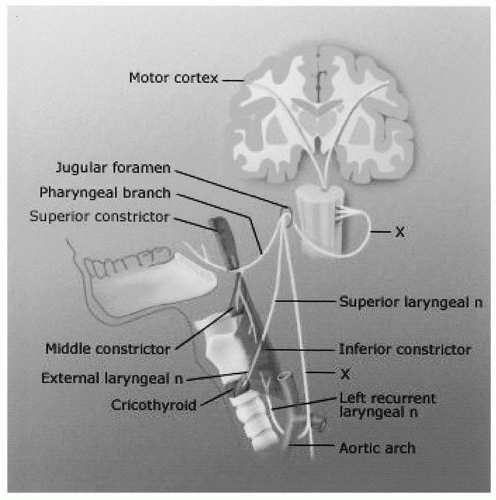
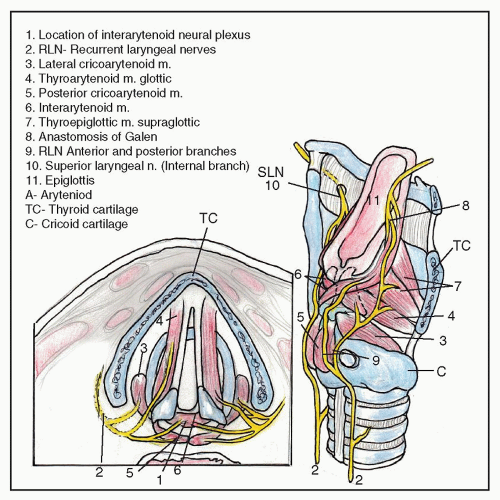
surgery are the causative etiology approximately twice as often as thyroidectomy, 31% versus 16% of all causes, respectively (3). Merati et al. (4) also reported a 47.6% rate of iatrogenic injury in 84 patients with acute vocal fold motion impairment, including 27.5% following cervical spine procedures and 15% after thyroidectomy. While iatrogenic VFP caused by anterior cervical spine surgery is clearly on the rise, it is unclear whether this increase is caused by a change in surgical technique or by a rise in the overall number of these surgeries being performed. The overall incidence of this complication has previously been reported to be around 1% but more recent studies have found a higher rate (11%) of early persistent RLN palsy (5). Netterville et al. (6) has noted that the complication is almost exclusively right-sided and is probably caused by a stretch injury of the RLN by the Cloward retractor. The shorter, more oblique course of the right RLN seems to predispose it to injury (6). Other common iatrogenic surgical causes of UVFP include esophagectomy, thymectomy, neck dissection, mediastinoscopy, and cardiothoracic surgery, including aortic surgery, coronary artery bypass grafting, and pulmonary lobar resection (2,7,8). Endotracheal intubation (9), prolonged nasogastric tube placement, and even esophageal stethoscope placement have been implicated as occasional causes of VFP (Table 69.2).
TABLE 69.1 UNILATERAL VOCAL FOLD IMMOBILITY: CAUSES | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
TABLE 69.2 SURGICAL OR IATROGENIC CAUSES OF VFP | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
Parkinson disease, Shy-Drager syndrome, progressive bulbar palsy, myasthenia gravis, multiple sclerosis, and postpolio syndrome. Almost all of these disorders will have other obvious signs of progressive neurologic disease in addition to unilateral VFP and, in many cases, the neurologic condition has already been diagnosed before the patient presents with voice complaints. Most of these progressive neurologic conditions present with much more complicated (yet often subtle) findings on head and neck examination, such as a bowed vocal fold or vocal fold atrophy (Parkinson, ALS, postpolio syndrome), unexplained vocal fatigue with subtle reduced vocal fold mobility (myasthenia gravis in particular), vocal spasms (ALS), tongue fasciculations (ALS), and dysarthria (ALS, bulbar palsy). It is important to obtain a neurology consultation when a progressive neurologic disease is suspected.
significance (e.g., that lateral vocal fold position indicated complete CN X paralysis caused by RLN and SLN involvement). This theory was later disproved by Woodson (20). The final position of the vocal fold after nerve injury seems to be caused entirely by the degree of reinnervation and synkinesis present, and nothing else.
Arytenoid cartilage edema
Difference in vocal fold level. Generally, an anterior arytenoid cartilage dislocation would result in a lower vertical position of the vocal process on the affected side. Note that vocal fold height differences are also commonly seen with VFP.
Absence of a “jostle sign,” which is a brief lateral movement of the arytenoid cartilage on the immobile side during glottic closure caused by contact from the mobile arytenoid.
and increased vocal fatigue. MPT values of 5 or less indicate severe, uncompensated VFP that may need arytenoid adduction (AA) in addition to medialization laryngoplasty (ML) (24). Poor pulmonary reserve from asthma or chronic obstructive pulmonary disease can reduce MPT significantly, so results need to be taken in context of the patient’s pulmonary status and effort levels. MPT should be expected to improve (i.e., increase) after successful medialization surgery for VFP. The main limitation of MPT is the inability to distinguish between upper and lower airway sources of dysfunction. A clinically more useful indicator of glottic incompetence is the S:Z ratio. In the presence of glottic insufficiency, duration of /z/ phonation is significantly decreased but the duration of /s/ phonation remains unchanged. This phenomenon results in an increased S:Z ratio (greater than 1.4) in these patients as compared to normal controls (1.0) (25).
Venereal Disease Research Laboratory, or fluorescent treponemal antibody absorption test, thyroid function tests, autoimmune panels, or erythrocyte sedimentation rate. Terris et al. (27) studied 84 patients with UVFP in which serologic studies had 0% yield in determining the cause. If additional elements of the history and physical examination point toward a systemic process as the cause of UVFP, directed serology tests may be indicated. In general, however, a “shotgun” approach to the workup of uncomplicated UVFP is unnecessary and wasteful.
Observation for 6 to 12 months, reserving treatment for patients with continued symptoms
Referral to speech pathology for voice strengthening or swallow therapy, as indicated
Get Clinical Tree app for offline access