Treatment of Advanced Stages of Retinopathy of Prematurity
Omesh P. Gupta
William Tasman
The incidence of retinopathy of prematurity (ROP) for early gestational age and low-birth-weight infants has decreased over the past decade despite the fact that medical advances continue to improve the survival rates in very premature infants.1,2,3,4 The use of surfactants and antenatal steroids coupled with improved neonatal nutrition, oxygen monitoring, and ventilation is believed to underlie this improvement.2,3,4
Infant mortality rates for infants treated in neonatal intensive care units correlate directly with weight and gestational age at birth. Infants who weigh between 500 and 749 g or are less than or equal to 23 weeks gestational age (GA) at birth have less than a 50% chance of survival.5 It is these very small infants who are most susceptible to ROP. ROP is identified in greater than 60% of infants of less than 750 g birth weight and less than 20% of infants of between 1,000 and 1,250 g.1,2 The incidence of ROP is less than 10% for infants between 1,250 and 1,500 g birth weight.6,7 The incidence of ROP declines from greater than 70% among infants of less than or equal to 23 weeks GA to less than 10% at 32 weeks GA.1 Fortunately, most cases of acute ROP undergo regression.8 In the minority of cases that progress, various treatments can lead to regression or stabilization of the disease.
INTERNATIONAL CLASSIFICATION
In 1984, the committee for the classification of ROP, a group of 23 ophthalmologists from 11 countries, all with experience in the treatment of ROP, published the international classification of ROP.9 This classification system provided an objective, reproducible method for ophthalmologists to grade the location, stage, and extent of ROP. It enabled the establishment of multicenter, randomized clinical trial for the treatment of ROP (CryoROP study).10,11,12,13 More recently, the Early Treatment for the Retinopathy of Prematurity Study (ETDRS) revised the guidelines.14
LOCATION
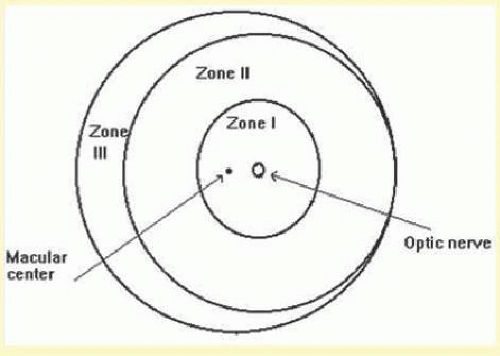
The retina is divided into three zones when identifying the location of ROP (Fig. 108.1). Zone I is a circle centered at the optic nerve with a radius of twice the distance from the optic nerve to the center of the macula. Clinically, this radius of 30 degrees approximates the diameter of field visualized with a 25D indirect lens.15 Zone II extends from the outer edge of zone I to the nasal ora serata, then forms a circle with the optic nerve at the center. Zone III is composed of the remaining crescent of retina temporal to zone II.
EXTENT
Extent is quantified by clock hours of retinal involvement.
STAGING
ROP is divided into stages of severity from stage 1 or demarcation line denoting minimal ROP to stage 5 or total retinal detachment (Table 108.1).
“PLUS” DISEASE
Vascular changes associated with ROP include tortuosity and dilation of the vessels in the peripheral retina and iris. When these changes progress to include the posterior pole, then a plus sign is added to the ROP stage number.
TYPE 1 AND TYPE 2 ROP
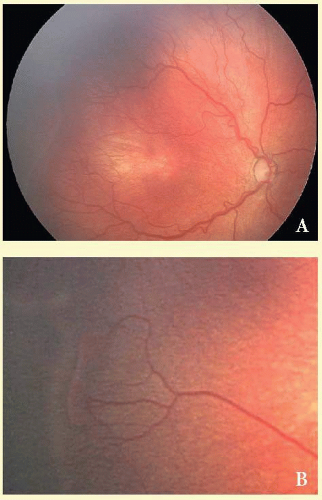
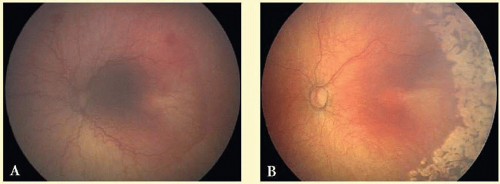
Type 1 ROP is defined as high-risk prethreshold ROP that would normally progress to threshold ROP if left untreated (Fig. 108.2). It is defined as zone 1 with any stage of ROP with plus disease, zone 1 with stage 3 ROP with or without plus disease, or zone II with stage 2 or 3 ROP with plus disease. These eyes should be treated within 48 hours of diagnosis.
Type 2 ROP is defined as zone II with stage 3 without plus disease or zone I with stage 1 or stage 2 ROP without plus disease. These eyes should be watched closely and treated if there is progression to Type 1 ROP. Approximately 22% of Type 2 ROP eyes will progress to Type 1 ROP.16
TABLE 108.1. Stage of ROP as Defined by the International Classification of ROP | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||
CRYOTHERAPY
Indications
Cryotherapy was the first treatment for ROP proven to be effective by multicenter randomized clinical trial. The multicenter trial of cryotherapy for retinopathy of prematurity (CryoROP) study assessed structural and functional outcomes in eyes randomized to either cryotherapy or observation for infants of less than 1,251 g birth weight with threshold disease.13 The presence of a retinal fold involving the fovea, a retinal detachment involving zone I, retrolental tissue, or mass was deemed an unfavorable structural outcome, whereas functional outcome was deemed unfavorable if final best-corrected visual acuity was 20/200 or worse.11,12 Preliminary study results at 3 months indicated a strong structural benefit for eyes with threshold disease treated with cryotherapy; therefore, patient enrollment was halted before the planned completion date.13
Contraindications
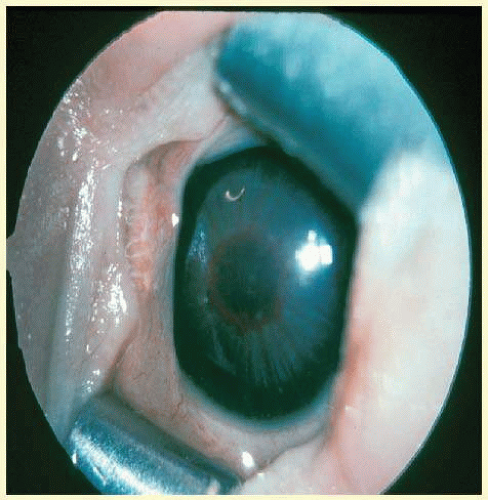
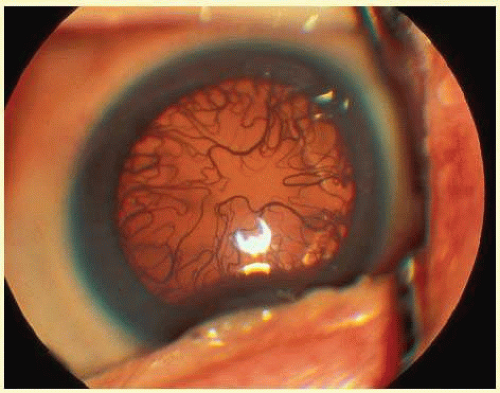
If the fundus view is obscured by anterior segment changes or vitreous hemorrhage, treatment should not be applied (Fig. 108.3). Ultrasonography should be performed when there is no fundus view. If obscuring vitreous hemorrhage is present during the active stage of ROP, vitrectomy should be considered to clear the view for treatment. If anterior segment changes, such as corneal opacity or cataract, are present in both eyes, or in a fellow eye when the other eye is blind, and if retinal detachment is seen on ultrasound, combined anterior segment and vitreous surgery should be considered. The infant’s systemic status, if unstable, may preclude treatment. Treatment should be deferred if the neonatologist believes that treatment may be too stressful for the infant.
Procedure
Cryotherapy can be applied with the use of either topical, local infiltration, or general anesthesia. If topical or local infiltration anesthesia is used, intravenous analgesic and sedative medication should be given.17 With topical anesthesia, the ballooning of the conjunctiva associated with subconjunctival infiltration does not occur. Sometimes, the swelling of the conjunctiva from injection of local anesthesia in addition to the edema that occurs during cryotherapy obscures the cornea, necessitating termination of the procedure. For topical anesthesia, proparacaine hydrochloride 0.5% or tetracaine hydrochloride 0.5% can be applied to the cornea every 20 minutes during treatment. Local infiltration anesthesia can be given by injecting 0.5 mL of lidocaine hydrochloride 1% into the subconjunctival space. To avoid possible cardiopulmonary complications, no more than 0.5 mL should be injected.18 General anesthesia can be used and was given to 28% of the patients in the CryoROP study.19
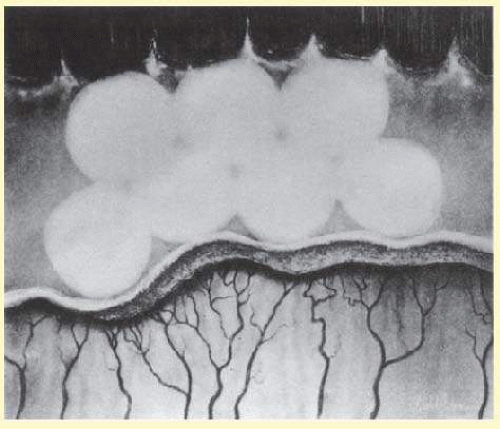
It is best to start treatment nasally because pressure from the cryoprobe will soften the globe and thus facilitate treatment temporally, when the avascular zone usually is more posterior. Contiguous spots of cryotherapy should be applied throughout the entire avascular retina anterior to the ridge and extraretinal fibrovascular proliferation. Usually, it takes just a few seconds for the avascular retina to whiten from the cryotherapy. This whitening is the endpoint for treatment (Fig. 108.4). Typically, depending on the extent of avascular retina, 30 to 50 applications of cryotherapy are necessary to complete treatment. Care should be taken to avoid prolonged scleral depression, which increases the intraocular pressure and risk for central retinal artery occlusion.
Occasionally, corneal clouding occurs during treatment, obscuring the view. Removing the lid speculum and waiting several minutes often results in clearing. If waiting fails, the epithelium may be removed with a cotton-tipped swab soaked in cocaine hydrochloride 4% solution. Use of the swab provides excellent topical anesthesia as well. If removing the corneal epithelium with cocaine does not result in clearing of the cornea, the procedure can be completed later.
Vitreous hemorrhage also may occur during treatment, necessitating termination of the procedure. The hemorrhage usually results from bleeding from a florid area of extraretinal fibrovascular proliferation caused by pressure on the globe from the cryoprobe. Treatment should be completed when the vitreous hemorrhage is clear enough to permit further photocoagulation.
Expected Results
The desired outcome after cryotherapy is regression of both plus disease and extraretinal fibrovascular proliferation. A reduction in both plus disease and extraretinal fibrovascular proliferation should be seen by 1 week after treatment.
Complications and Their Management
Both ocular and systemic complications may occur during cryotherapy for ROP. Systemic complications may be life threatening. All patients treated with cryotherapy have periorbital edema, conjunctival injection, and chemosis. The periorbital edema usually subsides in a few days, whereas the conjunctival injection and chemosis take 1 to 2 weeks to regress. In the CryoROP study, conjunctival or subconjunctival hematoma occurred in 11.7% of treated eyes, and unintended conjunctival laceration occurred in 5.3% of eyes.19 These complications are self-limited and do not lead to long-term problems.
The most frequent and most serious ocular complication in the CryoROP study was intraocular hemorrhage. Retinal, preretinal, and vitreous hemorrhage occurred in 22.3% of treated eyes.19 At times intraocular hemorrhage may prevent the completion of treatment. In such cases, treatment is aborted and the eye is carefully followed for clearing of the hemorrhage so that treatment may be completed.
Late-onset retinal detachment after cryotherapy has been reported.20 Breaks are often found at the border of the cryotherapy-treated and untreated retina. The retinal breaks may occur as a result of the inability of treated retina to stretch as the eye grows or as a result of vitreous traction.21 Surgical treatment includes standard buckling, vitrectomy, or combination techniques although the need for subsequent surgery is as high as 30%.22 An increased risk of rhegmatogenous retinal detachment is present for life.
The incidence of myopia is increased in children with a history of ROP with or without treatment. This increased risk of myopia correlates directly with severity of eye disease. Severe ROP leads more often to high myopia.23 When compared with observed control eyes with threshold ROP, eyes treated with cryotherapy have an increased risk of myopia greater than 8.00 Diopters (D).24
The visual field in eyes with a history of threshold ROP is decreased when compared with normal controls. Results from the CryoROP study at 10 years demonstrated a reduction in visual field area from 30% to 37% in eyes treated with cryotherapy when compared with healthy controls.25 This reduction in visual field was 5% greater for eyes that underwent cryotherapy than in eyes with threshold ROP that did not undergo cryotherapy.
There does not appear to be an increased incidence of amblyopia in eyes with threshold ROP treated with cryotherapy when compared to observed threshold ROP. In the CryoROP study, strabismus surgery was required for 10% of eyes treated with cryotherapy and 7% of control eyes with threshold ROP.26
LASER PHOTOCOAGULATION
Indications
Photocoagulation was the first modality of treatment of the anterior avascular zone for acute ROP. In 1967, Nagata27,28 used xenon arc photocoagulation to induce regression of acute ROP. This method of treatment was difficult to deliver, and it was supplanted by cryotherapy.
In the late 1980s, indirect ophthalmoscopic delivery systems for laser photocoagulation became available. Shortly thereafter, McNamara and colleagues,29 Iverson and colleagues,30 (Table 108.2) Hunter and Repka31 (Table 108.3) compared transpupillary laser photocoagulation with cryotherapy for threshold ROP in prospective, randomized trials. An argon laser was used to treat in the McNamara et al.29 and Iverson et al.30 studies, whereas a diode laser was used in the Hunter and Repka31 study. Similar favorable regression rates were reported with either treatment in each of these studies. McGregor and colleagues32 and Pearce and colleagues33 retrospectively compared laser photocoagulation with cryotherapy for threshold ROP. No statistical difference was found between the two treatment groups.
TABLE 108.2 Prospective, Randomized, Comparison of Indirect Ophthalmoscopic Delivery of Diode Laser to Cryotherapy for Threshold ROP | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||
Seiberth and colleagues34 compared transscleral with transpupillary diode laser photocoagulation for the treatment of threshold ROP. They found transscleral diode laser to be as effective as transpupillary diode laser in causing regression of threshold ROP.
Contraindications
Treatment of threshold ROP with laser photocoagulation requires clear media. Anterior segment changes, such as cataract and corneal clouding, and vitreous hemorrhage may preclude laser treatment, yet the media may be clear enough for cryotherapy to be performed. A persistent tunica vasculosa lentis may lead to cataract formation with argon laser photocoagulation.35 This risk may be decreased when using a diode laser36 (Fig. 108.5).
Systemic contraindications may be fewer with laser photocoagulation than with cryotherapy, perhaps due to the less invasive nature of the procedure.31
Procedure
Indirect laser photocoagulation for ROP should be performed with either topical anesthetic supplemented with intravenous analgesia and sedation or with general anesthesia.17 There is no need for local infiltration anesthesia because only gentle manipulation of the globe with a scleral depressor is necessary. McNamara and others29 used topical anesthesia alone, Hunter and Repka31 used topical anesthesia supplemented with systemic analgesia, whereas all infants treated by Iverson and colleagues30 and Landers and associates37 were given general anesthesia. Transient bradycardia occurred in three patients (19%) treated with laser and three (25%) treated with cryotherapy in the McNamara study. The bradycardia resolved when the scleral depressor or cryoprobe was removed for several seconds. Intubation following apnea and bradycardia was required for two patients (13%) treated with cryotherapy and none with laser in the Hunter and Repka31 study. No other systemic complications occurred.
TABLE 108.3 Prospective, Randomized, Comparison of Indirect Ophthalmoscopic Delivery of Argon Laser to Cryotherapy for Treatment of Threshold ROP | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||
Before laser photocoagulation, the pupils are dilated. Homatropine hydrobromide 2% and phenylephrine hydrochloride 2.5% drops instilled 3, 2, and 1 hours preoperatively provide adequate dilation. If topical anesthesia supplemented with systemic analgesia and sedation is to be used, treatment can be performed in the operating room or in the nursery. Proparacaine hydrochloride 0.5% is applied to the globe. A lid speculum is placed. Gentle manipulation with a scleral depressor to position the globe for viewing of the peripheral retina is performed. Although the peripheral retina is viewed through the indirect ophthalmoscope, the laser aiming beam can be seen. When the aiming beam is in focus on the peripheral avascular retina, laser discharge is accomplished by pressing a foot pedal. Care should be taken to assure that the aiming beam is focused before discharge to avoid laser burns in unwanted areas, such as the posterior vascularized retina, cornea, iris, and lens. The entire anterior avascular retina should be treated with avoidance of laser to the active ridge. Steinmetz and Brooks38 have suggested a possible benefit to treating the ridge in addition to the avascular retina, but to date no statistical benefit has been shown. Laser spots should be placed one-half burn width apart, with a dull white laser photocoagulation mark used as the endpoint. Banach and colleagues39 have demonstrated an increased risk of disease progression with a less dense laser photocoagulation pattern. For argon laser, the initial power setting should be 200 mW, with a duration of 0.1 second. The power can be increased gradually to achieve the appropriate laser mark in the fundus. For diode laser, the initial power setting should be slightly lower (150 mW), with a longer duration (0.2 second), to avoid causing choroidal hemorrhage and rupture of Bruch’s membrane. A neonatologist or anesthesiologist should monitor the infant during treatment.
The advent of portable diode and argon lasers has simplified the treatment of ROP and has largely supplanted cryotherapy as the treatment of choice for ROP at most centers. The portable laser can be brought directly into the nursery, which avoids the need for costly and sometimes hazardous transfer of the infant to a facility with a freestanding laser. For safety reasons, treatment should be performed in an isolated room in the nursery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree