Trauma: Ruptures and Bleeding
Earl R. Crouch Jr
Eric R. Crouch
Trauma to the Cornea
Blunt ocular trauma can cause a number of corneal complications. Minor ocular trauma results in corneal abrasion and avulsion of corneal epithelium. Mild cases can be treated with topical antibiotic ointment and observation for resolution. Large epithelial defects may benefit from a bandage contact lens, and poor healing may be indicative of an anterior basement membrane dystrophy. Ocular trauma can also involve corneal contusions and edema, which can clear with observation or can be treated with topical steroids. In severe cases, blunt trauma can even result in corneal penetration and rupture with release of aqueous fluid or intraocular contents. Care should be taken to thoroughly evaluate patients with ocular trauma for direct corneal lacerations or the less common lamellar corneal lacerations. Seidel testing with fluorescein should be applied and evaluated with blue light illumination to rule out an anterior chamber leak. Typically, lamellar corneal lacerations seem to occur more frequently in cases of oblique collision with the object of injury. Treatment of corneal lacerations should be promptly surgically repaired and then treated with topical broad spectrum antibiotic coverage.
Trauma to the Uveal Tract
Blunt ocular trauma frequently causes injury to the uveal tract.1,2,3 Injury to the uvea may be either direct or contrecoup. The sudden, transient increase of intraocular pressure (IOP) following impact is probably insufficient to produce much of the damage usually encountered. One possible mechanism of injury is direct contusion of the cornea, which produces a marked posterior displacement, first of the cornea and then of the iris-lens diaphragm. This places great stress on the area of the iris root and on the zonules attached to the ciliary body. In addition, the sclera expands in circumference in the area of the ciliary body. The ciliary body follows suit; however, its greater mass causes a lag in expansion, with possible subsequent separation within the uveal tissues or between the ciliary body and the sclera.1,4,5 An alternative explanation for damage is the creation of a pressure wave that travels posteriorly within the aqueous. As the pressure wave strikes the lens, it is directed laterally and circumferentially, which results in a concentration of its force at the iris root.
Trauma to the Iris
Iritis
Traumatic iritis is the mildest result of blunt injury to the iris. Contusion injury to the iris stroma results in anterior chamber flare and cells; it is accompanied by aching, photophobia, and ciliary injection. Disruption of the blood–aqueous barrier may accompany blunt trauma. Traumatic iritis is occasionally accompanied by pain with direct illumination into the uninjured, contralateral eye.6 Topical treatment of cycloplegia produces symptomatic improvement, and spontaneous recovery is the rule. In more severe iritis, topical steroids provide comfort and hasten recovery.
Pigmentary Changes
Visible scattering of intraocular pigment almost always follows ocular trauma. A fine dusting of iris pigment released from the iris pigmented epithelium may be observed on the corneal endothelium, trabecular meshwork, anterior lens capsule, iris surface, and occasionally in the vitreous. Small clusters of dense pigment may remain on the stromal surface of the iris, indicating previous hyphema.7 A Vossius lenticular ring may give additional evidence of ocular trauma. A Vossius ring is a full or partial circle of pigment deposited on the anterior capsule of the lens, corresponding to the pupillary margin. A Vossius ring is usually noted after dilation of the pupil with mydriatics. This imprint of iris epithelial pigment is probably deposited on the lens surface as the aqueous pressure wave momentarily compresses the pupillary margin of the iris against the lens. The Vossius ring usually disappears with time. Occasionally, remnants may remain permanently on the anterior surface of the lens.
Hemorrhage
Hemorrhage into the iris stroma is an infrequent posttraumatic finding. The vessels in the iris stroma have the capacity to retract and close spontaneously. Therefore, hemorrhage within the iris stroma is usually confined to very small areas that rapidly turn brown or totally disappear.
Sphincter Alterations
Miosis and Mydriasis
The response of the iris and the ciliary body to concussive force may be either irritative or paralytic. A mild concussion might cause a traumatic miosis and spasm of accommodation. Miosis may also accompany a posttraumatic iritis. More serious injury can cause a traumatic mydriasis and cycloplegia. Mydriasis and miosis are usually short lived and improve spontaneously. The ciliary and iris muscles are usually affected simultaneously; however, iridoplegia may be present without impairment of accommodation. Iris stromal tears may appear centrally as sphincter damage or peripherally at the iris root as iridodialysis.
Sphincter Rupture
Rupture of the iris sphincter is sometimes produced by blunt trauma. Single or multiple tears in the iris stroma (with iris sphincter involvement) can cause small triangular defects or teardrop defects in the pupillary border. Anterior concussion causes the aqueous to press the iris against the lens, resulting in forced pupillary dilation as the pupil and sphincter are forced to dilate over the dome of the anterior lens capsule. With blunt trauma, the pupil dilates rapidly and may induce shearing forces in the sphincter pupillae. At the same time, there is reflex constriction of the pupillary sphincter. When the limits of elasticity of the iris stroma are reached, the weakest areas rupture.
Part or all of the iris thickness may be involved. Small strands of anterior iris stroma may show separation; alternatively, only the posterior pigment epithelium and sphincter may be affected. Transillumination of the iris tissue is often required for recognition of the latter. Most frequently, all layers tear simultaneously, leaving a notch in the pupillary border or a triangular defect with its apex directed toward the iris root. Torn iris tissue is a permanent defect, because the iris is unable to repair its structure.
Iridoschisis
Blunt trauma can cause iridoschisis, that is, a portion of the anterior mesodermal leaf of the iris detaches from the posterior iris stroma or epithelium. The detached leaf of anterior stroma then floats forward into the anterior chamber. Alternatively, detached segments of the iris stroma may atrophy, leaving areas of thin, depigmented stroma or bare pigment epithelium.
Iridodialysis
In iridodialysis, the base of the iris separates from the ciliary body and scleral spur (Fig. 61.1). The iris root is the thinnest and weakest area of the iris. With severe concussive injuries, a large sector of the iris root may tear free, creating an accessory pupil of significant size at the limbus. This type of injury may be associated with a large hyphema; therefore, the iris injury may not be noted until the hyphema clears. The iridodialysis seldom requires treatment, except when it causes symptomatic monocular diplopia. Indeed, corrective measures should be deferred until the vision is stable, because iridodialysis is frequently associated with other, more serious injuries, which preclude good vision. Persistent diplopia can be corrected by resuturing the iris with 10-0 polypropylene suture.8 For McCannel iris techniques, a longer needle such as Ethicon CIF-4 is required.9,68 Various techniques have been used to repair an iridodialysis with a closed anterior chamber, using partial thickness scleral flaps similar to those used for sulcus fixation of posterior chamber intraocular lenses. The procedure should be undertaken with caution. The potential exists to produce a recurrent hyphema, because the incision may have to pass through a proliferative scar from the original injury.
Trauma to the Ciliary Body
Angle Recessions
Blunt contusion of the globe frequently causes damage to the anterior chamber angle, with or without bleeding into the anterior chamber.2 Concussive force produces a cleft in the ciliary body, usually between the circular and the longitudinal muscle fibers, beginning at the scleral spur. Less frequently, the cleavage may occur either more centrally in the circular muscle fibers or more peripherally, producing a cyclodialysis cleft.
Minor angle damage may be recognized as a separation of the strands of the uveal meshwork.10,11 The recessed meshwork tends to be disrupted in its midportion, resulting in small tufts of uveal tissue bunching up on the iris root and on the trabeculum above the scleral spur. This leaves the ciliary body bare in comparison with other quadrants of the angle or with the angle of the opposite eye. With mild injuries of this sort, the examiner should compare the gonioscopic appearance of both eyes simultaneously with two Koeppe lenses (compare two parts of the angle of one eye) to identify subtle changes in the injured angle.
In more severe injuries, the cleft extends into the ciliary body. The light gray portion of the ciliary band appears broadened, and the scleral spur is more distinctly white and prominent (Fig. 61.2). Small peripheral anterior areas of synechiae frequently appear at the lateral limits of an angle recession. In contrast, a cyclodialysis cleft, in which a sector of the ciliary body is totally detached from the sclera, is recognizable by the area of white sclera that is visible posterior to the scleral spur.
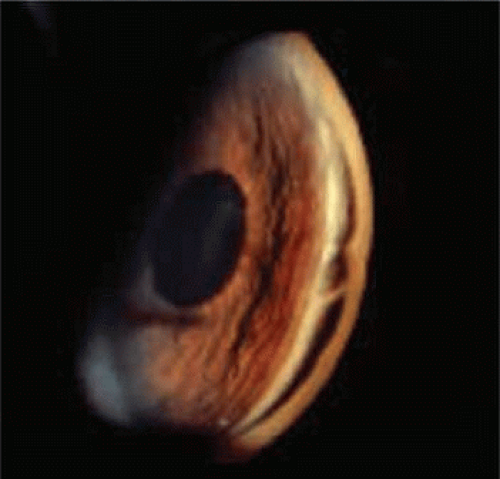 Figure 61.2. Angle recession involving left eye following traumatic hyphema in a 17-year-old boy after paintball injury. |
Although angle recessions can occur without anterior chamber hemorrhage, most patients have coexistent hyphemas. Careful gonioscopy has revealed that between 71% to 10% and 86% to 11% of traumatized eyes with hyphemas have an angle recession. The degree of angle recession is usually not related to the amount of hyphema; some small hyphemas had large, deep recessions, whereas some minor angle recessions were associated with large hyphemas.10
Angle recession is a frequent cause of unilateral glaucoma. Blanton12 described two posttraumatic periods of pressure elevation; these periods are exclusive of the initial elevation of IOP during the acute phase of the hyphema. The first period usually begins 2 months to 2 years after the injury. Ocular hypertension at this time is often related to the degree of angle recession and is difficult to control with medical therapy. This period of hypertension may last for several months to several years, and it frequently subsides spontaneously (provided a large portion of the angle is not obstructed by peripheral anterior synechiae). However, optic nerve damage can occur if hypertension is uncontrolled. Medical therapy, although it produces poor control at times, should be relied on to “weather” this early period of glaucoma.
A late period of elevated IOP may occur 10 to 15 years after the injury. Eyes with recessions greater than 180 degrees are particularly susceptible to this late-occurring glaucoma; nearly all patient whose angle damage approaches 270 to 360 degrees develop late elevations of IOP.12,13 The incidence of this glaucoma is approximately 7% in persons with angle recession.
In addition, angle recession of any degree seems to further elevate IOP in an eye in which primary open-angle glaucoma has coincidentally developed. Thus, inadequate IOP control and glaucomatous damage can occur more frequently in an injured eye that has concomitant open-angle glaucoma and an angle recession.
Patients with known blunt trauma should be examined gonioscopically for angle recession; if angle recession is found, they should be advised to have yearly examinations for the rest of their lives to detect late-occurring glaucoma.14
Cyclodialysis
Occasionally, the ciliary body attachment to the scleral spur separates. Temporary or permanent hypotony can result. A cyclodialysis cleft should be suspected if IOP is less than 9 or 10 mm Hg. Occasionally, the full thickness of the ciliary body detaches from the sclera, producing a cyclodialysis cleft. Localization of the cyclodialysis cleft can be visualized with scleral transillumination, biomicroscopy, and ultrasound.15,16 Traumatic cyclodialysis can occur as a small or a wide cleft; either size cleft can result in low intraocular tensions that continue as long as the cleft remains open. Spontaneous closure of the cleft (or operative closure to correct hypotony) may produce rapid elevation of IOP to dramatically high levels.
Rarely, blunt trauma produces complete ciliary and choroidal detachment, that is, 360 degrees. This allows the iris-lens diaphragm and ciliary body to shift markedly backward or forward, frequently resulting in hypotony and phthisis bulbi.
Traumatic Hyphema
The accumulation of blood in the anterior chamber is one of the most challenging clinical entities encountered by the ophthalmologist. Even a small hyphema can be a sign of major intraocular trauma with associated damage to other intraocular tissues. Blunt trauma causes anterior chamber angle distortion that can result in iris or ciliary body vessel rupture and bleeding into the anterior chamber. The lack of an ideal therapeutic program, the potential for secondary hemorrhage, and the secondary onset of glaucoma all threaten to turn an eye with an initially good visual prognosis into a complex therapeutic problem with poor final visual result.
Evaluation
The patient with traumatic hyphema should have a thorough ophthalmologic evaluation. The time of onset and type of injury should be accurately recorded. The object producing the injury and the method by which it produced the injury give additional insight into the amount of ocular damage. If several days have elapsed since the injury, the onset of diminished visual acuity or an increase in ocular pain is significant, because they may indicate secondary hemorrhage or elevated IOP. Any other history of ocular problems, including amblyopia, should be recorded.
The ocular examination should be thorough and include the surrounding tissues.13 Resist the temptation to abandon the examination when unable to see beyond the anterior chamber. Search the periorbital skin for areas of hypesthesia or anesthesia. Blow-out fractures frequently accompany hyphemas.13 Examine the lids and conjunctiva carefully for laceration. Disproportionate conjunctival edema should be regarded as a possible indication of scleral rupture until proved otherwise. Proptosis may indicate retrobulbar hemorrhage; enophthalmos or restrictions in extraocular muscle rotations suggest a blow-out fracture. Record visual acuity of both eyes with best optical correction. If the acuity of the injured eye is markedly reduced, test light projection and color sense, including evaluations of consensual reaction of the pupil of the uninjured eye. Poor visual acuity that does not correlate with the extent of the hyphema suggests concomitant optic nerve or macular damage.
Examine the cornea for damage or pre-existing disease, because any compromise of the endothelium increases its susceptibility to blood staining. Record the amount and character of the hyphema as it first presents. Careful documentation of the shape of the clot or of the level of free cells aids in evaluation of secondary hemorrhage or resolution of the hyphema. Note iris and lens details that are visible beyond the hyphema, including pupillary size and shape, sphincter tears, iridodialysis, and dislocation of the lens or other injuries. Attempt to examine the fundus, but it may be difficult until the hyphema has resolved.
Dilate the pupil if suspecting intraocular foreign body, rupture or perforation of the globe, or retinal tears or detachment. In most instances, a fundus evaluation is preferred when the media is clear following resolution of the hyphema. Record intraocular pressure by applanation tonometry after ruling out a ruptured globe. Avoid gonioscopy until day 14 to avoid a secondary hemorrhage.
Many patients with hyphema appear somnolent. The exact mechanism for this reaction is not clear. If associated head trauma is possible, obtain neurologic consultation and magnetic resonance imaging (MRI). Laboratory data should include platelet count, prothrombin time, and partial thromboplastin time (PTT). All black and Hispanic patients require a sickle cell prep and, if positive, hemoglobin electrophoresis.
Characteristics and Classification
Traumatic hyphema is most often encountered in children. Reports indicate a prevalence of 70% or greater in the pediatric population.17 Males are involved in three-fourths of cases.11,17 Studies in the United States indicate that hyphemas occur in approximately 20 per 100,000 population per year.18
The agent producing a hyphema is usually a projectile that strikes the exposed portion of the eye. Sources of trauma have included balls, rocks, projectile toys, air-gun or BB pellets, paint ball pellets, and the human fist19,20,21 (Figs. 61.3 and 61.4). Fists and belts have started to play a prominent role in cases of child abuse. Sports injuries, including soccer, hockey, baseball, and basketball injuries, account for up to 60% of traumatic hyphemas, which in many cases are related to lack of protective eye gear.
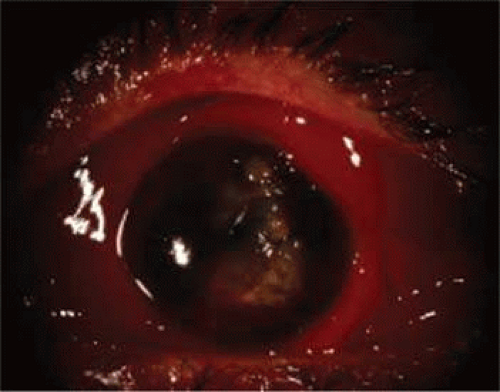 Figure 61.4. Penetrating globe injury with residual hyphema following primary repair in a 12-year-old boy after BB gun pellet injury. |
Rarely, spontaneous hyphemas may occur and be confused with traumatic hyphemas. Spontaneous hyphemas are secondary to neovascularization, ocular neoplasms including retinoblastoma and malignant melanoma, uveitis, and vascular anomalies (juvenile xanthogranuloma). Vascular tufts that exist at the pupillary border have been implicated in spontaneous hyphema.22
The level of hyphema has been classified into four grades, according to the amount of blood occupying the anterior chamber. Most hyphemas (58%) are classified as grade 1 (Fig. 61.5), meaning the hyphema consists of layered blood occupying less than one-third of the anterior chamber. Microhyphema is a grade 1 hyphema. It is defined as a suspension of erythrocytes in the anterior chamber without formation of a layered clot. In grade 2 hyphema, blood fills one-third to one-half of the anterior chamber; 20% of hyphemas are classified as grade 2. Grade 3 hyphema is layered blood filling one-half to less than the total anterior chamber, and 14% fall into this grade. Grade 4 is the least commonly occurring (8%) hyphema (Fig. 61.6), and it is defined as a total clotted hyphema, often referred to as blackball or eightball hyphema.
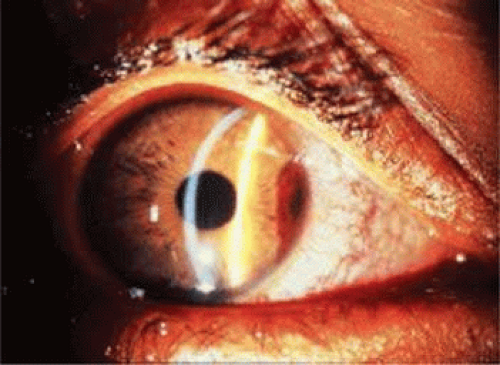 Figure 61.5. Grade 1 hyphema treated with systemic aminocaproic acid in a 9-year-old boy. Note the hyphema from the 2 to 4 o’clock position. The hyphema resolved after 4 days. |
 Figure 61.6. A total hyphema in an 11-year-old boy. Intraocular pressure (IOP) was normal initially, but within 24 hours increased to 40 mm Hg. |
Slightly fewer than half of all hyphemas settle inferiorly to form a level. Approximately 40% form a definite clot, usually adherent to the iris stroma; 10% have a dark clot in contact with the endothelium.11 An alternative method of grading hyphemas involves measuring (in millimeters) the hyphema from the inferior 6-o’clock limbus. This method may help in monitoring the progress of resolution or the occurrence of rebleeding. Digital imaging analysis is also useful and objective, but is available in only a few research or academic facilities.
The source of anterior chamber hemorrhage in contusion injuries is thought to be related to the displacement of tissues posteriorly or to the resultant fluid wave in the aqueous and vitreous, which stretches the limbal vessels and displaces the iris and lens. This usually results in a tear at the iris or ciliary body, usually at the angle structures.12,21 A tear at the anterior aspect of the ciliary body, the most common site of bleeding, occurs in about 71% of cases.11,18 The blood exits from the anterior chamber via the trabecular meshwork and Schlemm’s canal or the juxtacanalicular tissue. The usual duration of the uncomplicated hyphema is 5 to 6 days.
Elevated Intraocular Pressure
Increased IOP may accompany hyphemas of any size. Elevated IOP (>24 mm Hg) may be anticipated in approximately 24% to 32% of all patients with a hyphema at some time during their first 7 days of treatment.11 Higher, more prolonged elevations of IOP are more commonly associated with near total or total hyphemas.
The types of glaucoma in hyphema have a variety of mechanisms. The pathogenesis of the glaucoma related to erythrocytes in the anterior chamber includes a trabecular blockage with normal or sickle erythrocytes. Pupillary block glaucoma occurs as a result of large hyphemas. Glaucoma not related to the erythrocytes in the anterior chamber includes angle recession glaucoma, involving greater than 180 degrees of the trabecular meshwork. More than 270 degrees of angle recession definitely indicates significant long-term glaucoma. Blanton12 described two periods of elevated IOP, between 2 months and 2 years after the injury and 10 to 15 years after the injury. The degree of angle recession is not related to the amount of hyphema. Some small hyphemas produce large, deep recessions. In traumatic hyphema, if 180 degrees or more angle recession is found, the patient needs periodic examinations to detect late-occurring glaucoma.12 Glaucoma related to peripheral anterior synechiae occurs after 9 days of blood in the anterior chamber.12 Posterior synechiae with iris bombé may also occur.
These highly elevated IOP occur during the acute phase of the hyphema and are separate from those related to angle recession.11,12 In patients with pressure elevations, abnormal tonometric readings are frequently detected during the first 24 hours after injury. This initial period of elevated IOP is often followed by a period of normal or below normal pressure from the second to the sixth day.
The early period of elevated IOP is the result of trabecular plugging by erythrocytes and fibrin. The following period of reduced pressure is most likely owing to reduced aqueous production. A period of hypotony is commonly followed by a subsequent rise in IOP with the recovery of the ciliary body. In traumatic hyphema, control of elevated IOP is important to prevent complications of optic atrophy and corneal blood staining. Exceptions to this are those patients with a hyphema occupying greater than 75% of the anterior chamber and patients with total hyphema, in whom pressure elevation frequently has its onset simultaneously with the initial hyphema and remains continually elevated until the hyphema has had considerable resolution. When large segments of the anterior chamber angle are irreparably damaged or when organization of the fibrin or clot produces extensive peripheral anterior synechiae, the intraocular hypertension continues, becoming intractable glaucoma.
Ghost cell glaucoma with hyphema and vitreous hemorrhage may cause elevation of IOP 2 weeks to 3 months after initial injury.23 There is gradual clearing of the hyphema with erythrocytes losing hemoglobin and becoming so-called ghost cells in the vitreous cavity. The ghost cells then press forward into the anterior chamber, with resultant later elevation of IOP.
Secondary Hemorrhage
Secondary hemorrhage occurs in 18% of all patients with a hyphema, with a range between 6.5% and 38%.10,18,21,23,24,25,26,27,28,29 The incidence of secondary hemorrhage is higher in grade 3 and 4 hyphemas.21 Two reports indicate a higher rate of secondary hemorrhage in pediatric patients younger than 6 years.11,24 Frequently, a relationship exists between child abuse and hyphema in this younger age group. Secondary hemorrhage usually occurs on the third or fourth day, but it may occur from the second to seventh day after trauma.11
Secondary hemorrhage is caused by lysis and retraction of the clot and fibrin aggregates that have occluded the initially traumatized vessel. Approximately one-third of all secondary hemorrhages proceed to total hyphema. In patients with a total hyphema at onset, in which the blood is dark and clotted, bright red blood often appears at the periphery of the clot on the fourth to sixth day. This is the result of early dissolution of the clot and does not necessarily indicate a secondary hemorrhage.
Secondary hemorrhage into the anterior chamber results in a markedly worse prognosis. Some have noted a complication rate of 22% in eyes without secondary hemorrhage but in 58% of eyes that rebled.17 The secondary bleeding can result in increased IOP and corneal blood staining and is associated with a poorer visual prognosis. Eventual visual recovery to an acuity of 20/50 (6/15) (the metric equivalent is given in parentheses after the Snellen notation) or better occurs in approximately 64% of patients with secondary hemorrhage, as compared with 79.5% of those in whom no rebleeding occurred.10,11,21 Studies have indicated that the incidence of glaucoma following secondary hemorrhage may be as high as 50%. Improved surgical technique and earlier intervention have, however, improved the final visual outcome.27,28,29
Several studies have documented that secondary hemorrhage occurs more frequently in black and Hispanic patients. Spoor et al.24 observed secondary hemorrhage in 24.2% of blacks and in only 4.5% of whites. Three other studies have demonstrated greater secondary hemorrhage rates in black patients that were highly significant (P <0.05).21,25,26,27,28,29 In our recently reported series, 20% (11 of 54) of the controls had a positive test for sickle cell trait (SA hemoglobin), as measured by hemoglobin electrophoresis. Twelve patients in the control group had secondary hemorrhage, 10 (83%) of whom were black.27 Thus, 32% (10 of 31) of the black patients in the control group had secondary hemorrhage, and 40% of them had SA hemoglobin. Other reports have observed a higher incidence of complications and secondary hemorrhage in this population.18,21,24,28,29,30 In our previous series, three of eight (37%) such patients with SA hemoglobin had secondary hemorrhage.21
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree