Trigeminal Nerve
Leo H. Wang
Eric E. Kraus
Craig H. Smith
The trigeminal nerve (cranial nerve V [CN V]) is the main sensory nerve of the face responsible for tactile, proprioceptive, and nociceptive afferent signals from a large portion of the face, eyes, nose, and mouth. However, it is also a mixed nerve that carries motor efferent fibers to the muscles of mastication.
ANATOMY
Trigeminal Ganglion and Brainstem Nuclei
The trigeminal ganglion (Gasserian or semilunar ganglion) is the sensory ganglion that contains the nerve cell bodies of the pseudounipolar neurons of the sensory root of the trigeminal nerve. The trigeminal ganglion is located in Meckel’s cave near the apex of the petrous part of the temporal bone just behind the internal carotid artery and the posterior portion of the cavernous sinus.1 Proximally, the sensory root enters the pons and terminates in the trigeminal nuclei (which receive input from all sensory nerves innervating the face). From rostral to caudal, the trigeminal nuclei are divided into three parts: mesencephalic nucleus (in the midbrain and pons), principal sensory nucleus (in the pons), and spinal nucleus (spanning the pons to the upper cervical cord) (Fig. 36.1).
The mesencephalic nucleus receives muscle spindle information conveying proprioceptive afferents from the muscles of mastication. These are the only primary sensory afferents that have their cell body in the CNS, rather than outside in a sensory ganglion. A collateral branch synapses directly on the motor nucleus of the trigeminal nerve to activate jaw closure through the monosynaptic jaw jerk reflex. However, it also sends projections to the principal sensory nucleus and the spinal nucleus.
The principal sensory nucleus is thought to mediate large fiber modalities such as fine touch or other proprioceptive afferents. The spinal nucleus is thought to mediate small fiber modalities such as pain and temperature. The long spinal nucleus is organized in a somatopic fashion, with the most rostral region receiving innervation from the most midline regions (the mouth and nose), whereas the most caudal region receiving innervation from the most dorsal regions (outer ears). Therein lays the onionskin somatotopic organization of the spinal nucleus.2 Furthermore, the spinal nucleus receives proprioceptive information from mesencephalic nucleus projections.
Most of the fibers from the principal sensory and spinal nuclei cross to the contralateral side and ascend in the anterior trigeminothalamic tract to the ventral posteromedial nucleus of the thalamus. The anterior trigeminothalamic tract joins lateral to the medial lemniscus, which carries the analogous sensory fibers from the contralateral body to the ventral posterolateral nucleus of the thalamus. From the thalamus, the secondary neurons project to the primary somatosensory cortex located in the postcentral gyrus of the parietal lobe. There is much overlap of somatotopy of the face in the thalamus, while the inverted facial somatotopy is relatively more preserved in the primary somatosensory cortex (S1 region). From the superior-to-inferior axis in the coronal plane of the postcentral gyrus, the representation of the thumb is the more superior, followed by the most caudal facial region V1, than the more rostral facial regions. Muscle spindle information from the mesencephalic nucleus projects to the cerebellum.
The motor nucleus is ventral and medial to the principal sensory nucleus in the midpons. The motor root exits midpons and is anterior and medial to the larger sensory root. In Meckel’s cave, the motor root travels below the trigeminal ganglion and exits the skull through the foramen ovale along with the mandibular division. The motor nucleus receives input from sensory branches of the trigeminal and other sensory cranial nerves. It also receives input from bilateral motor cortex for voluntary control of chewing.
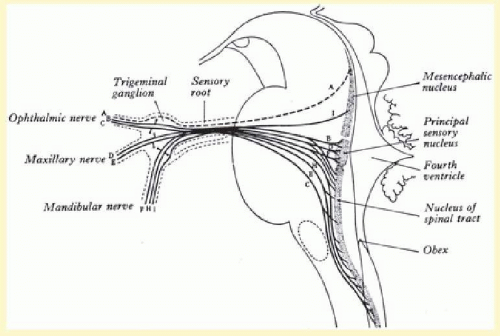 FIG. 36.1 Trigeminal nuclei and ganglion. (From Warwick R, Williams PL. Gray’s Anatomy. 35th ed. Edinburgh, UK: Churchill Livingstone; 1973:1001.) |
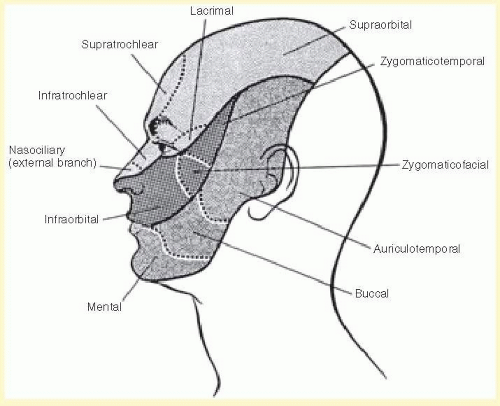
The three sensory divisions of the trigeminal nerve are the ophthalmic (V1), maxillary (V2), and mandibular (V3) (see Figs. 36.2 and 36.3). Although autonomic fibers do not originate from the trigeminal nerve, parasympathetic fibers from CN III (oculomotor), VII (facial), and IX (glossopharyngeal) and sympathetic fibers from superior cervical ganglia travel with the widely distributed trigeminal nerve to reach their destination.
Ophthalmic Division
The ophthalmic division/nerve exits the skull through the superior orbital fissure to enter the orbit. This division occupies the lateral wall of the cavernous sinus inferior to cranial nerves III and IV and lateral to cranial nerve VI. The ophthalmic nerve divides into the (1) frontal, (2) nasociliary, and (3) lacrimal nerves just before entering the superior orbital fissure (see Figs. 36.2 and 36.4). A tentorial branch returns to supply the dura of the cavernous sinus, sphenoid wing, anterior fossa, petrous ridge, Meckel’s cave, tentorium cerebelli, posterior falx cerebri, and dural venous sinuses.
1. The frontal nerve is the largest branch (and therefore considered a continuation) of the ophthalmic nerve. The nerve divides into the supraorbital and supratrochlear nerves, which innervate the conjunctiva, medial upper eyelid, forehead, scalp, frontal sinus, and bridge of the nose.
2. The nasociliary nerve gives off many branches, including the anterior ethmoidal, posterior ethmoidal, and infratrochlear nerves, innervating the sphenoidal sinus, ethmoidal air cells, the upper eyelid, the caruncle, the canaliculus sac, the lacrimal sac, the mucosa of the nasal septum, the mucosa of the inferior and middle turbinates, and the tip and side of the nose.
The nasociliary nerve contributes sensory fibers that innervate the iris and cornea to the 2 to 3 long ciliary nerves and 6 to 10 short ciliary nerves. In addition to the sensory fibers, the long ciliary nerves also carry postganglionic sympathetic nerves to the pupil dilators. The short ciliary nerves contain sensory, parasympathetic, and sympathetic nerve fibers. The sensory nerve fibers destined for the short ciliary nerves, split off from the nasociliary nerve to form the sensory root of the ciliary ganglion, and traverse (but do not synapse in) the ciliary ganglion. The parasympathetic fibers are postganglionic parasympathetic fibers that originate from the ciliary ganglion and innervate the pupil constrictors and ciliary muscle. In the ciliary ganglion, they form synapses with preganglionic parasympathetic fibers originating from the Edinger-Westphal nucleus that travel with the oculomotor nerve (specifically, the nerve fibers destined to innervate the inferior oblique muscle). The sympathetic fibers are postganglionic sympathetic fibers originating from the superior sympathetic ganglion in the neck and travel the internal carotid plexus to reach the pupil dilators and ciliary muscle. The cornea is supplied by 60 to 80 nerves that course radially from the limbus and branch into over 1,000 small axons that terminate as free nerve endings in the stroma and epithelium.
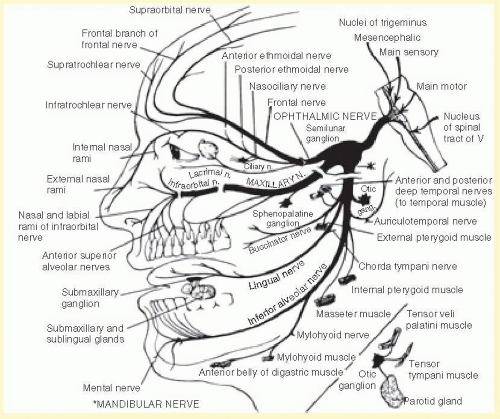 FIG. 36.2 Branches of the trigeminal nerve. (From Chusid JG. Correlative Neuroanatomy and Functional Neurology. Los Altos, CA: Lange Medical Publishers; 1976:92.) |
3. The lacrimal nerve innervates the conjunctiva and the skin of the lateral upper eyelid. The nerve is joined by postganglionic, parasympathetic facial nerve fibers from the pterygopalatine ganglion to innervate the lacrimal gland.
Maxillary Division
The maxillary division/nerve runs inferiorly in the cavernous sinus and passes through the foramen rotundum to the pterygopalatine fossa. Before the foramen rotundum, it gives off a meningeal branch that innervates the meninges of the anterior and middle cranial fossa. In the pterygopalatine fossa, it branches into the zygomatic, superior alveolar, palatine, pharyngeal nerves. The maxillary nerve then continues as the infraorbital nerve as it enters the orbit through the infraorbital fissure, travels in the infraorbital canal along the orbital floor, and exits on the face in the infraorbital foramen. One of the branches of the infraorbital nerves, the inferior palpebral branch, innervates the lower eyelid.
The maxillary division supplies sensation to the nasopharynx, maxillary sinus, roof of the mouth, soft palate, upper teeth, and an area of the face that extends from the upper lip to the lower eyelid and from the side of the nose to the cheek. In addition, the maxillary division receives lacrimal postganglionic parasympathetic fibers from the pterygopalatine ganglion, which it delivers to the lacrimal nerve of the ophthalmic division.
Mandibular Division
The mandibular nerve does not reach the cavernous sinus like the other two divisions; rather, it passes out of Meckel’s cave through the foramen ovale. Immediately, it gives off a recurrent meningeal branch that joins the middle meningeal artery and re-enters the skull through the foramen spinosum to innervate the meninges of the posterior cranial fossa. Its sensory branches, the auriculotemporal, buccal, lingual, and inferior alveolar nerves, provide sensation to the lateral scalp, posterior cheek and temporal areas, temporomandibular joint, anterior pinna, upper and outer walls of the external auditory canal, anterior half of the tympanum, lower lip and gums, chin, anterior two-thirds of the tongue, floor of the mouth, lower teeth, and lower half of the buccal surface.
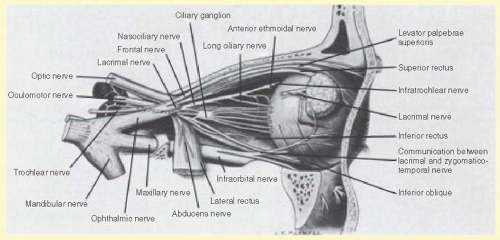 FIG. 36.4 Nerve supply to the right globe. (From Warwick R, Williams PL. Gray’s Anatomy. 35th ed. Edinburgh, UK: Churchill Livingstone; 1973:1003.) |
Motor fibers innervate four masticatory muscles, lateral pterygoid, medial pterygoid, temporalis, and masseter, and four other muscles, tensor tympani, tensor veli palati, mylohyoid, and anterior belly of the digastric.
CN VII and IX contribute nerve fibers that join branches of the mandibular nerve. Preganglionic parasympathetic nerve fibers from CN VII travel in the chorda tympani branch of CN VII and join the lingual nerve to travel to the submandibular ganglion where they synapse with nerve fibers that innervate the submandibular and sublingual gland. Special sensory (taste) nerve fibers travel the same route and transmit taste sensation from the anterior two-thirds of the tongue. Preganglionic parasympathetic nerve fibers from CN IX join the mandibular nerve and synapse in the otic ganglion. From the ganglion, the postganglionic parasympathetic nerve fibers join the auriculotemporal nerve to innervate the parotid gland.
PHYSIOLOGY
Ocular Sensation
Because of its high concentration of free nerve endings, the cornea is one of the most pain-sensitive structures in the body. Sensitivity is greatest in the center of the cornea and decreases toward the limbus. Pain receptors are also found in extraocular muscle, conjunctiva, uvea, sclera, and optic nerve sheath. In contrast, the retina, optic nerve, and lens are devoid of pain receptors.
Trigeminal Reflexes
Several trigeminal reflexes are of clinical importance. Pressure, or manipulation, of the ocular structures can result in the oculocardiac reflex, which is responsible for the bradycardia and other arrhythmias seen in ocular surgery, in particular strabismus surgery.3 The oculocardiac reflex, however, is only a portion of the broader trigeminocardiac reflex in which the afferent limb of the reflex is the trigeminal nerve, and the efferent limb is the vagus nerve.4,5 Vagal stimulation of the heart causes slowing and, rarely, asystole.
Other reflexes involving the trigeminal nerve include the jaw jerk, snout reflex, and sneeze reflex. Corneal stimulation can produce the corneal, corneolacrimal (reflex tearing), corneomandibular, and corneo-oculogyric reflexes.6 Nausea and vomiting may occur during an acute attack of glaucoma, with trauma, or with certain intraorbital inflammatory processes.
CLINICAL ASSESSMENT
Sensory Functions
Corneal sensitivity is tested by the light touch of a cotton-tipped applicator drawn to a fine point. If local cornea disease is present, each quadrant should be assessed separately, because it may be only a portion of the cornea that is denervated; otherwise, it is sufficient to test just the inferior cornea. The response to corneal stimulation is assessed both subjectively through the patient’s report and objectively by observation of the corneal reflex.
Cutaneous sensation is assessed as it is elsewhere in the body by testing light touch with cotton and pain with pinprick. The dermatomes of each of the three divisions of the trigeminal nerve should be tested separately (see Fig. 36.3). Temperature, vibration, stereognosis, and twopoint discrimination can also be assessed, but have little clinical relevance.
Motor Functions
The motor root of the trigeminal nerve is assessed by palpating the temporal and masseter muscles as the patient clenches his or her jaw and noting pterygoid strength during jaw opening (medial pterygoids) and lateral movement against resistance (lateral pterygoids). Weakness of the pterygoids will produce a deviation of the jaw to the ipsilateral side when the patient opens their mouth. Observation may reveal a more prominent zygomatic arch on the symptomatic side due to muscle wasting. Although more difficult to see, flattening or depression of one side of the palate and deviation of the uvula to the other side at rest may indicate tensor veli palatini dysfunction. Elevation of the palate voluntarily or to gag, however, is mostly a CN X function.
A unilateral supranuclear lesion rarely causes any weakness of the masticatory muscles because of bilateral cortical innervation of the trigeminal motor nuclei. An increased jaw jerk, evaluated by tapping over a finger placed on the middle of the chin with a reflex hammer while the patient relaxes his or her jaw, can be seen in supranuclear lesions. Normally, the jaw jerk is mild or absent in the nondisease state.
Observation for involuntary movements of the jaw is important. Rhythmic jaw tremor can be seen in both essential tremor and Parkinson disease. Mouth retraction and jaw opening or closing may be seen in Meige syndrome (see elsewhere in these volumes). Fatigable weakness of the jaw, or a history that the patient has to use his or her hand to close the jaw, is a feature of myasthenia gravis. A history of clonic jaw activity or chewing movements may indicate seizure activity. Jaw claudication (jaw/ear pain while chewing) is a frequent complaint in giant cell arteritis. Spasm of the masticatory muscles, with inability to open the mouth, is called trismus and can be seen in hemimasticatory spasm, tetanus, encephalitis, strychnine poisoning, and hysteria.
ELECTROPHYSIOLOGIC TESTING
Electrophysiologic testing of the trigeminal nerve can be accomplished in several ways. The blink reflex is the workhorse when evaluating the sensory system, and both the masseter reflex and direct electromyography (EMG) are used when evaluating the motor system.
The afferent component of the blink reflex is mediated through the trigeminal nerve, and the efferent component is mediated through the facial nerve. The stimulation site is over the supraorbital nerve, and the recording site is over the orbicularis oculi muscle. Connections are made between the trigeminal and facial nuclei in the brainstem at pontine and medullary levels. Identification of an abnormality in the reflex is based on the specific pattern of direct facial nerve stimulation and the early monosynaptic R1 and late oligosynaptic R2 latencies.7 The main application is in cases of facial pain in which clinical evaluation alone does not satisfactorily document trigeminal dysfunction. Unfortunately, demonstration of a lesion with this method may not be very high. The reported sensitivity of the blink reflex is 50% to 85% for tumor, infection, trauma, some polyneuropathies, multiple sclerosis (MS), and stroke, and it is usually normal in trigeminal neuralgia.7
Other tests of sensory function are the corneal reflex, in which direct stimulation of the cornea elicits bilateral R2 responses, but not R1 responses,8 and somatosensory evoked potentials,9 which are less useful.
The masseter or jaw reflex is a monosynaptic reflex involving proprioceptive fibers that travel by way of the mandibular division of the trigeminal nerve to the brainstem. Before reaching the mesencephalic nucleus, which lies in the midbrain, a branch synapses on the motor nucleus of the trigeminal nerve in the pons. Impulses are sent back to the masseter muscle by way of the motor nerve, causing the jaw to close.7 Use of both the blink and masseter reflexes may increase the clinical ability of demonstrating a trigeminal nerve lesion.10 EMG of the masseter muscle may document denervation.
TRIGEMINAL NERVE DYSFUNCTION
Trigeminal nerve disorders may present with loss of function (e.g., anesthesia and paresis of mastication) or abnormal sensation (e.g., pain and paresthesias). Most pathologic processes involving the nerve can produce both. In general, peripheral nerve lesions produce anesthesia on the face and inside the mouth, whereas central lesions only involve the face. Lesions peripheral to the trigeminal ganglion usually involve only one or two trigeminal divisions, whereas proximal lesions tend to affect the whole half of the face. Lesions of the ganglion itself may follow either pattern. Combined involvement of CN III, IV, VI, or a Horner syndrome may localize the lesion to the cavernous sinus.
Clues to a brainstem lesion should be sought when one is faced with trigeminal nerve dysfunction. As there is some dissociation of light touch with the main sensory nucleus, and pain and thermal sensation with the nucleus of the spinal tract of the trigeminal nerve, so too might this dissociation be appreciated clinically. Also, the compact nature of the pons and medulla makes it likely that other cranial nerves, as well as the ascending and descending tracts connecting the spinal cord with the cerebrum, will be affected.6
Supranuclear lesions most often have a wider distribution of dysfunction than just the face (e.g., ear, neck). Sensory loss can be seen in thalamic, internal capsule, and cortical disease. Clinical weakness usually requires bilateral disease such as would be seen with bilateral strokes or amyotrophic lateral sclerosis.
The following sections describe in more detail the diseases that can affect the trigeminal nerve, starting at the cornea and moving through the peripheral branches, cavernous sinus, trigeminal ganglion, middle cranial fossa, trigeminal root, brainstem and thalamus, basal ganglia, and sensory cortex. Finally, we briefly discuss a few common systemic diseases that may affect the trigeminal nerve. The diseases that present almost entirely with facial pain will be discussed separately (see section on Facial Pain). However, it should be remembered that there is often a great deal of overlap between the conditions that present with anesthesia and pain. Figure 36.5 depicts three diseases as a Venn diagram. Trigeminal neuralgia is the quintessential disease presenting with second long shocks and no sensory loss. Constant symptoms present as either atypical (chronic) facial pain with pain only or trigeminal sensory neuropathy with anesthesia only. Unfortunately, many people present with a mixture of several features and it can be difficult to know how to proceed with a clinical evaluation or provide treatment.
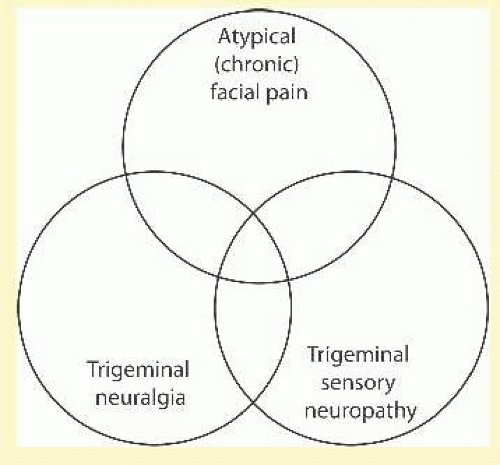 FIG. 36.5 Several diseases fall on a continuum between three features: timing (shock like to constant), anesthesia, and pain. |
Loss of Corneal Sensation
Corneal disease can be secondary to a lesion of the trigeminal nerve impairing cutaneous sensation in the ophthalmic division. Corneal nerve fibers and the corneal epithelium exert mutualistic trophic influences on each other with the trigeminal nerves secreting neurotransmitters and neuropeptides (such as substance P and calcitonin gene-related peptide) that maintain tissue integrity, physiologic renewal, and wound healing.
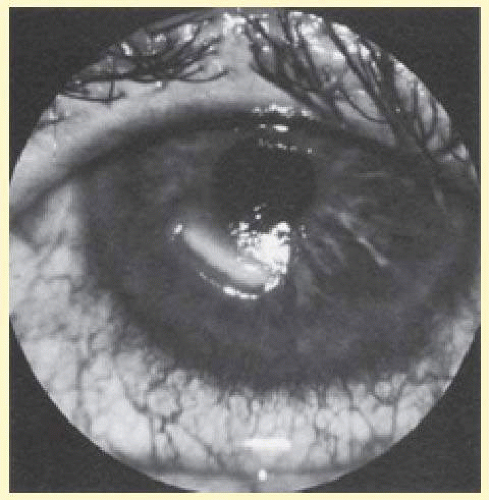
Corneal nerve fiber loss may occur iatrogenically from surgery (for trigeminal neuralgia or acoustic neuroma), medication effect (topical), diabetes, or infections (especially herpes simplex virus or herpes zoster).11 Photorefractive keratectomy and laser-assisted in situ keratomileusis (LASIK) are the newest iatrogenic causes of corneal nerve fiber loss.12 Loss of corneal sensation almost always accompanies trigeminal nerve lesions that impair cutaneous sensation in the ophthalmic division.13 Neurotrophic keratitis is the inflammation or loss of corneal epithelium associated with the loss of corneal nerve fibers (Fig. 36.6). The loss of trophic support from the corneal nerve fibers is thought to be a major factor, and much research is directed at developing treatment with neuropeptides to prevent this disease. However, other factors are thought to contribute, including desiccation of corneal surface due to diminished lacrimation, environmental exposure, inappropriate contact lens use, impaired corneal sensitivity, and therefore decreased protective blink reflexes and abnormal epithelial cell metabolism.
Treatment includes topical lubrication, punctal occlusion, environmental protective measures, and discontinuing topical ocular therapies that can decrease corneal sensitivity (e.g., timolol, betaxolol, sulfacetamide, diclofenac, ketorolac). Surgery may be needed to (1) close the eyelid (which can be by lateral tarsorrhaphy or botulinum A toxin injection into the levator muscle), (2) repair of corneal perforation or descemetocele secondary to severe stromal ulcerations, and (3) treatment of persistent epithelial defect. The second goal can be accomplished by amniotic membrane grafts.14,15,16
Peripheral Branches
Peripheral branches of the trigeminal nerve can be affected by numerous disease processes, including traumatic, vascular, inflammatory, demyelinating, infectious, or neoplastic processes.17 Facial trauma may impair branches of any of the three divisions, but most often damaged are the infraorbital, supratrochlear, and supraorbital nerves (see Fig. 36.3).18 Dental procedures are a common cause of trigeminal neuropathy, because the dental nerves course in the maxilla and mandible at the same depth as the roots of the teeth.19,20 Specifically, removal of the lower third molar can cause damage to the inferior alveolar nerve (innervating the lower teeth, lower lip, and chin) and the lingual nerve (tongue) at a rate of approximately 4%.21 The LASIK procedure has been reported to cause infraorbital nerve damage.22
The usual presentation of trigeminal neuropathy is either anesthesia or paresthesias, but after partial regeneration of the nerve, pain may also occur.23 Even after the inciting cause is removed, return of function is not guaranteed. With trauma, as few as 50% will recover by 12 weeks, with a minority of the rest recovering by 2 years.19
Trigeminal pain and numbness are rarely the first sign of tumors of the nasopharynx, paranasal sinuses, oral cavity, and orbit. Nasopharyngeal carcinoma has a tendency to spread perineurally from the cavernous sinus,24 as can adenoid cystic carcinomas and dermatologic malignancies.25 Sensory loss in orbital disease of all causes is relatively uncommon, occurring in only approximately 3% of the patients.26 Patients rarely complained of subjective numbness, approximately half complained of pain. This is also true in patients with orbital malignancies. Carcinomatous leptomeningitis can present with cranial neuropathies.27 Amyloidomas are also in the differential.28
Trigeminal neuropathy can be a sequela of autoimmune connective tissue and inflammatory disease (such as undifferentiated and mixed connective tissue diseases, scleroderma,29 Sjögren syndrome,30 and progressive systemic sclerosis31). Chronic immune-mediated demyelinating polyneuropathy (CIDP) or acute immune-mediated demyelinating polyneuropathy (Guillain-Barré syndrome) can also have trigeminal neuropathies present (albeit mostly subclinical in CIDP).32 Corticosteroids are not thought to provide symptomatic relief in Sjögren syndrome.30,33
In approximately 10% of cases, facial numbness occurs as a self-limited entity of unknown etiology.40,41,42,43 The term “trigeminal sensory neuropathy” is used by some authors to indicate this disease; however, the term is used nebulously and can be used to describe conditions that are also painful. The second and third trigeminal divisions are affected most often, and a sensory deficit can usually be detected in the involved dermatome. This condition is only rarely bilateral. Trigeminal motor function is typically normal. Involvement of the peripheral trigeminal branches is most likely the cause,42 and many authors have made an analogy with Bell’s palsy. Most patients recover sensory function over a period of a few weeks to months, but it may be recurrent. A diagnosis of idiopathic facial numbness should not be made until a thorough investigation is undertaken.41,43,44 The main differential is tumor, and there may be transient abnormalities of the trigeminal nerve on MRI, furthering the confusion.45 The etiology usually remains in question until the patient shows improvement. If no improvement occurs, frequent clinical evaluations are recommended to detect a pathologic process early.
Hemimasticatory spasm is a condition of unilateral involuntary jaw closure that is often associated with facial hemiatrophy, but not always.46,47 The contractions may be prolonged but are usually brief, resembling hemifacial spasm. The spasms are painful, and patients may break teeth, develop temporomandibular joint disease, or bite their tongue. Electrodiagnostic investigations have demonstrated peripheral motor nerve demyelination, but the EMG does not typically show denervation. Ephaptic transmission, which refers to the concept of an artificial synapse between contiguous nerves caused by the lateral spread of extraaxonal current through the interstitium, is one possible mechanism. Treatment includes botulinum toxin and carbamazepine, which is the most successful oral medication.
Cavernous Sinus
Masses or inflammation in the superior orbital fissure or cavernous sinus may affect the ophthalmic and maxillary divisions of the trigeminal nerve as well as the oculomotor, trochlear, abducens, and sympathetic nerves. Trobe and associates48 reported pain and impaired sensation in one of seven patients with meningiomas and in four of nine with aneurysms in the cavernous sinus. Trigeminal neurinoma or schwannoma can occur anywhere along the nerve and has also been reported in the cavernous sinus,49 as can metastatic carcinomas.50
Inflammation in the cavernous sinus has been called the Tolosa-Hunt syndrome.51,52,53 Pain is a prominent feature and may precede signs of involvement of the other nerves. It is typically described as a constant “boring” ache behind the eye. Loss of sensation in the ophthalmic more than the maxillary dermatomes is frequent. Corticosteroids can help with both the pain and the speed of cranial nerve recovery. In a related condition called orbital pseudotumor, the inflammation occurs more anteriorly in the actual orbit, rather than in the cavernous sinus. The distinction between Tolosa-Hunt syndrome and orbital pseudotumor may be difficult to make; the underlying immunopathologic deficit perhaps being the same.
Cranial polyneuropathy is a condition of multiple cranial nerve palsies.52,54 Involvement is not limited just to the cavernous sinus, as in the Tolosa-Hunt syndrome. The abducens nerve is the one most often affected, but any combination is possible, with the exception of the olfactory nerve. Causes include the Guillain-Barré syndrome, infections, tumors, carcinomatous meningitis, sarcoidosis, collagen vascular disease, and idiopathic causes. A retrospective review by Juncos and Beal52 found the trigeminal nerve to be involved in 5 of 14 cases of idiopathic cranial polyneuropathy they analyzed. Face and head pain were almost invariable, and corticosteroids provided symptomatic benefit.
Middle Cranial Fossa
Tumors at the base of the skull (chondroma, sarcoma, chordoma, meningioma, and nasopharyngeal tumors) can involve the trigeminal nerve, producing pain or a sensory deficit. In most cases, other cranial nerves are also affected.55,56
Cavernous hemangiomas may occur at Meckel’s cave and along the trigeminal nerve.55 V1 and V2 are usually involved; however, V3 is rarely involved and thought to be related to engorgement of the inferior petrosal sinus and vascular compression at the foramen ovale.57
Facial pain occurring with an oculosympathetic paresis is termed Raeder’s paratrigeminal neuralgia syndrome.58 When this is associated with ocular motor nerve involvement, a mass lesion is usually present in the middle cranial fossa.
Trigeminal Ganglion
The trigeminal ganglion can be affected by infection or tumor. It is frequently the latent reservoir for the neurotropic herpes family reservoir with herpes simplex virus/human herpes virus 1 (HSV/HHV) reactivating to cause herpes labialis and varicella zoster virus/human herpes virus 3 reactivating to cause herpes zoster ophthalmicus. By age 50, 80% to 90% of the population harbors HSV-1.7 Herpes zoster ophthalmicus behaves like zoster infections elsewhere in the body. The incidence is increased in the aged and immunosuppressed. Pain, often severe, may precede, be concurrent with, or follow the vesicular skin eruption. Occasionally, no skin lesions occur; this is referred to as zoster sine herpete. The vesicular eruption usually involves only one dermatome, but severe systemic eruptions can occur (the latter appear most often in immunocompromised persons or in persons with malignancy). Fever, malaise, headache, and lymphadenopathy may be present at the onset. Fifty percent of cases have ocular involvement, which can be predicted if vesicles appear on the lid margin or if Hutchinson’s sign is present.59,60 In 1865, Hutchinson61 noted that when the nasociliary branch to the tip of the nose is affected, intraocular involvement is likely. Vesicular lesions at the tip, side, root of the nose, representing the external nasal and infratrochlear branches of the nasociliary nerve, are associated with ocular changes and corneal denervation.62 Ocular changes include conjunctivitis, episcleritis, scleritis, keratitis, iritis, chorioretinitis, optic neuropathy, glaucoma, ocular motor palsies, and Horner syndrome.59,63,64 On rare occasions, a contralateral hemiplegia may occur (Fig. 36.7).59,65,66,67 A virus-induced angiitis of cerebral vessels has been shown by angiography and pathology in several cases. Although patients with neoplastic or other debilitating diseases are predisposed to the development of herpes zoster infections, a neoplasm is discovered in less than 1% of otherwise healthy persons with cutaneous zoster.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree