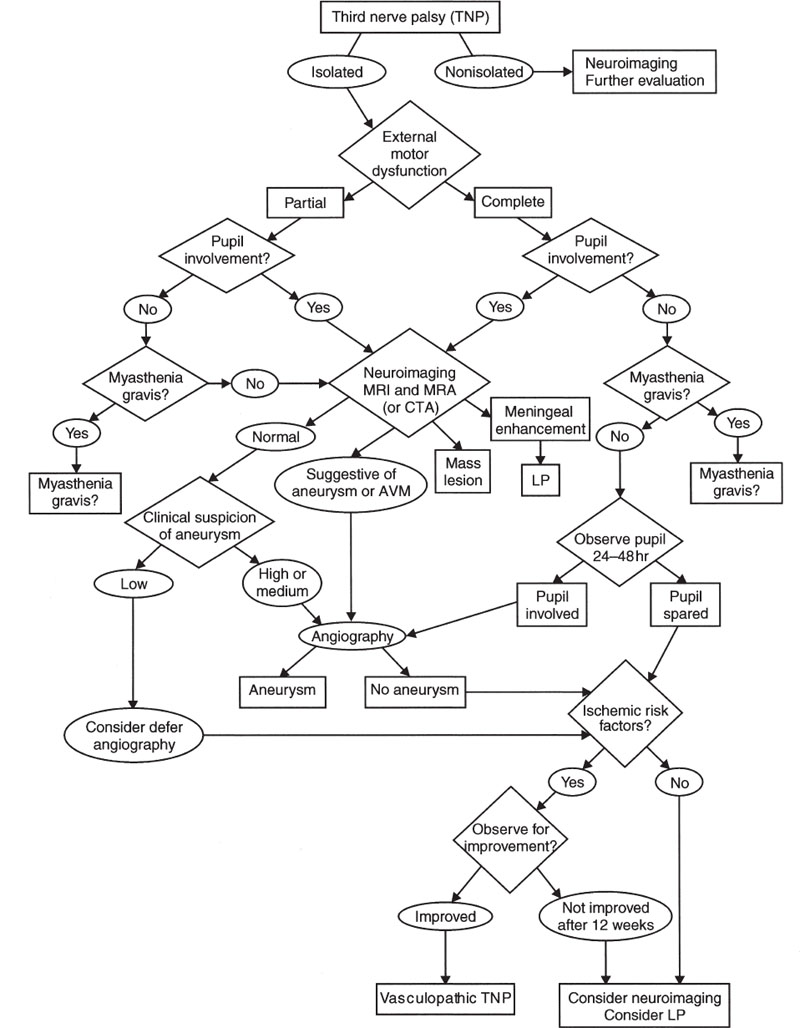
Third Nerve Palsies What Are the Clinical Features of a Third Cranial Nerve Palsy? The oculomotor nerve (third cranial nerve) supplies four extraocular muscles (medial, superior and inferior recti, and inferior oblique) as well as the levator of the lid, and contains parasympathetic fibers that supply the sphincter of the pupil and the ciliary body A complete peripheral third nerve palsy (TNP) thus causes ptosis, a fixed and dilated pupil, and a down (hypotropic) and out (exotropic) resting eye position. Partial TNPs may cause (in combination or isolation) variable ptosis; variable paresis of eye adduction, elevation, and depression; and variable pupillary involvement. In this section, we discuss the localization of TNPs associated with other neurologic signs (nonisolated TNPs) and TNPs without other associated neurologic or neuro-ophthalmologic deficits (isolated TNPs) (Lee, 1999). Is the TNP Isolated or Nonisolated? Can the TNP Be Localized? We classify TNPs as either nonisolated or isolated. The isolated TNPs were defined as TNPs without associated neurologic findings (e.g., headache, other cranial neuropathies). Patients with evidence for myasthenia gravis (e.g., variability, fatigue, Cogan’s lid twitch sign, enhancement of ptosis) are not included in the isolated TNP group. We define six types of TNP in Table 11–1. The localization of TNP is outlined in Table 11–2. Etiologies of TNPs by localization are outlined in Table 11–3. Type 1: nonisolated TNP is considered nonisolated if it has the following features: Orbital disease (e.g., chemosis, proptosis, lid swelling, injection, and positive forced ductions) Evidence to suggest myasthenia gravis (e.g., fatigability of the motility defect, Cogan’s lid twitch sign, orbicularis oculi weakness) Multiple cranial nerve palsies (including bilateral TNP) or radiculopathy Brainstem signs (e.g., hemiplegia, cerebellar signs, other cranial nerve deficits) Systemic, infectious, or inflammatory risk factors for TNP (e.g., history of previous malignancy, giant cell arteritis, collagen vascular disease) Severe headache Type 2: traumatic Isolated unilateral TNP, which has a clearly established temporal relationship to significant previous head trauma and does not progress, is considered traumatic in origin; patients with minor head trauma are not included Type 3: congenital Patient born with an isolated TNP Type 4: acquired, nontraumatic isolated Type 4A: TNP with a normal pupillary sphincter with completely palsied extraocular muscles Type 4B: TNP with normal pupillary sphincter and incomplete palsied extraocular muscles Type 4C: TNP with subnormal pupillary sphincter dysfunction and partial or complete extraocular muscle palsies Type 5: progressive or unresolved Patients with TNP that worsens after the acute stage (more than 2 weeks) or who develop new neurologic findings are considered to have progressive TNP; patients without resolution of TNP after 12 to 16 weeks are considered unresolved Type 6: signs of aberrant regeneration Is the TNP Due to a Nuclear Lesion? Lesions of the third nerve nucleus are rare and often associated with other signs of mesencephalic involvement, especially vertical gaze impairment (Bengel, 1994; Bogousslavsky, 1994; Chee, 1990; Gaymard, 1990; Nakao, 1998; Saeki, 2000b). Nuclear lesions may be due to infarction, hemorrhage, tumor, infection, or trauma and, thus, should be investigated by magnetic resonance imaging (MRI). Paresis of an isolated muscle innervated by the oculomotor nerve almost always results from a lesion in the orbit or from disease of the muscle or neuromuscular junction. For example, isolated inferior rectus paresis may develop with trauma, myasthenia gravis, or vascular disease and may also occur on a congenital or idiopathic basis (von Noorden, 1991). Lesions of the inferior rectus subnucleus, however, may also give rise to isolated weakness of the inferior rectus muscle (Chou, 1998; Lee, 2000b; Tezer, 2000). Isolated inferior rectus paresis may also occur on a supranuclear basis with a lesion selectively interrupting fibers descending from the right medial longitudinal fasciculus (MLF) to the inferior rectus subnucleus (Tezer, 2000). The levator palpebrae superioris muscles, the superior recti, and the constrictors of the pupils are affected bilaterally with nuclear lesions. Because medial rectus neurons probably lie at three different locations within the oculomotor nucleus, it is unlikely that a medial rectus paralysis (unilateral or bilateral) would be the sole manifestation of a nuclear lesion (Umapathi, 2000). Most characteristic of oculomotor nuclear involvement is unilateral TNP, weakness of the ipsilateral and contralateral superior rectus, and bilateral incomplete ptosis (Pratt, 1995). On rare occasions the ipsilateral superior rectus is spared while the contralateral superior rectus is paretic. Bilateral TNPs with sparing of the lid levators may also be caused by nuclear lesions (the central caudal levator subnucleus is spared) (Bryan, 1992). Isolated bilateral ptosis with sparing of the extraocular muscles and pupils may occur with lesions involving the levator subnucleus and sparing more rostral oculomotor subnuclei (Martin, 1996). After surgery for a fourth ventricle ependymoma, bilateral nuclear oculomotor palsies affecting only the levator and superior recti subnuclei have been described, resulting in third nerve paresis affecting only the levators and superior recti bilaterally (Sanli, 1995). Bilateral total ophthalmoplegia, bilateral complete ptosis, and large, unreactive pupils have been described with midbrain hematoma (Worthington, 1996). This constellation of findings was thought due to bilateral third nerve nuclear or fascicular damage or both, bilateral involvement of the interstitial nucleus of Cajal and the rostral nucleus of the MLF, and involvement of bilateral horizontal saccadic and smooth pursuit pathways. Lesions affecting the third nerve nucleus Oculomotor nucleus: ipsilateral complete cranial nerve (CN) III palsy; contralateral ptosis and superior rectus paresis Oculomotor subnucleus: isolated muscle palsy (e.g., inferior rectus) Isolated levator subnucleus: isolated bilateral ptosis Lesions affecting the third nerve fasciculus Isolated fascicle: partial or complete isolated CN III palsy with or without pupil involvement Paramedian mesencephalon: plus-minus syndrome (ipsilateral ptosis and contralateral eyelid retraction) Fascicle, red nucleus/cerebellar peduncle: ipsilateral CN III palsy with contralateral ataxia and tremor (Claude) Fascicle and cerebral peduncle: ipsilateral CN III palsy with contralateral hemiparesis (Weber) Fascicle and red nucleus/substantia nigra: ipsilateral CN III palsy with contralateral choreiform movements (Benedikt) Lesions affecting the third nerve in the subarachnoid space Oculomotor nerve: complete CN III palsy with or without other cranial nerve involvement; superior or inferior division palsy Lesions affecting the third nerve in the cavernous sinus Cavernous sinus lesion: painful or painless CN III palsy; with or without palsies of CN IV, VI, and VI; CN III palsy with small pupil (Horner syndrome); primary aberrant CN III regeneration Lesions affecting the third nerve in the superior orbital fissure Superior orbital fissure lesion: CN III palsy with or without palsies of CN IV, VI, and VI; often with proptosis Lesion affecting the third nerve in the orbit Oculomotor nerve: CN III palsy; superior or inferior CN III branch palsy CN III and optic nerve or other orbital structures: visual loss; proptosis; swelling of lids; Chemosis Source: Modified from Brazis, 2001, with permission from Lippincott Williams & Wilkins. Nuclear TNP Infarction or hemorrhage (Bengel, 1994; Bogousslavsky, 1994; Bryan, 1992; Chee, 1990; Gaymard, 1990; Saeki, 2000a; Tezer, 2000; Worthington, 1996) Tumor (Chou, 1998; Nakao, 1998; Sanli, 1995) Infection Trauma Multiple sclerosis (Lee, 2000b) Fascicular TNP Infarction or hemorrhage (Breen, 1991; Castro, 1990; Gaymard, 1990, 2000; Getenet, 1994; Guy, 1989a; Hopf, 1990; Kim, 1993; Ksiazek, 1994; Liu, 1991; Messe, 2001; Oishi, 1997; Onozu, 1998; Saeki, 1996, 2000a,b; Thömke, 1995; Umapathi, 2000) Tumor (Andreo, 1994; Barbas, 1995; Eggenberger, 1993; Ishikawa, 1997; Landolfi, 1998; Vetrugno, 1997) Multiple sclerosis (Newman, 1990; Onozu, 1998; Thömke, 1997) Stereotactic surgery (Borras, 1997) Subarachnoid space Aneurysms of the internal carotid–posterior communicating, superior cerebellar, basilar, or posterior cerebral arteries (Birchall, 1999; Branley, 1992; DiMario, 1992; Friedman, 2001; Giombini, 1991; Good, 1990; Greenspan, 1990; Griffiths, 1994; Horikoshi, 1999; Keane, 1996; McFadzean, 1998; Mudgil, 1999; Park-Matsumoto, 1997; Ranganadham, 1992; Renowden, 1993; Richards, 1992; Striph, 1993; Teasdale 1990; Tomsak, 1991; Tummala, 2001; Walter, 1994; Weinberg, 1996; Wolin, 1992; Zimmer, 1991) Ectatic vessels (Hashimoto, 1998b; Nakagawa, 1991; Zingale, 1993) Tumors, especially meningiomas, chordomas, metastases, or primary tumors of the third nerve (Cullom, 1993; Egan, 2001; Hardenack, 1994; Ide, 1997; Jacobson, 2001; Kadota, 1993; Kajiya, 1995; Kawasaki, 1999; Kawase, 1996; Kaye-Wilson, 1994; Kodsi, 1992; Mehta, 1990; Norman, 2001; Ogilvy, 1993; Reifenberger, 1996; Robertson, 1998; Sanchez Dalmau, 1998; Schultheiss, 1993; Takano, 1990; Winterkorn, 2001) Infectious or inflammatory processes of the meninges (e.g., sarcoidosis and Wegener’s) and carcinomatous or lymphomatous meningitis (Balm, 1996; Galetta, 1992; Guarino, 1995; Ing, 1992; Ishibashi, 1998; Jacobson, 2001; Keane, 1993; Mark, 1992; McFadzean, 1998; Newman, 1995; Renowden, 1993; Straube, 1993; Ueyama, 1997) Ophthalmoplegic migraine (O’Hara, 2001) Subarachnoid hemorrhage with leukemia (Papke, 1993) Pseudotumor cerebri Spontaneous intracranial hypotension (Ferrante, 1998) Trauma, especially during neurosurgical procedures (Balcar, 1996; Hedges, 1993; Horikoshi, 1999; Kudo, 1990; Lepore, 1995) Nerve infarction from diabetes, atherosclerosis, giant cell arteritis, or systemic lupus erythematosus (nerve infarction may also occur in the cavernous sinus or anywhere along the course of nerve) (Berlit, 1991; Bondenson, 1997; Capo, 1992; Cullom, 1995; Davies, 1994; Jacobson, 1994, 1995, 1998a, 2001; Naghmi, 1990; Renowden, 1993; Richards, 1992) Uncal herniation Hydrocephalus Cavernous sinus/superior orbital fissure Aneurysm of the internal carotid or posterior communicating artery (Hahn, 2000; Ikeda, 2001; Jacobson, 2001; Keane, 1996; Lanzino, 1993; Silva, 1999; Zingale, 1997) Dural carotid cavernous sinus fistula (Acierno, 1995; Brazis, 1994; Keane, 1996; Lee, 1996; Miyachi, 1993; Perez Sempere, 1991; Uehara, 1998; Yen, 1998) Ballon test occlusion of the cervical internal carotid artery (Lopes, 1998) Cavernous sinus thrombosis or infection (e.g., tuberculoma); superior ophthalmic vein thrombosis (Bikhazi, 1998; Grayeli, 1998; Holland, 1998; Polito, 1996) Tumors, including pituitary adenoma, meningioma, esthesioneuroblastoma, arachnoid cyst, neurinoma, nasopharyngeal carcinoma, myeloma, lymphoma, Hodgkin’s disease, and metastases (Barr, 1999; Cullom, 1993; Ing, 1997; Kasner, 1996; Keane, 1996; Kurokawa, 1992; Lee, 2000c; Liu, 1993; Manabe, 2000; Moster, 1996; North, 1993; Shen, 1993; Tao, 1992; Wake, 1993) Pituitary infarction or hemorrhage (pituitary apoplexy) (Lee, 2000c; Robinson, 1990; Rossitch, 1992; Seyer, 1992) Gammopathy Intraneural hemorrhage (Miyao, 1993) Mucocele of the sphenoid sinus (Ashwin, 2001) Sphenoid sinusitis (Chotmongkol, 1999) Tolosa-Hunt syndrome, Wegener’s granulomatosis, or other granulomatous diseases (Herman, 1999; Jacobson, 2001; Keane, 1996) Orbit Infections, inflammations, and granulomatous processes (e.g., orbital pseudotumor) (Kondoh, 1998; Ohtsuka, 1997; Stefanis, 1993) Sphenoid sinus mucocele (Sethi, 1997) Tumors (Goldberg, 1990a,b) Dural arteriovenous malformation (Gray, 1999) Trauma Unknown localization Congenital (Good, 1991; Hamed, 1991; Ing, 1992; Mudgil, 1999; Parmeggiani 1992; Patel, 1993; Pratt, 1995; Schumacher-Feero, 1999; Tsaloumas, 1997; White, 1992) Migraine (Mark, 1998; O’Halloran, 1999; Prats, 1999) Viral infections (including herpes zoster ophthalmicus or Ramsay Hunt syndrome) and immunizations (Capoferri, 1997; Chang-Godinich, 1997; Mansour, 1997; Saeki, 2000c; Sood, 1999; Zurevinsky, 1993) Lyme disease (Savas, 1997) Diffuse neuropathic processes (e.g., Fisher’s syndrome and chronic inflammatory polyradiculo-neuropathy (CIDP) (Arroyo, 1995; Nagaoka, 1996) Cervical carotid artery dissection, stenosis, or occlusion (Balcar, 1997; Hollinger, 1999; Koennecke, 1998; Mokri, 1996; Schievink, 1993) Subdural hematomas (Okuchi, 1999; Phookan, 1994) Glioblastoma multiforme (Al-Yamany, 1999) Unintentional subdural catheter (Haughton, 1999) Submucosal diathermy to the inferior turbinates to improve the nasal airway (Green, 2000) Toxic effects of drugs (Pacifici, 1993; Soysal, 1993) Cocaine (Migita, 1998) Sildenafil citrate (Viagra) (Donahue, 1998) Internal carotid cisplatin infusion (inferolateral trunk carotid artery neurovascular toxicity) (Alderson, 1996; Wu, 1997) Dental anesthesia Radiation therapy (Ebner, 1995) Partial TNP associated with elevated anti-galactocerebroside and anti-GM 1 antibodies (Go, 2000) Is the TNP Due to a Fascicular Lesion? Lesions of the third nerve fascicle often accompany nuclear lesions because infarction is a common cause of a nuclear TNP, and the paramedian branches near the top of the basilar artery often feed both structures. For example, infarction of the dorsal paramedian midbrain may cause bilateral ptosis associated with unilateral paresis of all other muscles innervated by the oculomotor nerve (pupil spared) with sparing of the contralateral superior rectus muscle (Liu, 1991). These unique findings suggest a lesion of the proximal third nerve fascicles and the central caudal subnucleus. Third nerve fascicular lesions are often caused by infarction, hemorrhage, or demyelination. Pure fascicular lesions cause a unilateral peripheral type of oculomotor palsy. Involvement of brainstem structures other than the fascicle of the third nerve identifies the mesencephalic location of the lesion (Liu, 1992). Concomitant damage of the red nucleus and superior cerebellar peduncle causes contralateral ataxia and outflow tract cerebellar tremor (Claude’s syndrome), whereas a more anterior lesion, affecting the peduncle, gives rise to oculomotor palsy with contralateral hemiparesis (Weber’s syndrome). The TNP with Weber’s syndrome may affect or spare the pupillary fibers (Saeki, 1996). Larger lesions that affect the oculomotor fascicle and the red nucleus/substantia nigra region may produce TNP with contralateral choreiform movements or tremor (Benedikt’s syndrome) (Borras, 1997), sometimes associated with contralateral hemiparesis if the cerebral peduncle is also involved. A pupil-sparing TNP associated with binocular ocular torsion to the contralateral side—thereby indicating a left-sided midbrain lesion that included the fascicle of the third nerve and the supranuclear integration centers for torsional eye movements, the interstitial nucleus of Cajal, and the rostral interstitial nucleus of the MLF—has been described with a paramedian rostral midbrain infarction in a diabetic with giant cell arteritis (Dichgans, 1995). Ipsilateral TNP and contralateral downbeat nystagmus may be caused by unilateral paramedian thalamopeduncular infarction (Oishi, 1997). Rarely, a unilateral or bilateral fascicular third nerve lesion may occur in isolation without other ocular motor or neurologic signs or symptoms (see below) (Andreo, 1994; Barbas, 1995; Getenet, 1994; Kim, 1993; Newman, 1990; Thömke, 1995). Fascicular lesions, even when bilateral, may occasionally spare the pupil(s). Bilateral preganglionic internal ophthalmoplegia has been described with bilateral partial oculomotor fascicular lesions (Hashimoto, 1998a). Because of the intraaxial topographic arrangement of fibers, fascicular lesions may cause TNP limited to specific oculomotor-innervated muscles (Ksiazek, 1994). Fascicular lesions have resulted in the following: 1. Isolated inferior oblique paresis (Castro, 1990) 2. Unilateral fixed, dilated pupil unassociated with other neurologic dysfunction (Shuaib, 1989) 3. Paresis of the superior rectus and inferior oblique without other evidence of oculomotor nerve involvement (Gauntt, 1995) 4. Paresis of the superior and medial rectus (Saeki, 2000a) 5. Paresis of the levator muscle, superior rectus, and medial rectus (Onozu, 1998) 6. Paresis of the inferior oblique, superior rectus, medial rectus, and levator muscle with sparing of the inferior rectus muscle and pupil (Naudea, 1983; Schwartz, 1995; Shuaib, 1987) 7. Paresis of the inferior oblique, superior rectus, medial rectus, levator, and inferior rectus with pupillary sparing (Breen, 1991; Naudea, 1983) 8. Paresis of the left inferior rectus, left pupil, right superior rectus, convergence, and left medial rectus (Umapathi, 2000). Based on these clinical studies, it has been proposed that individual third nerve fascicles in the ventral mesencephalon are arranged topographically from lateral to medial as follows: inferior oblique, superior rectus, medial rectus and levator palpebrae, inferior rectus, and pupillary fibers (Castro, 1990). A rostral-caudal topographic arrangement has also been suggested with pupillary fibers most superior, followed by fibers to the inferior rectus, inferior oblique, medial rectus, superior rectus, and levator, in that order (Saeki, 2000a; Schwartz, 1995). This model also accounts for the description of superior and inferior division oculomotor palsies. The superior division paresis involves the superior rectus and levator muscles without involvement of other groups (Guy, 1989a; Hriso, 1990; Ksiazek, 1989). The inferior division oculomotor palsies cause paresis of inferior rectus, inferior oblique, medial rectus, and pupillary fibers with sparing of the superior rectus and levator (Abdollah, 1990; Eggenberger, 1993; Ksiazek, 1989). Both divisional palsies may be associated with intraaxial midbrain lesions. Thus, although superior and inferior divisional TNP have classically been localized to anterior cavernous sinus or posterior orbital lesions, a divisional TNP may occur from damage at any location along the course of the oculomotor nerve, from the fascicle to the orbit (Ksiazek, 1989). Fascicular TNP may occasionally be associated with ipsilateral ptosis and contralateral eyelid retraction (plus-minus lid syndrome) (Gaymard, 1992; Vetrugno, 1997). This syndrome occurs with a small lesion located in the paramedian mesencephalon. There is involvement of the ipsilateral levator palpebrae fascicles as they emerge from the central caudal nucleus (the central caudal nucleus is spared), and the inhibitory pathways projecting on the levator palpebrae motor neurons immediately before their entrance in the central caudal nucleus. The plus-minus syndrome has been described with bilateral glioma extending to the paramedian midbrain and thalamic-mesencephalic infarction; it also may occur with peripheral processes such as peripheral TNP, myasthenia gravis, orbital myositis, congenital ptosis, or orbital trauma. Is the TNP Due to a Subarachnoid Lesion? An isolated pupil spared peripheral TNP is most often related to an ischemic neuropathy or a lesion affecting its subarachnoid portion. Subarachnoid lesions may distort or injure the brainstem, and diffuse processes will show signs of meningeal irritation. Etiologies of TNP due to a subarachnoid lesion are outlined in Table 11–3. Third nerve schwannoma may cause a painful relapsing-remitting TNP mimicking the clinical syndrome of ophthalmoplegic migraine (Kawasaki, 1999). Monocular elevator paresis from isolated superior rectus and/or inferior oblique dysfunction may occur in neurofibromatosis type 2-related schwannoma (Egan, 2001). The third nerve is also susceptible to trauma in the subarachnoid space, especially during neurosurgical procedures (Hedges, 1993; Horikoshi, 1999; Kudo, 1990). Closed head trauma may cause TNP due to shearing injury resulting in distal fascicular damage or partial root avulsion (Balcar, 1996). Walter et al described two patients with TNP precipitated by minor head trauma with negative brain computed tomography (CT) scans who were subsequently discovered to have ipsilateral posterior communicating artery aneurysms (Walter, 1994). Park-Matsumoto and Tazawa described a similar case (Park-Matsumoto, 1997). Compression of the third nerve by an aneurysm characteristically causes dilatation and unresponsiveness of the pupil. Compressive subarachnoid lesions may occasionally spare the pupil, however. Two explanations have been proposed: (1) compression may be evenly distributed and the relatively pressure-resistant, smaller-caliber pupillomotor fibers escape injury; or (2) the lesion compresses only the inferior portion of the nerve and spares the dorsally situated pupillomotor fibers. The TNP due to an aneurysm may be incomplete with at least one element of nerve dysfunction (i.e., ptosis, mydriasis, or extraocular muscle weakness) being absent. Ptosis has been described in isolation as the sole manifestation of third nerve compression by a posterior communicating artery aneurysm (Good, 1990). Rarely, aneurysmal TNP may even be transient and clear spontaneously (Greenspan, 1990). A normal pupil in the setting of a complete somatic oculomotor paresis, however, essentially excludes a diagnosis of aneurysm (see below). A single patient has been described in whom a painless, pupil-sparing but otherwise complete oculomotor paresis was the only sign of an aneurysm arising from the basilar artery (Lustbader, 1988). Conversely, an isolated pupillary paralysis without ptosis or ophthalmoparesis is rarely caused by an aneurysm or other subarachnoid lesion (Kaye-Wilson, 1994; Wilhelm, 1995). Koennecke and Seyfert reported a patient with a common carotid artery dissection from intraoperative trauma whose mydriasis preceded a complete TNP by 12 hours (Koennecke, 1998). Is the TNP Due to a Cavernous Sinus Lesion? Lesions of the third nerve in the cavernous sinus often also involve the other ocular motor nerves, the ophthalmic branch of the trigeminal nerve, and sympathetic fibers. Sensory fibers from the ophthalmic division of the fifth cranial nerve join the oculomotor nerve within the lateral wall of the cavernous sinus. The frontal-orbital pain experienced by patients with enlarging aneurysms could thus be caused by direct irritation of the third nerve (Lanzino, 1993). Compressive cavernous sinus lesions may also spare the pupil because they often preferentially involve only the superior division of the oculomotor nerve that carries no pupillomotor fibers (Silva, 1999) or the superior aspect of the nerve anterior to the point where the pupillomotor fibers descend in their course near the inferior oblique muscle. The pupillary “sparing” with anterior cavernous sinus lesions may be more apparent than real, resulting from simultaneous injury of nerve fibers to both the pupillary sphincter and dilator, causing a mid-position fixed pupil or from aberrant regeneration (see below). Ikeda et al described a patient with a painful, “severe” TNP with normal pupils due to a cavernous sinus aneurysm (Ikeda, 2001). Lesions in the neighborhood of the posterior clinoid process may for some time affect only the third nerve as it pierces the dura (e.g., breast and prostatic carcinoma) (Cullom, 1993). Medial lesions in the cavernous sinus, such as a carotid artery aneurysm, may affect only the ocular motor nerves but spare the more laterally located ophthalmic branch of the trigeminal nerve, resulting in painless ophthalmoplegia. Lesions that begin laterally present with retro-orbital pain first, and only later does ophthalmoparesis supervene. Lesions located in the cavernous sinus causing TNP are outlined in Table 11–3. The clinical findings and etiologies for processes located in the superior orbital fissure are similar to those of the cavernous sinus syndrome. Is the TNP Due to an Orbital Lesion? Lesions within the orbit that produce third nerve dysfunction usually produce other ocular motor dysfunction as well as optic neuropathy and proptosis (Goldberg, 1990a,b). Lesions may extend from the cavernous sinus to the orbital apex and vice versa so that a clear distinction between the two syndromes may be impossible. Isolated involvement of the muscles innervated by either the superior or the inferior oculomotor branch has classically been localized to an orbital process: often trauma, tumor, or infection, or a sphenocavernous lesion (Stefanis, 1993). However, as we noted, the functional division of the third nerve is present probably even at the fascicular level, and a divisional pattern may occur from damage anywhere along the course of the nerve. Superior division or inferior division third nerve paresis may occur with subarachnoid lesions (Guy, 1985), and isolated superior division paresis has been described with a superior cerebellar-posterior cerebral artery junction aneurysm that compressed and flattened the interpeduncular third nerve from below (Guy, 1989b). Superior branch palsy has also been described with basilar artery aneurysm, intracavernous carotid aneurysm, migraine, diabetes, lymphoma, sphenoidal abscess, sphenoid sinusitis, frontal sinus mucocele, viral illness, meningitis, and after craniotomy (Chotmongkol, 1992, 1999; Ehrenpries, 1995; Guy, 1989b; Manabe, 2000; O’Halloran, 1999; Saeki, 2000c; Silva, 1999; Stefanis, 1993). Even ophthalmoplegic migraine may cause recurrent paroxysmal superior division oculomotor palsy. Isolated superior division-like paresis may be mimicked by myasthenia gravis (Dehaene, 1995). Isolated inferior division involvement has occurred with trauma, mesencephalic infarction and tumor (Abdollah, 1990; Eggenberger, 1993; Ksiazek, 1989), basilar artery aneurysm (Kardon, 1991), parasellar tumors (e.g., meningioma, schwannoma) (Carlow, 1990), viral illness, orbital dural arteriovenous malformation (Gray, 1999), as part of a more generalized vasculitic or demyelinating neuropathy (Cunningham, 1994), and in association with elevated antigalactocerebroside and anti-GM1 antibodies (Go, 2000). Inferior division involvement with tumors may be pupil-sparing, perhaps because of insidious tumor growth sparing pressure-resistant pupillomotor fibers. Partial or complete TNP may rarely follow dental anesthesia, presumably due to inadvertent injection of an anesthetic agent into the inferior dental artery or superior alveolar artery with subsequent retrograde flow into the maxillary, middle meningeal, and finally the lacrimal branch of the ophthalmic artery. What Is the Evaluation of Nonisolated TNP? Nonisolated TNP should undergo neuroimaging, with attention to areas suggested topographically by the associated neurologic signs and symptoms. Appropriate investigations and neuroimaging studies are directed at the precise area of interest, and this area is determined by the associated localizing features with the TNP (Brazis, 1991; Lee, 1999). In general, MRI with and without gadolinium enhancement is the neuroimaging modality of choice for all these processes (Renowden, 1993). Contrast-enhanced CT scanning with narrow (2-mm) collimation is reserved for those patients who cannot tolerate MRI or in whom MRI is contraindicated (e.g., pacemaker, claustrophobia, metallic clips in head, etc.) (Renowden, 1993; Teasdale, 1990). CT scanning is also the appropriate first-choice neuroimaging study in patients being evaluated for acute head trauma or acute vascular events (infarction or hemorrhage). If there are clinical signs of a meningeal process, lumbar puncture should be performed. The evaluation of a patient with TNP is summarized in Figure 11–1. Is the TNP Due to Trauma? Traumatic isolated TNP (type 2) should undergo CT scanning to search for associated central nervous system damage (e.g., subdural or intracerebral hematoma) as indicated by associated neurologic signs and symptoms (Balcar, 1996; Hedges, 1993; Kudo, 1990; Lepore, 1995; Phookan, 1994). TNP after mild head trauma have been observed in association with otherwise asymptomatic lesions (e.g., cerebral aneurysm) (Park-Matsumoto, 1997; Walter, 1994). Although uncommon, neuroimaging may be warranted in patients with TNP after minimal or trivial trauma to exclude mass lesions or cerebral aneurysms (class III–IV, level C). Is the TNP Congential? Congenital isolated TNP (type 3) is rare, usually unilateral, and may occur in isolation or in association with other neurologic and systemic abnormalities, including congenital facial nerve palsies or other cranial neuropathies, facial capillary hemangioma, cerebellar hypoplasia, gaze palsy, ipsilateral nevus sebaceous of Jadassohn, mental retardation, and digital anomalies (Good, 1991; Hamed, 1991; Ing, 1992; Parmeggiani, 1992; Patel, 1993; Pratt, 1995; Shumacher-Feero, 1999; White, 1992). All patients have some degree of ptosis and ophthalmoplegia, and nearly all have pupillary involvement. In most cases, the pupil is miotic rather than dilated, probably because of aberrant third nerve regeneration, and usually trace reactive or nonreactive to light. Rarely the pupil may be spared. Amblyopia is common (Schumacher-Feero, 1999). Most cases are spontaneous, but familial cases have been described. We recommend MRI in all patients with congenital TNPs, mainly to search for associated structural abnormalities of the brain (class III–IV, level C). Is the Isolated TNP Acquired and Nontraumatic? Acquired, nontraumatic isolated TNP (type 4) may occur with lesions localized anywhere along the course of the third nerve from the fascicle to the orbit (Renowden 1993). For clinical purposes, isolated TNP may be divided into three types (types 4A–4C) (Jacobson, 1999; Lee, 1999; Trobe 1985) (Table 11–1). Figure 11–1. Evaluation of third nerve palsy (TNP). Does the Patient Have an Acquired Isolated TNP with a Normal Pupillary Sphincter with Completely Palsied Extraocular Muscles (Type 4A TNP)? TNP with a normal pupillary sphincter and completely palsied extraocular muscles is almost never due to an intracranial aneurysm. However, a single patient has been described in whom a painless, pupil-sparing, but otherwise complete TNP was the only sign of an aneurysm arising from the basilar artery (Lustbader 1988). A similar painful TNP palsy has been described with an aneurysm in the cavernous sinus (Ikeda, 2001), and pupillary sparing may rarely occur with pituitary adenoma. This type of TNP is most commonly caused by ischemia, especially associated with diabetes mellitus. In a retrospective review of 34 consecutive cases of isolated atraumatic TNP, diabetes mellitus was the most common etiology accounting for 46% of the cases (Renowden 1993). Ischemic TNP may also occur with giant cell arteritis (Berlit, 1991; Bondenson, 1997; Da vies, 1994; Renowden, 1993; Richards, 1992) and systemic lupus erythematosus. Pupil-sparing TNP has also been reported with sildenafil citrate (Viagra) (Donahue, 1998) and cocaine use (Migita, 1998). Significant risk factors for ischemic oculomotor nerve palsies include diabetes, left ventricular hypertrophy, and elevated hematocrit (Jacobson, 1994). Obesity, hypertension, and smoking are also probable risk factors. Ischemic damage to the trigeminal fibers in the oculomotor nerve may be the source of pain in ischemic-diabetic TNPs (Bortolami, 1993).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


