two-piece Krupin shunt.29,64,65 Lavin et al.66 suggest that the one-piece design minimizes the possibility of fibrous ingrowth at the tube–plate junction.
The OptiMed implant (no longer commercially available) was composed of a silicone tube connected to a PMMA plate. The flow-restricting element contained 180 microtubules, each 60 μm in diameter, providing a pressure gradient governed by Poiseuille’s law.
Flow Characteristics of Shunts
Fluid–flow dynamics through the various tube shunts has been examined experimentally.30,52,69,70,71 As expected, there is no open-air closing pressure for the non-valved shunts. It is unclear whether the valve shunts perform in vivo according to their specifications. The opening and closing pressures for the respected valve shunts are 10 and 8 mm Hg for the Ahmed and 11 and 9 mm Hg for the Krupin valve. The Ahmed was the only valve shunt that varied resistance according to flow rate.71,72
Indications for Tube Shunt Implantation
Patients who have a poor surgical prognosis with a guarded filtration procedure are candidates for either a tube shunt73,74 or a ciliodestructive procedure.3 The latter procedure usually is reserved for eyes that have poor vision or patients whose general health or preference precludes tube placement. In some cases, the combination of tube shunt and cyclophotocoagulation is necessary.75,76
Candidates for tube shunts include patients with the following conditions:
Active neovascular glaucoma
Active uveitic glaucoma
Severe limbal conjunctival scarring
Any failed filtering procedure with antimetabolite use
Epithelial downgrowth
Associated vitreoretinal disease when used in combination with pars plana vitrectomy or penetrating keratoplasty, especially in aphakic or pseudophakic eyes
Refractory infantile glaucoma
Need for avoidance of limbal filtration blebs (e.g., contact lens wearer)
Primary surgical management of chronic open angle and chronic angle closure glaucomas
When the surgeon cannot wait for panretinal argon laser photocoagulation to promote regression of florid neovascularization, a standard filtering procedure—even with antimetabolites—has little chance of providing long-term benefit. Ciliodestructive procedures, although equally effective lowering the intraocular pressure,2 are not ideal if the goal is visual preservation rather than palliative relief of ocular pain or control of pressure.54 Tube shunts have comparatively high success rates, about 75% in these difficult cases.2 These procedures can be combined with vitrectomy and posterior segment laser photocoagulation.42,46,74 The tube can also then be placed in the vitreal cavity through the pars plana rather than the anterior chamber.77,78,79,80,81,82,83
With active uveitis, there is persistent stimulation for subconjunctival fibrosis at the limbus, which often leads to failure of any limbal filtering procedures. Shunting the aqueous to posteriorly placed reservoirs is more likely to provide long-term control of the secondary inflammatory glaucoma.84,85
Epithelial downgrowth usually rapidly closes limbal sclerostomies with epithelial sheets, but does not appear to grow over the tubes. This feature allows control of intraocular pressure,86 and restoration of useful vision is possible if a penetrating keratoplasty is performed later.87
If limbal filtration surgery has failed or extensive limbal subconjunctival scarring is seen (e.g., after cataract or retinal detachment surgery), tube shunts are especially appropriate.88 The old filtration sites should be avoided whenever possible. After the conjunctiva is dissected from the old filtration site, the drainage area often becomes functional, and overfiltration can occur unless the area is sutured closed.
Penetrating keratoplasty with aphakia or pseudophakia predisposes eyes to peripheral anterior synechiae. This tendency makes a limbal-filtering procedure challenging to perform and likely to fail. Placement of a tube shunt controls intraocular pressure in most of these cases, but it may promote graft decompensation.89,90,91,92,93,94,95 Often it is preferable to place the tube into the vitreous cavity through the pars plana after a thorough vitrectomy has been performed.77
Congenital and infantile glaucoma can be frustrating to control after goniotomies, trabeculotomies,96 and filtering procedures fail.97 Previously, ciliodestructive procedures were used in such cases in an attempt to control the intraocular pressure. It is clear that tube shunts are preferable to ciliodestructive procedures, and they have been beneficial for recalcitrant cases of glaucoma.52,54,98,99,100 Despite short-term success (>80% for 1 year), long-term success (<57% to 75% over 4 years) is still a concern in the pediatric population.55,101 Delayed tube retraction because of continued growth of the eye may require repositioning of the implant,102 or repositioning or extension of the tube.
In young patients who have a history of bleb infections, preference may be to avoid another limbal bleb and opt for a tube shunt with a more posterior, thicker walled bleb. No reports are found of bleb infections over tube reservoirs unless there has been an extrusion and exposure.103 If contact lenses are worn, they pose a serious increased risk for bleb infection and endophthalmitis with limbal blebs. It is preferable either to consider cataract or refractive surgery to eliminate contact lenses before a trabeculectomy or to go straight to a tube shunt.
Glaucoma drainage implants may gain popularity as the primary surgical treatment for open angle glaucoma or chronic angle closure, conditions commonly treated with filtering procedures. Whereas trabeculectomy may result in a lower intraocular pressure in the first year of follow up, the surgical success rate is comparable at 41- to 52-month follow-up for trabeculectomy (68.1%) versus the Ahmed valve implant (69.8%).98,104.
Choosing a Glaucoma Drainage Device
Conventionally, the intraocular pressure-lowering effect of glaucoma drainage devices was found to correlate with the size of the implant. Smaller implants (e.g., the pediatric Ahmed valve or the single plate Molteno implant), in general, result in less pressure reduction than larger devices (i.e., double-plate Molteno61 or the Baerveldt-350 tube shunt44). The Baerveldt-500, however, does not appear to offer much benefit over the easier to handle Baerveldt-350 plate in terms of pressure-lowering effect.105,106,107 Recent studies demonstrate equal surgical success rates (65.6% each) for Baerveldt-350 and Ahmed valve implants at 1-year follow up108 and a qualified success of 83.4% or higher109 for both devices. Another comparative study indicates greater surgical success for the Ahmed valve (82.9%) compared with the Baerveldt implant (72.9%) 1 year after surgery. The authors also noted, however, a higher prevalence (60.4% vs. 27.1%) and earlier onset of bleb encapsulation with the Ahmed device.110
Surgical Technique
Anesthesia
The operation can be lengthy, but virtually all adult patients can be treated safely and comfortably with local anesthesia. The local anesthesia can be supplemented by subconjunctival infiltration with local anesthetic, which in turn is helpful in separating scarred subconjunctival tissue from the sclera. If the intraocular pressure is high (>35 mm Hg), intravenous mannitol should be given (1 to 2 g/kg) over 30 to 45 minutes whenever medically acceptable. Once the intraocular pressure is lowered, the magnitude of the reduction of intraocular pressure with surgery is not as dramatic and the risk of suprachoroidal hemorrhage or massive choroidal effusion may be reduced.
Exposure of the Scleral Bed
The conjunctival flap is raised by sharp dissection from the insertion at the limbus. The fornix-based flap is elevated, which allows direct visualization for further subconjunctival dissection to bare sclera. Alternatively, a conjunctival incision is made 4 to 6 mm from the limbus to raise a limbal-based conjunctival flap. This is helpful when there is a previous limbal filtration site, which is best avoided. If the old site is inadvertently reopened, the globe becomes hypotonus and it will be difficult to place the shunt reservoir.
Use of antimetabolites such as 5-fluorouracil or mitomycin C with tube shunt reservoirs remains controversial. Although it was demonstrated in an animal study that the blebs overlying the reservoir are thinner and contain less connective tissue,111 the clinical use of antimetabolites has not been shown to have any benefit.101,112,113
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree