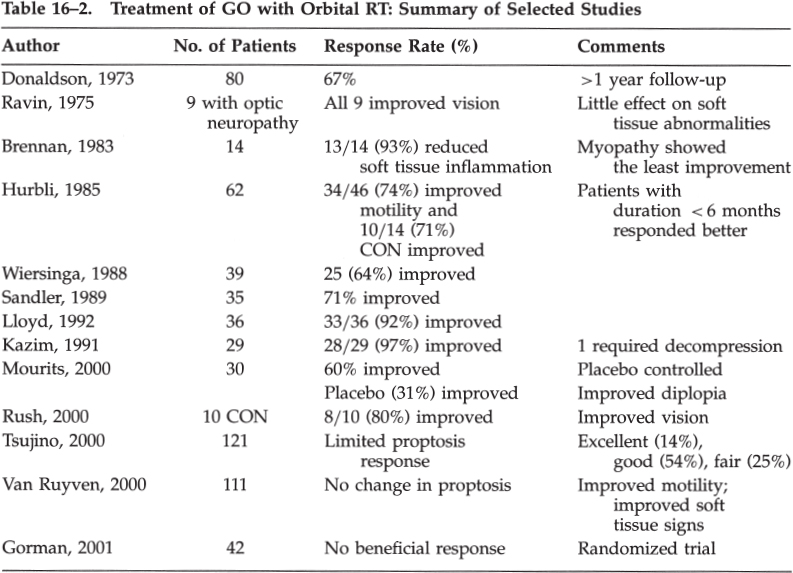
Thyroid Eye Disease: Graves’ Ophthalmopathy What Are the Typical Clinical Features of Graves’ Ophthalmopathy? Graves’ ophthalmopathy (GO) is characterized clinically by the signs listed in Table 16–1. Patients without the typical features of GO should undergo further evaluation for other etiologies of their signs: proptosis (e.g., orbital tumor or pseudotumor), strabismus (e.g., myasthenia gravis), and lid retraction (see Chapter 19). Although we do not typically image GO without compressive optic neuropathy, we do recommend neuroimaging and orbital imaging for patients with atypical features for GO (class IV, level C). What Are the Imaging Findings in Graves’ Ophthalmopathy? Orbital imaging, such as computed tomography (CT) and magnetic resonance imaging (MRI) scans, often demonstrate proptosis, extraocular muscle (EOM) enlargement sparing the tendons (Just, 1991; Ozgen, 1999), increased orbital fat volume (Chang, 1990; Char, 1991; Firbank, 2000), and sometimes engorgement of the superior ophthalmic vein. MRI may be superior to CT scan in differentiating EOM edema (with elevated T2 relaxation times) from fibrosis (Just, 1991; Muller-Forell, 1999; Nagy, 2000). Serial short tau inversion recovery (STIR) sequence MRI correlates with the clinical activity score (Mayer, 2001). Ultrasonography of the orbit can also demonstrate EOM enlargement consistent with GO. MRI, however, is usually more costly than CT imaging. Apical compression of the optic nerve in compression optic neuropathy (CON) may be seen on CT or MRI. Coronal as well as axial images are useful in the radiographic diagnosis of CON in GO (So, 2000). We recommend orbital imaging in patients with clinical evidence of an optic neuropathy and in cases where the diagnosis is uncertain or atypical features are present (class IV, level C). Eyelid signs Lid retraction (the most common clinical feature of GO) (Bartley, 1994, 1995, 1996) Stare Lid lag in downgaze Exophthalmos Enlargement of extraocular muscles Increased orbital fat volume (Hudson, 1991) Increased intraocular pressure (Danesh-Meyer, 2001; Kalmann, 1998) Diplopia/ophthalmoplegia secondary to extraocular muscle inflammation or fibrosis Visual loss Exposure keratopathy Compressive optic neuropathy (CON) from extraocular muscle involvement in the orbital apex(Liu, 1992; Mourits, 1990a,b; Nagy, 2000; Trobe, 1978) Stretching of the optic nerve due to proptosis (Liu, 1992) Flashing lights (Danks, 1998) Signs and symptoms of orbital congestion Due to proptosis with or without venous outflow obstruction Conjunctival injection and chemosis Eyelid and periorbital edema Tearing, photophobia, and orbital discomfort (Bahn, 1990; Nunery, 1991; Srivastava, 2000) Newer technologies such as 1H-magnetic resonance spectroscopy of the retrobulbar tissues have been used to estimate the concentration of chondroitin sulfate proteoglycan in retrobulbar tissue. Because the concentration of glycosaminoglycans is increased in patients with GO, this clinical tool may thus assist in the evaluation of patients with thyroid orbitopathy (Ohtsuka, 1999a). Octreotide scintigraphy may also be a useful test for determining activity of disease by demonstrating orbital uptake in GO (Gerding, 1999; Krassas, 1999a,b). What Is the Relationship between Graves’ Disease and Systemic Thyroid Status? Although GO is often associated with systemic hyperthyroidism, GO may occur in primary hypothyroidism, Hashimoto’s thyroiditis, and sometimes in euthyroid individuals (Gleeson, 1999; Salvi, 1990; Weetman, 2000). What Is the Treatment for Graves’ Ophthalmopathy? Treatment of the underlying systemic thyroid abnormalities is the logical first step in the management of thyroid disease. The evidence is controversial regarding the effect of the degree of thyroid abnormality or the speed, type (medical or surgical), or completeness of systemic therapy (Feldon, 1990) on the incidence or severity of GO (class III–IV, level C). Nevertheless, we recommend that systemic thyroid control be achieved and this may improve the signs and symptoms of GO (Prummel, 1990; Tallstedt, 1992) (class III–IV, level B). Prummel et al studied 90 patients with GO and hyperthyroidism in whom the severity of GO and thyroid function were assessed. Patients were assigned to four groups with increasingly severe GO. More dysthyroid patients were in the groups with severe GO than in the other groups (Prummel, 1990). Other uncontrolled studies, however, failed to show regression of GO after careful treatment of hyperthyroidism (class III–IV, level C). The relation between therapy for hyperthyroidism and the course of GO was studied by Bartalena et al (Bartalena, 1998a). Patients with Graves’ hyperthyroidism and slight or no GO (443 patients) were randomly assigned to receive radioactive iodine (RAI), RAI followed by a 3-month course of prednisone, or methimazole for 18 months. The patients were evaluated at intervals of 1 to 2 months for 12 months. Among the 150 patients treated with RAI, GO developed or worsened in 23 (15%) at 2 to 6 months after treatment. The change was transient in 15 patients, but it persisted in 8 (5%), who subsequently required treatment for GO. None of the 55 other patients in this group who had GO at baseline had improvement. Among the 145 patients treated with RAI and prednisone, 50 (67%) of the 75 with GO at baseline had improvement and no patient had progression. The effects of RAI on thyroid function were similar in these two groups. Among the 148 patients treated with methimazole, three (2%) who had GO at baseline improved, four (3%) had worsening of eye disease, and the remaining 141 had no change. The authors concluded that RAI therapy for Graves’ hyperthyroidism is followed by the appearance or worsening of GO more than is therapy with methimazole. Worsening of GO after RAI therapy is often transient and might be prevented by the administration of low-dose prednisone (Dietlein, 1999; Marcocci, 1999a). The authors concluded there was worsening of GO in 15% of the patients treated with RAI, but in none of those treated with RAI and prednisone. Only 3% of those treated with methimazole experienced any worsening of GO (Keltner, 1998). Bartalena et al studied 26 patients treated with RAI alone and 26 treated with RAI and systemic prednisone for 4 months (Bartalena, 1989). The initial dose of prednisone was 0.4 to 0.5 mg/kg of body weight for 1 month, with a gradual taper over 3 months. Before RAI, 15 patients had no evidence for GO and none of these developed GO after RAI. Of the patients treated with RAI alone with initial GO, 56% worsened and 44% were unchanged in soft tissue abnormalities and EOM function. Conversely, there was an improvement in GO in 52% and no change in 48% of RAI patients treated with steroids. These authors and others (Bartalena, 1989; Rasmussen, 2000) have recommended systemic corticosteroid treatment to prevent exacerbation of GO in patients undergoing RAI who have some degree of ocular involvement before treatment, but there is some disagreement with this recommendation (Beck, 1990). We recommend a short course of oral prednisone during RAI therapy for GO (class III–IV, level C). Some authors believe that patients may experience worsening of GO after any systemic thyroid treatments (e.g., thyroid surgery, RAI, and neck radiotherapy for nonthyroidal neoplasms). The presumed mechanism for worsening GO is leakage of thyroid antigens and an increase in circulating thyroid autoantibodies. In contrast to RAI, Marcocci et al did not find any effect on GO of near-total thyroidectomy in patients with nonsevere or absent GO (Marcocci, 1999b). Several studies have shown that smoking is associated with worsening GO, and we recommend discontinuing tobacco to all of our patients with GO (class III, level B) (Balazs, 1990; Bartalena, 1998b; Mann, 1999; Nunery, 1993; Pfeilschifter, 1996; Prummel, 1993; Shine, 1990; Solberg, 1998; Tallstedt, 1993; Tellez, 1992). Insulin-dependent diabetes mellitus is also a risk factor for GO, and optic neuropathy occurs much more frequently (33.3%) in patients with GO and diabetes (and seems to have a worse prognosis) than in a total group of patients with GO (3.9%) (Kalmann, 1999). The natural history of the GO is variable, and although most GO appears within a few months of the diagnosis of hyperthyroidism, it may develop many months to years before or after the onset of the systemic diagnosis of thyroid abnormality. Some patients never show clinical or laboratory evidence for systemic thyroid abnormalities (euthyroid GO). In many patients GO is a self-limited disease that may not require any therapy and the disease often stabilizes within 1 to 3 years. Therefore, treatment is usually directed at short-term control of the inflammatory component of the disease (usually within the first 6 to 36 months); acute intervention for vision-threatening proptosis or CON; and long-term reconstructive management of lid retraction, strabismus, and proptosis. Medical and other conservative therapy should generally precede consideration of surgical intervention. Shorr and Seif described a logical stepwise approach to the surgical rehabilitation of GO (Shorr, 1986). These authors proposed the following four stages for GO: (1) orbital decompression, (2) strabismus surgery, (3) lid margin repositioning surgery, and (4) blepharoplasty. The rationale for this sequential approach to GO is that orbital decompression often results in worsening, new, or changed EOM dysfunction as well as changes in lid position. Therefore, orbital decompression should precede strabismus and lid surgery in patients who require all three surgeries (class III–IV, level C). Patients with CON should undergo treatment to preserve or improve vision (class III–IV, level B). Trobe summarized the natural course of untreated CON in three series of 32 eyes. In this report, 21% of these eyes were left with a visual acuity of 20/100 or less, including a final vision of counting fingers to no light perception in five eyes (Trobe, 1978). What Therapies Are Suggested for Local Ocular and Orbital Inflammatory Signs? Patients with lid or ocular irritation, mild inflammation, or exposure keratopathy may benefit from conservative treatments, such as topical artificial tears and/or lubricating ointments, tinted or wrap-around glasses, elevation of the head of the bed, or taping the eyelids shut during sleep (Liu, 1992). Although some authors have advocated the use of topical, peribulbar, or retrobulbar steroids, we do not usually employ these routes of steroid therapy for periorbital swelling (class IV, level C). The evidence to support the use of steroids in this manner is anecdotal at best, and these treatments may be associated with complications, such as secondary increased intraocular pressure (class III–IV, level U). Should Immunosuppressive Therapy Be Considered in GO? Although systemic corticosteroids have been employed for GO, there is limited evidence outlining the specific indications for their use or the results of treatment, except in patients with CON (Liu, 1992). In addition, although medical therapy usually consists of prednisone, other immunosuppressive agents (e.g., azathioprine [Imuran], methotrexate, cyclophosphamide [Cytoxan], or cyclosporine) have also been recommended. Experience with these agents is limited (class IV, level U). Perros et al reported negative results with azathioprine for moderate GO in a matched study of 20 patients (Perros, 1990). Other studies have reported little effect with azathioprine, methotrexate (Smith, 2001), or cimexone (Kahaly, 1990). Anecdotal success has been reported with plasmapheresis and bromocriptine, but the relative efficacy of these therapies compared with traditional treatment for GO remains to be defined. Prummel et al reported a single-blind randomized clinical trial comparing the efficacy of prednisone (60 mg/day with a tapering dose) versus cyclosporine (7.5 mg/kg of body weight/day) (Prummel, 1989). During the 12-week treatment period, 11 prednisone-treated and 4 cyclosporine-treated patients responded to therapy (61% vs. 22%; p = 0.018) as defined by decrease in EOM enlargement and proptosis, improved vision, and subjective eye scores. There were no differences at baseline between the patients who responded later and those who did not, but the prednisone was less well tolerated than the cyclosporine, and combination therapy was better tolerated than prednisone alone. These authors concluded that single-drug therapy with prednisone was more effective than cyclosporine in patients with severe GO, but that the combination may be effective in patients who did not respond to either drug alone (Prummel, 1989). Corticosteroids have been shown to reduce EOM enlargement in CON (Liu, 1992). Many authors recommend relatively high doses of prednisone (1 to 1.5mg/kg/day) for the treatment of CON (Liu, 1992). Kazim et al reviewed 84 cases of acute GO treated with either high-dose systemic steroids or radiotherapy (RT) and reported that RT (1 of 29 required decompression) was more effective than corticosteroids (6 of 16 patients required decompression) and that RT had fewer complications than steroids (Kazim, 1991). Guy et al reported the use of high-dose pulse intravenous corticosteroids (Guy, 1989). Macchia et al compared oral (prednisone 60 to 80mg/d) and high-dose intravenous (two weekly injections of 1 g for 6 weeks) corticosteroid therapy for GO in 25 patients. High-dose IV steroids were better tolerated but all patients showed significant improvement in proptosis, inflammation, and diplopia (Macchia, 2001). Many authors believe that improvement in CON following prednisone therapy is usually evident within the first few weeks of treatment and that there is no justification for maintaining patients with CON on prolonged corticosteroid regimens (Trokel, 1993). Claridge et al investigated combined immunosuppression with primary bilateral orbital radiotherapy (20Gy in 10 fractions). These authors studied RT with azathioprine (up to 3mg/kg/d) and low-dose prednisolone (maximum dose 40mg/kg/d) (Claridge, 1997). Forty consecutive patients with active GO were recruited. Before treatment, 15 had CON, 35 had significant motility restriction, and 38 had marked soft tissue signs. On average, GO became inactive after 1.2 years of immunosuppression, and treatment was well tolerated. Compared with previously reported treatments, the authors thought that this therapy regimen was more effective than either treatment alone and led to fewer side effects than high-dose steroids. In particular, there was more than a fourfold reduction in the requirement for orbital decompression and strabismus surgery. Baschieri et al performed a prospective nonrandomized study of intravenous immunoglobulin (IVIG) versus corticosteroids for GO (Baschieri, 1997). Twenty-seven patients treated with IVIG were followed for an average of 21 months (12 to 48 months). Soft tissue involvement improved or disappeared in 32 of 35 patients (91%) treated with IVIG and 25 of 27 (93%) of patients treated with steroids. Diplopia improved or disappeared in 22 of 29 (76%) patients treated with IVIG and 16 of 20 (80%) of patients treated with steroids. A significant reduction of EOM thickness on CT imaging was observed after treatment in both groups. Proptosis improved or disappeared in 20 of 31 (65%) of patients treated with IVIG and in 15 of 24 (63%) of patients treated with steroids. The authors suggest that IVIG is safe and effective in reducing the eye changes in patients with GO. Prophet et al used immunoadsorption therapy (20 sessions of Plasmaselect/Therasorp Anti-IgG) in two patients with refractory GO (Prophet, 2001). Balazs et al performed a pilot study of pentoxifylline on moderately severe GO in 10 patients (Balazs, 1997). At 12 weeks, 80% of patients had improvement of soft tissue involvement, but not of proptosis or EOM involvement. This agent may have a future role in the treatment of GO. Krassas and Heufelder reviewed the immunosuppressive treatment available for GO, and concluded that cyclosporin A, azathioprine, cyclophosphamide, and cimexone had modest results but with unfavorable risk-benefit ratios (Krassas, 2001). Somatostatin analogs, octreotide, and lanreotide are emerging therapies but remain costly and as yet unproven alternatives (Krassas, 2001). Uysal et al reported a positive response to octreotide in seven of nine patients with GO (1999). We recommend immunosuppression therapy only for GO in the active phase. Usually a short course (2- to 4-week trial) of oral corticosteroids (prednisone 1 mg/kg per day) would be a reasonable first-line therapy (class IV, level C). We generally do not recommend long-term corticosteroid therapy in GO because of the systemic side effects. Concomitant corticosteroid treatment, however, may be useful as an adjunct to radiotherapy (see below). The evidence for using other immunosuppressive agents in GO is not sufficient to support a definitive recommendation on the indications, drug, dosage, or duration of therapy (class IV, level U). What Is the Role of Radiotherapy in GO? Low-dose orbital RT has been reported with good results for the treatment of acute inflammation (soft tissue signs) and/or CON due to GO. The typical cumulative RT dose of 2000 cGy is well tolerated and generally considered to be safe (class III, level B). RT may also be useful in reducing the dosage or eliminating the need for corticosteroid or other immunosuppressive treatments (Kazim, 1991; Petersen, 1990; Rush, 2000). Table 16–2 summarizes selected cases of orbital RT in the treatment of GO. Marcocci et al compared the efficacy and tolerance of intravenous or oral glucocorticoids and orbital RT in a prospective, single-blind, randomized study of 82 patients with severe GO (Marcocci, 2001). There was a significant reduction in proptosis, diplopia, and CON in both groups. IV steroids were tolerated better than oral steroids. The clinical activity score was significantly lower in the IV group than the oral group. These authors concluded that high-dose (15mg/kg for four cycles, then 7.5mg/kg for four cycles, each cycle consisted of two infusions on alternate days at 2-week intervals) IV steroids and oral steroids (prednisone 100mg/d, withdrawal after 5 months) associated with orbital RT were effective in severe GO. IV steroids, however, were more effective, had fewer side effects, and were better tolerated than oral steroids (Marcocci, 2001). Tsujino et al reported 121 patients with GO treated with orbital RT (20Gy in 10 fractions). The clinical response was excellent in 14%, good in 54%, fair in 25%, no response in 6%, and worse in 1% (Tsujino, 2000). Kahaly et al reported improvement using three RT dosing protocols: group A (1 Gy weekly for 20 weeks), group B (10 fractions of 1 Gy), and group C (2 Gy daily over 2 weeks). Improvement was noted in 12 patients in group A (67%), 13 in group B (59%), and 12 in group C (55%). They concluded that the response rates were similar in low and high RT dose treatment but that the 1 Gy per week protocol was most effective and better tolerated (Kahaly, 2000).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


