Chapter 4 Thyroiditis
Introduction
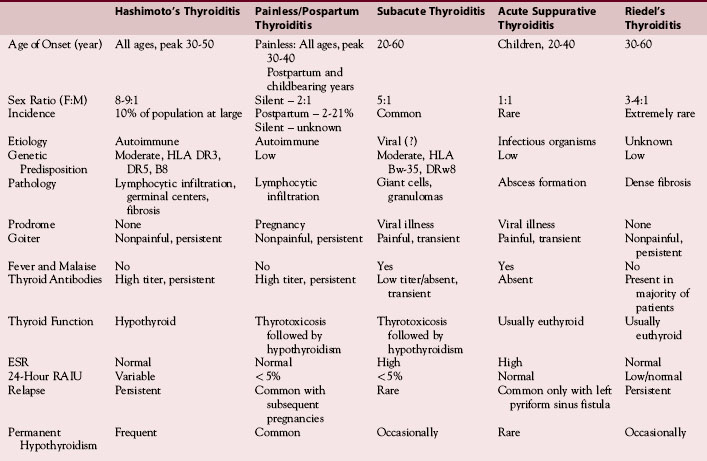
Thyroiditis comprises a diverse group of disorders that are among the most common endocrine abnormalities encountered in medical endocrine clinical practice as well as by surgeons managing the thyroid. These disorders range from the extremely common chronic lymphocytic thyroiditis (Hashimoto’s thyroiditis) to the extremely rare invasive fibrous thyroiditis (Riedel’s thyroiditis) (Table 4-1). Clinical presentations are also diverse, ranging from an incidental finding of a goiter to potentially life-threatening illness, from hypothyroidism to thyrotoxicosis. The term thyroiditis implies that the disorders described in this section are inflammatory processes involving the thyroid gland, although some of the lesions are not inflammatory and are included in the thyroiditis category largely for convenience. A rational approach to such patients, including history, physical examination, laboratory evaluation, radionuclide or ultrasonographic imaging, and fine-needle aspiration biopsy, will allow the appropriate diagnosis to be made in the majority of cases. This chapter reviews the evaluation, diagnosis, and management of thyroiditis in descending order of clinical frequency.
Table 4-1 Types of Thyroiditis (most common to least common)
| Chronic lymphocytic thyroiditis (Hashimoto’s thyroiditis) |
| Subacute lymphocytic thyroiditis |
| —Postpartum thyroiditis |
| —Sporadic silent thyroiditis |
| Subacute granulomatous thyroiditis (De Quervain’s thyroiditis) |
| Drug-induced thyroiditis |
| Radiation thyroiditis |
| Acute suppurative/infectious thyroiditis |
| —Bacterial, fungal, parasitic |
| Invasive fibrous thyroiditis (Riedel’s thyroiditis) |
| Miscellaneous |
| —Sarcoid, amyloid, traumatic, and palpation-induced thyroiditis |
Hashimoto’s Thyroiditis
Autoimmune thyroiditis, also known as struma lymphomatosa, chronic lymphocytic thyroiditis, and Hashimoto’s thyroiditis, was first described by Hashimoto in 1912 (Table 4-2). He described four patients with goiters, the thyroid histology of which were all characterized by diffuse lymphocytic infiltration, atrophy of parenchymal cells, fibrosis, and eosinophilic change in some of the parenchymal cells. Although this condition is common, there are several variants that differ somewhat from the one initially described by Hashimoto.1 Classically, the disorder occurs as a painless diffuse goiter (goitrous form) in a young or middle-aged woman and often presents as an incidental finding during a routine physical examination. The atrophic form of Hashimoto’s thyroiditis is less common and is usually diagnosed by the presence of thyroid antibodies in the hypothyroid patient with a normal-sized or atrophic thyroid. Although high circulating titers of antibodies to thyroid peroxidase (primarily) or thyroglobulin (less often) are almost always present, some patients with Hashimoto’s thyroiditis do not have antibodies but do have a heterogeneous pattern on thyroid ultrasound.2
In iodine-sufficient countries, the most common cause of goiter, hypothyroidism, and elevated thyroid antibody levels is Hashimoto’s thyroiditis. The incidence of autoimmune thyroiditis has increased over the past three generations, perhaps because of the increase in iodine intake that has occurred in the Western world.1 Elevated serum thyroid antibody concentrations are found in approximately 10% of the United States population and in up to 25% of U.S. women over the age of 60.1 About 45% of older women will have lymphocytic infiltration within the thyroid gland. Autoimmune thyroiditis has a female predominance with reported female-to-male ratios ranging between 5 to 1 and 9 to 1.
Pathogenesis
Although it is clear that Hashimoto’s thyroiditis is an autoimmune disease, the nature of the autoimmune process is still debated. These disorders tend to aggregate in families, and a genetic link has been suggested. There have been associations between human leukocyte antigen (HLA)-DR3, HLA-DR4, and HLD-DR5, and Hashimoto’s thyroiditis; however, this was demonstrated only in a cohort of Caucasian individuals.1 Although HLA genes may be critical to the development of Hashimoto’s thyroiditis, this weak association makes it clear that there are other genes that have not been identified as yet which play a role in this multigenic disease. Smoking has also been interestingly identified both as a risk factor for hypothyroidism3 and to protect against hypothyroidism.4
The defect in immunoregulation is currently a matter of debate. Human T lymphotropic virus-1 (HTLV-1) has been reported to be associated with autoimmune diseases, and carriers of the virus have been shown to have a higher frequency of thyroid antibody positivity as well as a higher incidence of Hashimoto’s thyroiditis compared to controls.5,6 Other theories hold that thyrocyte expression of class I and class II genes allows the thyrocyte to present antigen and thus induce autoimmune thyroid disease; however, in contrast, available evidence indicates that thyrocyte expression of these genes promotes anergy and thus may protect against autoimmune thyroid disease.7 The prime defect probably lies in antigen-presenting genes in antigen-presenting cells, like macrophages, such that specific regulatory T lymphocytes are not fully activated.8 This, together with environmental factors that may serve to down-regulate the immune system, may act together to disturb immunoregulation and allow for the development of autoimmune thyroid disease.
Many antibodies are often present in patients with Hashimoto’s thyroiditis. Antithyroid peroxidase antibodies are complement fixing and are detectable in about 90% of patients with Hashimoto’s thyroiditis. Antithyroglobulin antibody, a non-complement-fixing antibody, is found in about 20% to 50% of patients with Hashimoto’s thyroiditis.9 Thyrotropin (TSH) receptor antibodies that block TSH binding but do not stimulate the thyroid cell function may play a role in the clinical presentation of Hashimoto’s thyroiditis, producing or exacerbating hypothyroidism in the absence of significant thyroid gland destruction.10 Such antibodies have been reported to bind to epitopes near the carboxyl end of the TSH receptor extracellular domain, in contrast to thyroid-stimulating antibodies, which bind to epitopes near the amino terminus.11 The prevalence of TSH receptor blocking antibodies in adult hypothyroid patients has been reported to be as high as 10%12 and a decrease in the titer of these antibodies is likely to be responsible for “remission” of hypothyroidism in some patients with Hashimoto’s thyroiditis.13 Antibodies to colloid antigen, other thyroid autoantigens, T4 and T3, as well as other growth promoting and inhibiting antibodies may also be present.
Pathologically, there is lymphocytic infiltration of equal proportions of T and B cells and the formation of germinal centers (see Chapter 44, Surgical Pathology of the Thyroid Gland). The follicular cells undergo metaplasia into larger, eosinophilic cells known as Hurthle or Askanazy cells, which are packed with mitochondria. These cells exhibit high metabolic activity but ineffective hormonogenesis. There is progressive fibrosis and the quantity of parenchymal tissue left in the thyroid is variable, as the pathologic involvement ranges from focal regions to an entire lobe to the entire gland. Although usually clear, the pathology needs to be differentiated from lymphoma of the thyroid.
Clinical Manifestations
Hashimoto’s thyroiditis occurs most frequently in middle-aged women but can occur at any age. The usual presentation is as an incidental finding of a goiter during routine physical examination. Although usually asymptomatic, some patients may complain of an awareness of fullness in the neck. The usual course is for slow enlargement of the thyroid over years; however, the thyroid occasionally may enlarge rapidly and can produce compressive symptoms of dyspnea or dysphagia. Rarely, Hashimoto’s thyroiditis may be painful1,14 and must be distinguished from subacute thyroiditis (discussed later). Systemic symptoms of hypothyroidism will be present in up to 20% of patients at the time of diagnosis,15 although this incidence is a bit higher with the atrophic form of the disorder. Conversely, Hashimoto’s thyroiditis is found to be the etiology in the majority of patients in the United States with hypothyroidism.
Physical examination typically reveals a firm, lobulated, nontender goiter, which is generally symmetric, often with a palpable pyramidal lobe. Regional lymph node enlargement may be observed. Although nodular thyroid disease can, and frequently does, occur in Hashimoto’s thyroiditis, single nodules and dominant nodules in a multinodular gland should be evaluated with a fine-needle aspiration biopsy to rule out a coexistent malignancy. Ophthalmopathy is present in a small subset of patients with Hashimoto’s thyroiditis.16 Further, there is evidence of chronic autoimmune thyroiditis in many patients with euthyroid Graves’ ophthalmopathy.
The hallmark of Hashimoto’s thyroiditis is elevated thyroid antibody levels. The majority of individuals with elevated thyroid antibody levels are biochemically euthyroid. Up to 10% of postmenopausal women with an elevated thyroid antibody level have an increased TSH, but a minority of these (~ 0.5%) will have overt hypothyroidism.1 Women with elevated thyroid antibody levels have been reported to develop overt hypothyroidism at a rate of 2% to 4% per year.1,15,17 Mild thyrotoxicosis (“Hashitoxicosis”) has been reported to be the initial manifestation in some patients with Hashimoto’s thyroiditis,18 especially in children. The clinical course in these patients follows a pattern similar to that observed in sporadic silent or postpartum thyroiditis (discussed later), suggesting that differentiation between these disorders may be largely semantic.
The diagnosis of Hashimoto’s thyroiditis is confirmed by the presence of antithyroid antibodies. Serum T4 and TSH concentrations depend on the level of thyroidal dysfunction that is present and are not specific to hypothyroidism caused by Hashimoto’s thyroiditis. Serum T3 concentrations are often preserved in all but the most severely hypothyroid patient and, thus, are of little clinical utility. Similarly, the radioactive iodine uptake is usually not helpful, as it may be elevated, normal, or depressed. Thyroid isotope scanning usually reveals patchy uptake and, in general, provides little useful information unless a dominant thyroid nodule is present. Ultrasound examination of the thyroid frequently reveals marked hypoechogenicity with pseudonodules.2
When imaged, an enlarged thymus gland is frequently found in Hashimoto’s thyroiditis and may be important in the pathogenesis of the condition. In both affected patients and their relatives, there is an association with other autoimmune diseases including insulin-dependent diabetes mellitus, pernicious anemia, Addison’s disease, and vitiligo. Thyroid lymphoma is rare; however, the risk is increased in those individuals with Hashimoto’s thyroiditis by a factor of 67.1,19 In patients in whom a fine-needle aspiration biopsy is performed, lymphocyte subsets should be determined on the biopsy specimen if the more typical pathologic features of Hashimoto’s thyroiditis are not present.
Clinical Management
Treatment of Hashimoto’s thyroiditis consists of thyroid hormone replacement if hypothyroidism is present. Levothyroxine is the hormone of choice for thyroid hormone replacement therapy because of its consistent potency and prolonged duration of action. The average daily adult replacement dose of levothyroxine sodium in a 68-kg person is 112 μg. Institution of therapy in healthy younger individuals can begin at full replacement doses. Because of the prolonged half-life of thyroxine (7 days), new steady-state concentrations of the hormone will not be achieved until 4 to 6 weeks after a change in dose. Thus, reevaluation with determination of serum TSH concentration need not be performed at intervals less than 6 to 8 weeks. The goal of thyroxine replacement therapy is to achieve a TSH value in the normal range, as over-replacement of thyroxine suppressing TSH values to the subnormal range may induce bone loss (especially in postmenopausal women) and cardiac dysfunction, most often atrial fibrillation.20 In noncompliant, young patients, the cumulative weekly doses of levothyroxine may be given as a single weekly dose, which is safe, effective, and well tolerated. In individuals over the age of 60, institution of therapy at a lower daily dose of levothyroxine sodium (25 μg per day) is indicated to avoid the exacerbation of underlying and undiagnosed cardiac disease. Daily doses of thyroxine may be interrupted periodically because of intercurrent medical or surgical illnesses that prohibit taking medications by mouth. A lapse of several days of hormone replacement is unlikely to have any significant metabolic consequences. However, if more prolonged interruption in oral therapy is necessary, levothyroxine may be given intravenously at a dose 25% to 50% less than the patient’s daily oral requirements.
The treatment of the euthyroid, asymptomatic patient is not so clear-cut and the recommendations for treatment of a mild increase in TSH without a corresponding low T4 concentration are divided.20,21 In addition to replacement therapy, thyroid hormone therapy may be considered in the patient with a serum TSH in the normal range in an attempt to decrease the size of a goiter or as a preventative measure to preclude the development of hypothyroidism. However, goiter regression with L-T4 is frequently not generally significant, even in the subset of patients early in the course of the disease and before fibrosis. The goal of such levothyroxine suppression therapy is to decrease the serum TSH into the subnormal range. Patients on levothyroxine suppression therapy should be reevaluated periodically and the suppressive levothyroxine dose should be reduced or discontinued if the goiter does not reduce significantly. Surgery is, of course, indicated for compressive goiters with local obstructive symptoms (see Chapter 7, Surgery of Cervical and Substernal Goiter).
Sporadic Silent and Postpartum Thyroiditis
Sporadic silent thyroiditis and postpartum thyroiditis, also known as subacute lymphocystic thyroiditis, along with subacute granulomatous thyroiditis (discussed later), are a group of disorders also known as the destruction-induced thyroididities.22–25 The clinical hallmarks of the destruction-induced thyroiditides begin with the abrupt onset of thyrotoxic symptoms, which is associated with a low-thyroid radioactive iodine uptake (Table 4-3) and elevated serum thyroid hormone and thyroglobulin concentrations, consistent with the leakage of preformed hormone from a damaged gland. Development of a nontender goiter or enlargement of a preexisting goiter almost always accompanies this. Following a brief euthyroid phase as the thyroid hormones fall back into the normal range, hypothyroidism ensues, which is a consequence of depleted thyroid hormone stores and, possibly, recent TSH suppression during the thyrotoxic phase. Because the destructive process is self-limited, however, recovery generally occurs.
Table 4-3 Causes of Low Radioactive Iodine Uptake Thyrotoxicosis
| Postpartum lymphocytic thyroiditis |
| Silent lymphocytic thyroiditis |
| Subacute granulomatous thyroiditis |
| Iodine-induced thyrotoxicosis |
| Drug-induced thyroiditis |
| Thyrotoxicosis factitia |
| Metastatic thyroid cancer |
| Struma ovarii |
Pathogenesis
Sporadic silent thyroiditis and postpartum thyroiditis are probably variants of the same disorder, distinguished only by their relationship to pregnancy. Their histopathologies are similar, as are their clinical courses and laboratory features (see Chapter 44, Surgical Pathology of the Thyroid Gland). Postpartum thyroiditis is more clearly defined than sporadic silent thyroiditis, because it is easier to study the disorder in a prospective fashion and it is far more common than sporadic silent thyroiditis. Both of these disorders are autoimmune in nature. Women who test positive for antithyroid antibodies at delivery or during the first trimester have a higher risk of developing postpartum thyroiditis.22–25 With the onset of postpartum thyroiditis itself, antithyroid antibody titers increase further. Elevated circulating levels of antithyroid antibodies have also been reported in sporadic silent thyroiditis. In addition, postpartum thyroiditis occurs more frequently in individuals with other autoimmune diseases, particularly those with type 1 diabetes mellitus.26 Finally, patients with postpartum thyroiditis are more likely to have a family history of autoimmune thyroid disorders.27
Thyroid autoimmunity in sporadic silent and postpartum thyroiditis is probably inherited and it has been proposed that “numerous genetic susceptibility factors contribute to the clinical spectrum of postpartum thyroiditis,”28 which would be consistent with the observations that postpartum thyroiditis is weakly associated with genes that regulate immune function.29 These include, in certain populations, HLA-DR4, -DR5, and -DR3 in combination with –A1 and –B8.24,29 Initial studies of an association between postpartum thyroiditis and CTLA-4 gene polymorphisms28 have been negative; however, studies in larger populations may be needed to establish an association. CTLA-4 polymorphisms are associated with Graves’ disease and autoimmune hypothyroidism.30,31 A role for fetal microchimerism has been proposed as a mechanism of immunomodulation in postpartum thyroiditis.32
Histopathology shows extensive lymphocytic infiltration, collapsed follicles, and degeneration of follicular cells23–25,28,33 (see Chapter 44, Surgical Pathology of the Thyroid Gland). The changes can be either focal or diffuse with lymphoid follicles being present in about half of the patients.33 However, unlike Hashimoto’s thyroiditis, there is usually no stromal fibrosis, oxyphilic changes, or germinal centers. Most intrathyroidal lymphocytes have the T-cell phenotype, and the distribution of T and B cells is similar in Hashimoto’s thyroiditis and sporadic silent thyroiditis.33 There is focal or diffuse chronic thyroiditis in the early stages of this disorder; during the recovery phase follicular disruption and hyperplastic changes of the follicles are common. The inflammatory changes noted earlier are seen as hypoechogenicity on ultrasound.34 The hypoechogenicity resolves with the resolution of the thyroiditis; persistence of hypoechogenicity is frequently observed in those individuals with permanent hypothyroidism.
Smoking increases the risk of developing postpartum thyroiditis, whereas age, parity, the sex or birth weight of the neonate, and the duration of breastfeeding has little influence in this regard.23–25 Adaptations of the immune system during pregnancy and their reversal after delivery may play a role in the induction of postpartum thyroiditis. During pregnancy the maternal immune response is biased toward antibody production and away from cell-mediated immunity. Normally in pregnancy the T-cell helper/suppressor ratio falls, and there are T-cell subset changes. Women who develop postpartum thyroiditis have lesser changes in their T-cell helper suppressor ratio.35 Environmental factors, such as iodine intake, may influence the incidence and severity of postpartum thyroiditis.36 Recently, a role for selenium in thyroid autoimmunity has been proposed.37 Indeed, a recent study of selenium supplementation during pregnancy and the postpartum period decreased the incidence of postpartum thyroiditis.38
Clinical Manifestations
The incidence of postpartum thyroiditis after delivery is variable, ranging from 2% to 21% of postpartum women in different studies.23–25 Sporadic silent thyroiditis accounts for less than 1% of patients with newly diagnosed thyrotoxicosis39 and is more common in women than in men.
Sporadic silent and postpartum thyroiditis are usually transient disorders consisting of four phases (Figure 4-1), although not all phases are seen in all patients.23–25 Clinical presentation is similar in both sporadic silent and postpartum thyroiditis and varies from overt thyrotoxicosis to thyroid enlargement with minimal thyroid dysfunction or hypothyroidism, depending on the stage in which the diagnosis is made. A nontender goiter is usually present. The initial phase is one of thyrotoxicosis, caused by the “leak” of thyroid hormone from the regions of lymphocytic infiltration within the thyroid gland. In postpartum thyroiditis, this occurs within a few weeks to 2 months following delivery. Tachycardia, palpitations, heat intolerance, and emotional disturbances are characteristic of the thyrotoxic phase of sporadic silent thyroiditis and postpartum thyroiditis. The ratio of serum T3 to serum T4 is lower in sporadic silent thyroiditis and postpartum thyroiditis than it is in Graves’ disease40; therefore, patients are less symptomatic. Features that develop with long-standing thyrotoxicosis, such as substantial weight loss or severe muscle weakness, are unusual.
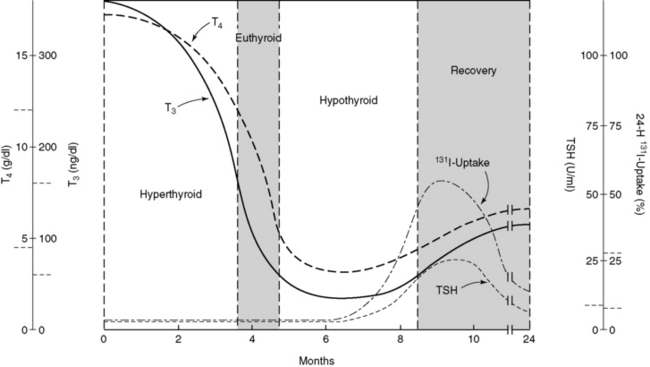
Figure 4-1 Clinical progression of destruction-induced thyroiditis.
(Modified from Woolf OD: Transient painless thyroiditis with hyperthyroidism: a variant of lymphocytic thyroiditis? [Review]. Endocr Rev 1:411, 1980.)
A brief period of euthyroidism follows the thyrotoxic phase and may last for a few months.41 As the gland becomes depleted of hormone, hypothyroidism ensues and may last for up to a year. Fatigue, malaise, impaired concentration, carelessness, and symptoms related to depression are common in patients in the hypothyroid phase. Indeed, positive tests for antithyroid antibodies during gestation are associated not only with the development of postpartum thyroiditis but also with postpartum depression.42 However, postpartum depression is not related to hypothyroidism alone, as the majority of patients with postpartum depression have normal thyroid function tests. Although severe depression may occur in a small number of women with postpartum thyroiditis, this disorder is not an important contributor to most cases of postpartum depression.
In general, the duration of hypothyroidism in those patients who initially present with thyrotoxicosis is likely to be brief. Hypothyroidism tends to be more severe and prolonged if it is the presenting disorder. Most patients do return to a euthyroid state, but up to 20% remain permanently hypothyroid,23–25 although some recent studies suggest a higher incidence of permanent hypothyroidism.43 The features associated with the development of permanent hypothyroidism are multiparity, a history of spontaneous abortion, high titers of antithyroid antibodies, the lack of a detected thyrotoxic phase, more severe degrees of hypothyroidism during postpartum thyroiditis, and persistent hypoechogenicity on ultrasound. Recurrences are common, as women with postpartum thyroiditis have a 70% risk of developing the syndrome after a subsequent pregnancy.23–25 Follow-up is essential in sporadic silent thyroiditis as well, as recurrences are not unusual and patients are likely to eventually develop permanent hypothyroidism. Further, these patients are susceptible to iodine-induced hypothyroidism years later as may be seen in other disorders of the thyroid.44,45
Although the major clinical feature that distinguishes sporadic silent or postpartum thyroiditis from subacute thyroiditis (discussed later) is the presence of thyroid pain and tenderness, thyroidal pain has occasionally been reported in sporadic silent thyroiditis. Fine-needle aspiration biopsies consistent with chronic lymphocytic thyroiditis were described in five of eight patients presenting with destruction-induced thyrotoxicosis and thyroidal pain.46 These patients also had high titers of antithyroid antibodies, and many of them developed permanent hypothyroidism. Conversely, painless destruction-induced thyrotoxicosis with pathologic changes characteristic of subacute thyroiditis has been reported in 10 of 12 patients in the Netherlands.47
Differential Diagnosis
Subacute thyroiditis, sporadic silent thyroiditis, and postpartum thyroiditis are not the only forms of thyrotoxicosis in which the radioactive iodine uptake is low (see Table 4-3).48 This is also the case for thyrotoxicosis of extra-thyroidal origin, such as struma ovarii and ingestion of supraphysiologic amounts of thyroid hormone. The most useful laboratory test in the diagnosis of thyrotoxicosis factitia is the serum thyroglobulin concentration. Serum thyroglobulin is low in patients who are ingesting extra thyroid hormone and is elevated in all other causes of thyrotoxicosis, including struma ovarii.49 Additionally, thyroid enlargement is common in destruction-induced thyrotoxicosis but unusual in thyrotoxicosis of extrathyroidal origin.
Sporadic silent thyroiditis and postpartum thyroiditis can be mistaken for Graves’ disease leading to the inappropriate use of antithyroid drugs, which are contraindicated in patients with thyrotoxicosis. Radioisotope studies are recommended in thyrotoxic patients who do not have a goiter, if they have recently been pregnant, or if they have had an abrupt onset of thyrotoxicosis. However, radioiodine studies are relatively contraindicated in the nursing mother and, if performed, require pumping and discarding breast milk for two days following the study. Measurement of the thyroid-stimulating immunoglobulin (TSI) is also helpful in differentiating between these two conditions, as it is usually elevated in Graves’ disease and negative in postpartum thyroiditis.50 It is important to measure serum TSH at the time the radioisotope test is performed because the radioactive iodine uptake may be normal or elevated as the patient recovers from the acute thyrotoxic phase. Postpartum thyroiditis is not the only autoimmune postpartum thyroid disorder. Exacerbation or relapse of Graves’ disease is prone to occur after delivery, and a few patients experience both Graves’ disease and postpartum thyroiditis.23–25 Serum thyroglobulin concentrations are elevated in both conditions, but in postpartum thyroiditis the rise in serum thyroglobulin precedes the onset of thyrotoxicosis, whereas in Graves’ disease, increases in serum thyroglobulin and thyroid hormone occur together.51 Sporadic silent thyroiditis and postpartum thyroiditis are distinguished from each other by the setting in which they occur, because postpartum thyroiditis, by definition, occurs within weeks to months after delivery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree