Chapter 3 Thyroid Physiology and Thyroid Function Testing
Introduction
The lifetime risk of developing thyroid dysfunction is common.1,2 Subclinical and overt hypothyroidism3,4 occurs in 4.6% to 9.5%, whereas subclinical and overt hyperthyroidism1,3,4 occurs in 1.3% to 2.2% of the population. Thyroid disease occurs in women two to three times more commonly than men.1,3–5 Thyroid dysfunction has a variable clinical presentation depending on the age of the patient, degree of dysfunction, concomitant disease, and duration of disease, making the clinical diagnosis difficult. Fortunately, the presence of thyroid dysfunction can be easily confirmed biochemically. Although there are many tests, including biochemical serum tests and imaging studies, the clinical picture, together with the judicious use of a limited number of the tests recommended in this chapter, can be used to diagnose most of the thyroid illnesses encountered by primary care physicians and specialists in ambulatory care. This chapter reviews the basics of thyroid testing, including imaging of the thyroid gland, and develops a straight forward approach to the diagnosis of hypothyroidism and hyperthyroidism and the laboratory tests used for monitoring the course of differentiated thyroid carcinoma.
Thyroid Physiology
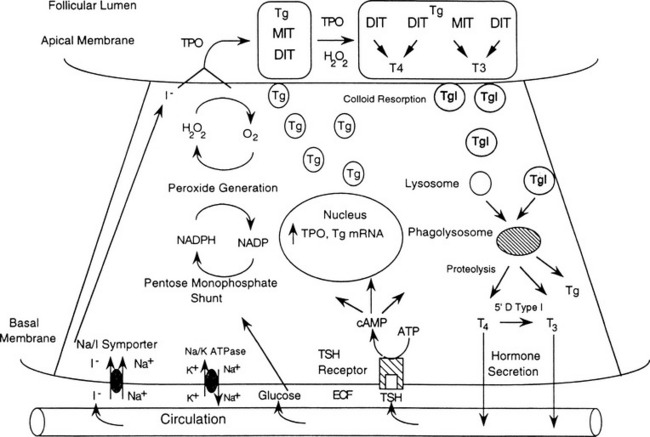
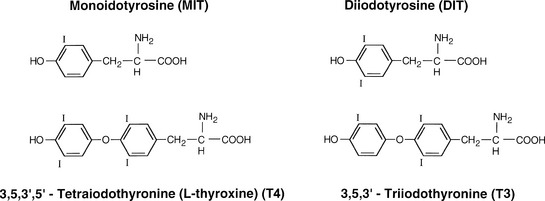
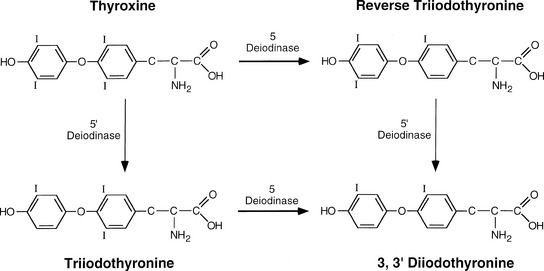
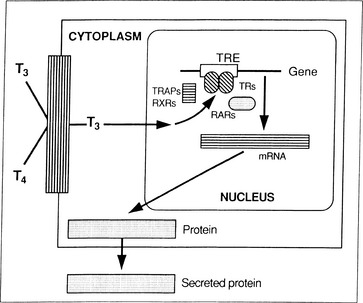
The basic functional unit of the thyroid is a follicle. The follicle is a single layer of cells that forms a sphere that surrounds a protein aggregate called colloid. Thyroid follicular cells are polarized with the side toward the colloid called the apical membrane and the outer side of the cell in contact with capillaries at the basal membrane (Figure 3-1). The synthesis of thyroid hormone is activated after binding of thyrotropin-stimulating hormone (TSH) to the basal membrane surface receptor, the TSH receptor. TSH stimulates all the steps of thyroid hormone synthesis and secretion, including iodide transport, synthesis of thyroglobulin, iodination of thyroglobulin, and secretion of thyroid hormones. TSH binding of the receptor activates adenylate cyclase to increase intracellular cAMP, which activates a cascade of numerous steps in the thyroid hormone synthetic pathway (see Figure 3-1).6 The first step is transport of iodide across the basal membrane into the follicular cell in an energy-dependent manner by the Na+/I symporter.7,8 The iodide becomes covalently attached to the precursor thyroid hormone glycoprotein, thyroglobulin at the interface between the apical membrane and the colloid by the enzyme thyroperoxidase (TPO). The iodide is attached to the tyrosine molecules in the thyroglobulin molecule to form monoiodotyrosines (MITs) and diiodotyrosines (DITs) (Figure 3-2). TPO enzymatically couples two iodotyrosines to create bioactive thyroid hormones, L-thyroxine (T4) and triiodothyronine (T3) (see Figure 3-2). The T4 and T3 remain part of the thyroglobulin molecule and is stored as colloid within the interior of the follicle. The thyroid gland is a unique endocrine organ because it stores large amounts of thyroid hormones as colloid that is released as needed through TSH stimulation. In healthy and iodine-sufficient individuals, the majority of thyroid hormone is stored as T4 with a small amount, less than 20%, stored as T3. TSH receptor stimulation leads to colloid uptake into the cytoplasm by pinocytosis to form a cytoplasmic vesicle (see Figure 3-1). The cytoplasmic vesicles fuse with lysosomes and proteases hydrolyze the peptide bonds of thyroglobulin to release T4 and T3 (see Figure 3-2) into the cytoplasm where it diffuses into the bloodstream. Approximately 90 mcg of T4 is secreted from the thyroid each day in adults. T4 and T3 travel in the circulation bound 99.97% and 99.5%, respectively, to a group of serum thyroid hormone binding proteins synthesized in the liver, which include thyroxine binding globulin (TBG), transthyretin (also known as prealbumin), and albumin. TBG has the highest affinity to bind thyroid hormone and is clinically the most important member of this group. TBG carries about 68% of the circulating T4 and 80% of the T3. Transthyretin, formally named prealbumin, binds with a lower affinity and carries 11% of the circulating T4 and 9% of T3. Albumin has the lowest affinity for thyroid hormone but the largest capacity, binding 20% of the T4 and 11% of the T3.9 More than 99% of thyroid hormones circulate bound to these carrier proteins, and are biologically inactive. The half time of T4 in the blood is 7 to 10 days. The thyroid hormones not associated with protein, free T4 and free T3, can enter the cells and are biologically active. T4 is made exclusively by the thyroid gland, whereas T3 is made primarily in peripheral tissues by deiodination of circulating T4 by a group of enzymes called deiodinases.10 Deiodinase enzyme activity is tightly regulated to maintain a normal T3 despite fluctuations in T4.11 T3 binds with a much higher affinity to the thyroid hormone receptor and is more biologically active than T4. The activity of a specific 5′-deiodinase and the resulting T3 level can be reduced by hyperthyroidism, drugs (beta-blockers, ipodate, amiodarone, dexamethasone, propylthiouracil), malnutrition, and severe illness (Figure 3-3). Conversely, during hypothyroidism, the 5′ deiodinase is activated to ensure that T4 is converted to the more bioactive T3. Normally, 20% of the daily T3 requirement is directly synthesized and secreted by the thyroid gland. During starvation and illness, the 5′ deiodinase converts the bioactive T4 and T3 to biologically inactive molecules, reverse T3 (rT3) and 3, 3′ diiodothyronine (see Figure 3-3). The small quantity of free T3 binds to its intranuclear thyroid hormone receptor to alter gene expression, which in turn alters cellular function and determines the thyroidal status (Figure 3-4). The thyroid hormone receptor (TR) is a nuclear protein that is a member of a superfamily of receptors that bind steroid hormone such as retinoic acid, vitamin D, and estrogen.
Thyroid hormone nuclear receptors (TRs) mediate the biologic activities of T3. Two TR genes, alpha and beta, encode four TR isoforms (alpha 1, beta 1, beta 2, and beta 3). The transcriptional activity of TRs is regulated by the binding of T3, the type of thyroid hormone response elements located on the promoters of the T3 regulated gene, by the developmental- and tissue-dependent expression of TR isoforms and by many nuclear co-factors or co-regulatory proteins (see Figure 3-4). There are also nongenomic actions of iodothyronine (T4) that are not mediated by intranuclear TR. Plasma membrane-initiated actions begin at a thyroid hormone receptor on integrin alphavbeta 3 that activates ERK1/2, which leads to changes in membrane ion transport, such as the Na(+)/H(+) exchanger, and are also involved in other important cellular events such as cell proliferation.12,13
Thyroid Physiology and Pregnancy
Significant changes in thyroid physiology take place during pregnancy that can make the interpretation of thyroid function tests challenging.14–16 TBG levels increase by 50% by the end of the first trimester of pregnancy, which greatly increases the protein-bound levels of T4 and T3, resulting in an apparent elevation of measured T4 and T3.16 The changes in TBG are thought to be due to the direct effect of estrogen with an increase in the liver production and glycosylation of TBG. Despite the elevation of protein-bound thyroid hormones during pregnancy, the active or free levels of T4 and T3 remain normal in euthyroid patients as reflected by a normal serum TSH level. During the first trimester, the placental human chorionic gonadotropin (hCG) increases and peaks at approximately 12 to 14 weeks of pregnancy and then decreases to a lower plateau in the second and third trimesters in the euthyroid individual.16 hCG has a weak TSH-like activity, resulting in a small increase in free T4, which usually remains in the normal range, and a concomitant decrease in TSH.14,15 Up to 13% of women during the first trimester have unmeasurable TSH levels (< 0.1 mud/L) with a normal or slightly elevated FT4I and are clinically euthyroid.16,17 This suppression of TSH usually normalizes after the first trimester of pregnancy.
Thyroid Physiology and Nonthyroidal Illness (Euthyroid Sick Syndrome)
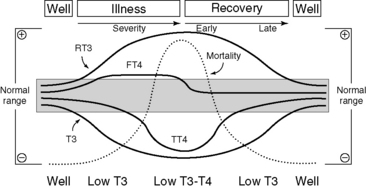
Severe nonthyroidal illness is accompanied by major alterations in thyroid physiology that can interfere with the interpretation of thyroid tests.18 The total T4 is decreased primarily because of a reduction in all thyroid binding proteins, whereas Free T4 measured by equilibrium dialysis is normal or slightly low.19 Total T3 is further decreased secondary to a decrease in the function of the 5′deiodinase, which results in a reduction in the conversion of T4 to T3 (see Figure 3-3). Instead, T4 is metabolized to the inactive form, reverse T3 (rT3), by 5′deiodinase (Figure 3-5). Measurement of rT3 does not reliably distinguish nonthyroidal illness from hypothyroidism.20 The serum TSH may be low, normal, or high in nonthyroidal illness.21,22 Serum TSH is often low secondary to medications (glucocorticoids, dopamine)23 or a form of acquired central suppression of the hypothalamus, which occurs in severe nonthyroidal illness. The degree of TSH, and especially free T3 suppression, correlates with mortality (see Figure 3-5), length of stay, and mechanical ventilation in the intensive care unit (ICU).24,25 It can be difficult to diagnose primary thyroid dysfunction in the setting of severe nonthyroidal illness. Severe nonthyroidal illness total T4, THBR, and FT4I are usually low; total T3 is relatively lower than expected for the total T4 level, but the serum TSH (measured by a third-generation assay with a sensitivity of < 0.01 mIU/L) is almost always measurable. During recovery from the acute illness with release of the central suppression, the TSH tends to rise for a short period of time prior to returning to normal.22
Thyroid Tests
TSH Assays
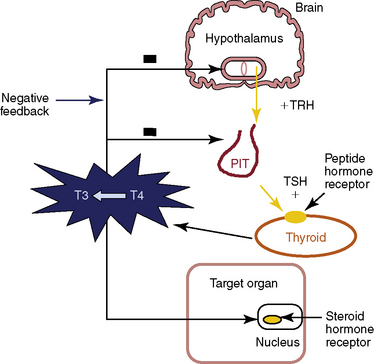
Thyroxine-stimulating hormone (TSH) is produced from the pituitary after stimulation by thyrotropin-releasing hormone (TRH), a modified three-amino acid peptide produced by neurons of the paraventricular nucleus of the hypothalamus. TRH reaches the pituitary via the portal venous system in the infundibulum (pituitary stalk). Both TRH and TSH gene expression is repressed via a negative feedback inhibition by excess thyroid hormone (Figure 3-6). Pituitary TSH is secreted in a pulsatile manner with an increase in pulse frequency at night. TSH concentrations have a diurnal rhythm with the peak occurring around 11 p.m. and lower levels during the day.26 Generally, laboratory testing of TSH during work hours, between 8 a.m. and 8 p.m., is not affected by the diurnal variation of TSH secretion, but results outside the reference range may occur in euthyroid individuals outside these times. TSH stimulates the synthesis and release of thyroid hormone and growth of the thyroid gland. TSH production from the anterior pituitary is inversely regulated by the serum thyroid hormone concentration. When thyroid hormone levels in the circulation are low, the TSH level rises to stimulate thyroid hormone production by the thyroid gland to return the system to normal function or equilibrium. The relationship or negative feedback loop between serum TSH and serum-free thyroid hormone maintains thyroid levels in a tight range (Figures 3-6 and 3-7). The TSH level changes by an inverse log-linear manner, such that small changes in the serum-free T3 levels result in large changes in the serum TSH concentration (see Figure 3-7). Small changes in a patient’s thyroid hormone level that may not be clinically apparent or result in abnormal thyroid hormone levels measured in the serum will be reflected in a significant change in the serum TSH concentration. Serum TSH is the preferred initial or screening diagnostic test for evaluation of thyroid function of the ambulatory patient, whether or not the patient is taking thyroid hormone replacement medication.27,28 For the healthy patient in an ambulatory setting, the diagnosis of thyroid hypothyroid or hyperthyroid dysfunction may be determined with approximately 98% sensitivity and 92% specificity.27 Another consideration is that TSH has a narrow within-person variability of +/− 0.5 mIU/L,29 and if there are larger changes in TSH level over time in an individual, even within the reference range, it may indicate thyroid dysfunction. In certain situations, however, such as known or suspected pituitary or hypothalamic dysfunction, recent thyrotoxicosis, critical illness, starvation, use of certain medication (dopamine or high-dose glucocorticoid therapy), interference with autoantibodies, and thyroid hormone resistance syndromes, the TSH level is inaccurate for the thyroidal status and should not be used alone to determine thyroid function.30 The presence of interfering heterophile antibodies, antibodies against the animal-derived antibodies used in the immunometric assay, may rarely cause an abnormally high or low level of reported TSH levels.31 Fortunately, these conditions can be easily determined clinically or are very rare. Further, the peripheral hormone levels will not correspond to the expected changes that occur with elevated or suppressed TSH levels.
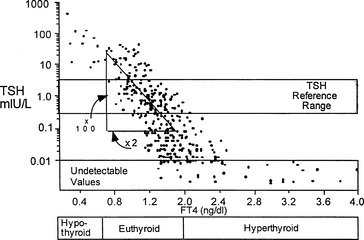
TSH assays have evolved considerably since the early 1980s. The normal range of TSH in most laboratories is approximately 0.3 to 4.5 mIU/L but depends on the specific assay used. The commonly used second-generation TSH assays should have a functional lower limit of 0.10 mIU/L or less.28,32 This TSH assay is able to differentiate between euthyroid and hyperthyroid states but does not indicate the degree of hyperthyroidism. The third-generation immunometric TSH assays utilizing a sensitive chemiluminescent detection system have a detection limit of < 0.01 mIU/ L and are capable of determining the degree of hyperthyroidism.32–34 Most clinical laboratories use a second-generation TSH assay, which is adequate for routine thyroid function testing. A third-generation TSH assay is needed only in conditions such as severe nonthyroidal illness with difficult-to-interpret thyroid function tests with a suppressed TSH.33 Spencer found that hospitalized patients with sick euthyroid syndrome or treated with high doses of glucocorticoid always had a measurable third-generation TSH > 0.01 mIU/L but the TSH was suppressed to < 0.01 mIU/L with hyperthyroidism.33
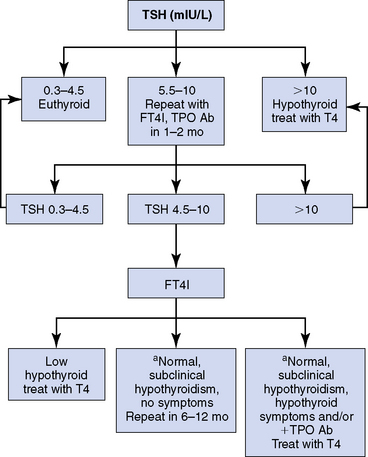
A TSH level measured in an ambulatory population that lies within the reference range is generally considered evidence of normal thyroid function and requires no additional testing34 (Figures 3-8 and 3-9). Current guidelines recommend using an upper TSH limit that is lower than the laboratory referenced range. The American Association of Clinical Endocrinology (AACE) recommends using a TSH reference range of 0.3 to 3 mIU/L,35 but the National Academy of Clinical Biochemistry (NACB) Guidelines recommend a TSH upper limit of 2.5 mIU/L.36 The Endocrine Society pregnancy guideline has recommended a trimester specific range for pregnant women of < 2.5 mIU/L during the first trimester and < 3 mIU/L in the second and third trimesters or assay-specific trimester-specific reference ranges.15 It is important to realize that these data are based on empiric and epidemiology distribution of TSH in populations, but the true upper limit is not yet definitively defined. After an abnormal TSH result has been obtained, then measurement of the circulating thyroid hormone levels should be performed (see Figures 3-8 and 3-9). The specific tests will depend on whether hypothyroidism, hyperthyroidism, or nonthyroidal illness is clinically suspected.
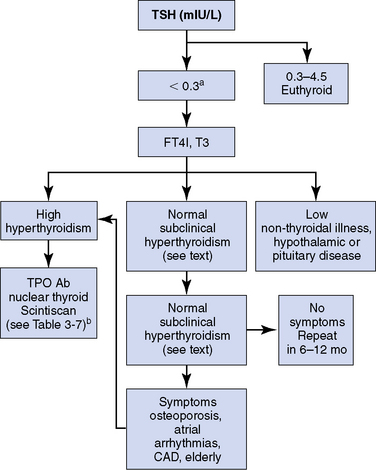
Figure 3-9 Algorithm for the evaluation of thyrotoxicosis. a, Rule out other conditions such as nonthyroidal illness. The etiology of the thyrotoxicosis can be made with TPOAb and thyroid scintiscan (see Table 3-7). b, The treatment of Graves’ disease, toxic multinodular goiter, and toxic adenoma includes thyroid surgery, antithyroid medication such as methimazole, and radioactive iodine ablation. The treatment of subacute thyroiditis is symptomatic, as the thyroid function abnormality will spontaneously return to normal in > 90% of patients. The approach to thyrotoxicosis from metastatic thyroid carcinoma is to treat the thyroid malignancy.
Measurement of Thyroid Hormone Levels
Thyroid panel is a term commonly used and often misused. The thyroid tests that make up a thyroid panel often do not include the most important diagnostic test, a TSH level. The measurement of TSH should replace any thyroid panel as the initial step in the assessment of thyroid function in the healthy ambulatory patient.27,28 However, when the TSH is not believed to be sufficient by itself for diagnosis (first-trimester pregnancy, pituitary/hypothalamic dysfunction, critical illness, high-dose glucocorticoid or dopamine therapy, etc.) or to accurately access the degree of hyperthyroidism when the TSH is low, thyroid hormone levels must also be measured.27,28,34
Total T4 and Total T3
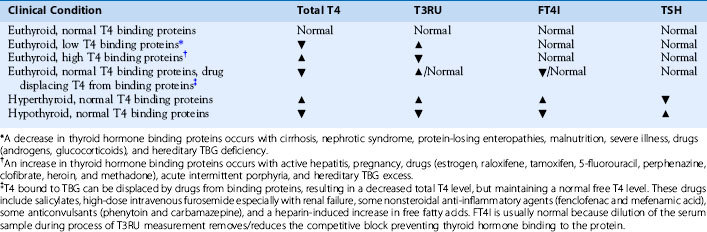
The bioactive thyroid hormones include T4 and T3. rT3, which may be elevated during nonthyroidal illness, is not biologically active, and other forms of iodothyronine are not routinely measured. Serum total T4 (TT4) and total T3 (TT3) measure both bound and free hormone levels of these two hormones. Total T4 or total T3 should never be used alone as an indication of thyroid function because many clinical conditions and medications change the amounts of thyroid hormone binding proteins or compete with the binding of thyroid hormones to the binding proteins. Changes in binding to thyroid binding proteins can greatly affect the measured TT4 and TT3 levels without altering of the bioactive free levels or thyroidal status (Table 3-1). Measurement of serum TT3 is not part of the initial evaluation of thyroid function if hypothyroidism is suspected (see Figure 3-8). During hypothyroidism, the activity of the 5′deiodinase that converts T4 to the more biologically active T3 increases, resulting in normal T3 levels until the overall thyroid levels are very low. T3 is useful in the diagnosis and management of patients with thyrotoxicosis34 (see Figure 3-9) and, occasionally, to help with differentiating Graves’ disease (higher T3/T4 ratio > 20) from subacute thyroiditis (lower T3/T4 ratio < 12).37,38 rT3 rarely needs to be measured during the evaluation of thyroid function. The mouse-antihuman antibodies (HAMAs) that interfere with TSH testing can interfere with thyroid hormone assays. HAMA may result in artificially elevated or reduced TT4, TT3, FT4, and FT3 levels.39 Patients who have received therapeutic monoclonal antibody treatments seem to be especially prone to develop interfering HAMA.40
Equilibrium Free T4 and Estimates of Free T4
Assessment of the bioactive free T4 can be estimated using the following three assays: (1) free T4 index (FT4I), (2) freeT4 by immunoassay, and (3) free T4 detected by equilibrium dialysis. Free T4 by equilibrium dialysis is the gold standard and measures the 0.03% of T4 that is biologically active and not bound to protein. This assay is available only at specialty laboratories and is technically difficult. The measured levels of free T4 have a significant interassay variation because of the minute amount of T4 being measured. Generally, local laboratories estimate free T4 by either the analog free T4 assay or a calculated FT4I corrected to thyroid hormone binding capacity. The free T4 test that results the same day or overnight in your institution or laboratory does not measure the free T4 concentration but is based on one-step, two-step, or labeled antibody approches.41 This type of free T4 test is especially sensitive to abnormal albumin levels and should not be used with conditions such as familial dysalbuminemic hyperthyroxinemia,42 pregnancy15,43 or severe nonthyroidal illness.18 For example, it is common to have a free T4 lower than the reference range in a euthyroid female who is pregnant. The high estrogen state of pregnancy greatly increases the TBG in the serum, making the FT4 inaccurate. FT4I is a calculated value that is the product of the total T4 and a correction factor related to the number of free thyroid hormone binding sites (Table 3-1). This correction factor may be called a THBR, T3 resin uptake (T3RU), or T3 uptake (T3U).34 Although the FT4I seems to be one of the most difficult laboratory tests for the nonendocrinologist to understand, these tests give important information about the T4 binding capacity of the serum that is necessary to make a diagnosis of thyroid dysfunction (see Table 3-1). The T3RU is inversely related to the free thyroid hormone binding sites. For example, a large number of free sites may occur during hypothyroidism when there is less T4 than normal to occupy the sites or when there is an excess number of free sites because of estrogen stimulation of the TBG level. Both of these situations will result in a low T3RU. The T4 binding capacity is corrected by using the equation TT4 × T3RU = FT4I. This concept is shown in Table 3-1. Note that when true thyroid dysfunction occurs, the TT4, T3RU, and FTI all move in the same direction: higher with hyperthyroidism and lower with hypothyroidism (see Table 3-1). When serum thyroid hormone binding protein levels are grossly normal, the FT4I provides a reliable index of the patient’s thyroid status. Occasionally the thyroid binding protein levels markedly change in various conditions44 (pregnancy, severe illness, malnutrition, dysproteinemia; see Table 3-1), resulting in changes in the ratio of bound to free hormone, making the FT4I a poor estimate of the free T4 level. When there is a binding abnormality such as with hepatitis or pregnancy, generally the total T4 and T3RU move in opposite directions (see Table 3-1). In these cases, TSH with a T3RU and FT4I or a measurement of the free thyroid hormone level by equilibrium dialysis is required to correctly assess thyroid function. Direct measurement of the TBG level should not routinely be ordered, because its value rarely contributes to the assessment of the patient’s thyroid status that cannot be otherwise predicted by the T3RU.
Thyroid Antibodies
In the adult, hyperthyroidism and hypothyroidism are often the result of autoimmune disease, where immunoglobulin G (IgG) antibodies are formed against thyroid proteins, such as thyroglobulin (TgAb), thyroid peroxidase (TPOAb, previously known as anti-microsomal antibodies), and the TSH receptor (TSHRAb).34 The TSHRAb is a group of immunoglobulins that can either stimulate the TSH receptor (thyroid-stimulating immunoglobulins [TSI]), causing Graves’ hyperthyroidism, or less commonly inhibit the receptor from binding TSH (thyroid hormone binding inhibiting immunoglobulins [TBII]) causing hypothyroidism. Without thyroid dysfunction, thyroid antibodies should generally not be measured except in special circumstances such as a history of hyperthyroidism during pregnancy45 or recurrent miscarriages.46 Both the stimulating and inhibiting TSHRAb can cross the placenta to affect fetal thyroid function and promote goiter. A high level of TPOAb during pregnancy will predict a higher risk for postpartum subacute thyroiditis.47 More than 90% patients with autoimmune thyroid disease (Graves’ disease and Hashimoto’s thyroiditis) will have elevated titers of second-generation assays for TPOAb and TgAb.48,49 The measurement of TPOAb can be helpful clinically, as it provides additional information regarding the autoimmune nature of the thyroid dysfunction or thyroid enlargement. It is important, however, to remember that TPOAb is diagnostic of autoimmune thyroid disease49,50 but does not guarantee the development of autoimmune hypothyroidism or hyperthyroidism. The NHANES III study has shown that 11% of the population with a normal TSH will have elevated TPOAb, but after 4 years of follow-up only 12% of these individuals will develop an elevated TSH.52 TPO antibodies can be used to help predict which patients with subclinical hypothyroidism will progress to overt hypothyroidism. When patients with subclinical hypothyroidism (i.e., TSH between 5 and 10 mIU/L) were followed for 3 years, 20.5% of the patients had spontaneous normalization of their TSH, 27.3% required replacement therapy with T4 because of progression to overt hypothyroidism or persistence of serum TSH > 10 mIU/L, and 52.1% continued to have a TSH ≤ 10 mIU/L. Patients with a positive TPOAb or ultrasound (US) changes of chronic thyroiditis had a threefold increased risk of developing overt hypothyroidism (31.2% versus 9.5%, respectively).51 Elevated titers of thyroid antibodies occur in all types of autoimmune thyroid diseases, but low titers, especially TgAb, can be measured in individuals with normal thyroid function, especially the elderly and patients with other autoimmune conditions.3 Epidemiologic studies suggest that a positive TgAb level is not highly predictive of thyroid dysfunction.3,52 As almost all patients with TPOAb will have TgAb, the measurement of TgAb adds little information to the characterization of thyroid dysfunction and should not be routinely measured.
Thyroglobulin
Thyroglobulin (Tg) is a protein precursor and storage form of thyroid hormone. This large glycoprotein is stored as colloid in the interior of each thyroid follicle. Tg continuously leaks into the circulation from the thyroid gland. Serum Tg reflects the mass of normal and malignant thyroid, TSH stimulation of thyroid tissue, and injury of the thyroid tissue. Its current primary use is as a tumor marker in patients with differentiated thyroid cancer to detect recurrent disease and evaluate the efficacy of treatment after thyroidectomy and radioactive iodine (131I).53–56 Its clinical value for evaluating thyroid function or thyroid disease (i.e., goiter) is limited in the era of modern thyroid function testing and imaging. The demonstration of suppressed serum Tg levels can be useful in differentiating factitious thyrotoxicosis (from exogenous thyroid hormone ingestion) from excessive endogenous thyroid hormone release of any etiology.57 When excess thyroid hormone is due to ingestion of levothyroxine, the normal thyroid function is suppressed and serum thyroglobulin levels become very low. But if the excess thyroid hormone is from the thyroid, thyroglobulin levels are elevated. Measurements of Tg are used primarily as a tumor marker for tumor recurrence or persistence after thyroidectomy and 131I therapy.53–56 Detection of tumor recurrence depends on the thyroglobulin immunometric assays that currently have a less than optimal sensitivity and high between-assay variability. Virtually all immunometric methods will report an undetectable Tg in euthyroid TgAb positive controls.54,58 Currently, most Tg assays have only first-generation functional sensitivity between 0.5 and 1 ng/mL but second generation Tg assays have a functional sensitivity of 0.05 to 0.1 ng/mL. The Tg assay can be made more sensitive to detect persistent or recurrent tumor after stimulated by either recombinant human TSH (rhTSH) or endogenous hypothyroidism, which results in an approximate eightfold increase in Tg. Investigators have shown at a single institution that the second-generation Tg assay correlates with the recombinant human TSH (rhTSH) stimulated Tg.53 If the basal Tg were < 0.1 ng/mL, then 99.7% of the rhTSH stimulated Tg was less than 2 ng/mL, suggesting absence of recurrent or persistent tumor.
For clinicians to know how to use the Tg level as a tumor marker for differentiated thyroid cancer, they must understand a number of important facts (see Chapter 50, Postoperative Management of Differentiated Thyroid Cancer). TgAb interference will usually make elevated Tg levels read as unmeasurable in the commercially available immunometric Tg assays. Thyroid cancer patients have a higher prevalence of TgAb than euthyroid controls (20% versus 12%).3,59,60 Commercially available TgAb assays will detect circulating antibody in only 65% of individuals with TgAb detected with a highly sensitive assay. Thus, when a node or mass is detected in a thyroid cancer patient, an unmeasurable basal or TSH-stimulated Tg with a “negative” commercial TgAb level does not exclude a thyroid cancer recurrence.58 It is reasonable in this uncommon category of patients to measure a Tg by radioimmunoassay (RIA) available at some specialty endocrine laboratories.58 The thyroglobulin antibody level may be used as surrogate marker of tumor recurrence. When TgAb declined by more than 50% within the first year following thyroidectomy, none of the patients had persistent disease, but when the TgAb increased in the same time period, 37% of the patients had persistent thyroid cancer.61 Thyroid cancer patients with a rising level of thyroglobulin antibodies are at high risk for thyroid cancer recurrence and should be evaluated promptly.61–63 The sensitivities and absolute values reported by different methods of measuring Tg and TgAb are highly variable. It is essential to always use the same Tg and TgAb method58 when following an individual over time for tumor recurrence. The presence of interfering heterophile antibodies, antibodies against the animal-derived antibodies used in the immunometric assay, may rarely cause an abnormally high or low level of reported thyrogloglobulin.63 The most common interfering antibodies are human antimouse antibodies (HAMAs). Clinically, this can be suspected when the elevated thyroglobulin level is not appropriate for the clinical situation and does not increase with TSH stimulation. When this is suspected, the clinician should repeat the test using a commercially available heterophile-blocking tube (HBT) or with an RIA for thyroglobulin.58,63,64
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree