Therapeutic Options for Patients Who Fail Primary Chemoradiation
Steven B. Cannady
Mark K. Wax
Single institution reports, randomized controlled trials, and meta-analyses have established that chemoradiotherapy protocols improve locoregional control and reduce distant metastases in patients with head and neck squamous cell carcinoma (HNSCC) (1,2,3,4,5,6,7,8,9). These protocols have been collectively termed “organ preservation strategies” as they inherently maintain the anatomic structure being treated. Increased usage of organ preservation strategies has led to scenarios in which surgical salvage for recurrent or persistent disease requires removal of an organ from a significantly treated soft tissue and vascular bed. Studies have confirmed that operating in the previously treated field leads to a greater risk of complications resulting in added morbidity after surgery (10,11). In patients with advanced recurrent HNSCC, long-term survival is rare, and the quality of life (QOL) cost to such added morbidity and decreased function may outweigh the benefit of surgery unless adequate reconstruction is feasible (12,13). The advent of microvascular free tissue transfer has reduced the risk of complication and increased the possibility of regaining function in the posttreatment patient, rendering salvage more appealing even for short-term control of disease, amelioration of pain, or improvement of function (14,15,16,17,18,19,20,21,22,23,24).
Free tissue transfer has revolutionized the management of posttreatment sequelae encountered following nonsurgical upfront treatment of HNSCC. The functional consequences of high dose radiation therapy with concomitant chemotherapy can now be surgically addressed in the cured patient to improve QOL (14), swallowing, and deglutition (25). Microvascular techniques also allow for preservation of normal tissue outside the body during chemotherapy and radiotherapy (CRT) with reimplantation afterward as in the case of submandibular gland preservation techniques.
Within this chapter, treatment options following failed chemoradiation are discussed. The applications of microvascular free tissue transfer in the treatment of recurrent HNSCC following previous nonsurgical treatment are reviewed. The relative merits of free tissue transfer in the treatment of each subsite of HNSCC are assessed. The use of free tissue transfer in the reconstruction of treatmentrelated complications is also discussed.
Surgical salvage after failed chemoradiation is by far the most common treatment option available for these patients. Other options that that have been described include chemotherapy, reirradiation (external beam and brachytherapy), and hospice care. Each of these is briefly described.
(CHEMO) RADIOTHERAPY EFFECTS ON TISSUES OF THE HEAD AND NECK
Before we can discuss the possible treatment options for patients that have failed chemoradiation, we must understand the underlying tissue consequences of the initial treatment.
The role of chemotherapy in the treatment of head and neck oncology is well discussed in a previous chapter in this textbook. There are many agents available for treatment in the primary setting. Investigational agents have been used in Phase I and Phase II trials, and there is some question about whether, in Phase III trials, chemotherapy as a single modality treatment may be efficacious. Chemotherapy in the majority of head and neck tumors is combined with radiation therapy in a protocol known as chemoradiation. Here, the chemotherapy may be used as a sensitizing agent or as a direct combination therapy with the radiation to kill the tumor cells. Consequently, the majority of patients who present for salvage treatment, who have had chemotherapy, will have had this as a combined modality treatment with radiation therapy.
Chemotherapy has been demonstrated to not only enhance the tumor kill effectiveness of the radiation therapy but also to increase the postoperative complications in
patients who require salvage surgery. The effects of radiation therapy on the tissues have been described both from a short- and long-term toxicity perspective. Chemotherapy will adversely affect patients in the short term as well as in the long term. Patients who recur and who require salvage treatment in the close term will oftentimes be debilitated, have lost significant amounts of body weight, and still be suffering from some of the acute toxicity, such as mucositis and soft tissue necrosis. These patients will have underlying metabolic disorders and will be in a high catabolic state. Their ability to heal their wounds will be less than patients who have not undergone this treatment. Consideration of these issues should be undertaken with nutritional supplementation, and g-tube placement will need to be considered in these patients.
patients who require salvage surgery. The effects of radiation therapy on the tissues have been described both from a short- and long-term toxicity perspective. Chemotherapy will adversely affect patients in the short term as well as in the long term. Patients who recur and who require salvage treatment in the close term will oftentimes be debilitated, have lost significant amounts of body weight, and still be suffering from some of the acute toxicity, such as mucositis and soft tissue necrosis. These patients will have underlying metabolic disorders and will be in a high catabolic state. Their ability to heal their wounds will be less than patients who have not undergone this treatment. Consideration of these issues should be undertaken with nutritional supplementation, and g-tube placement will need to be considered in these patients.
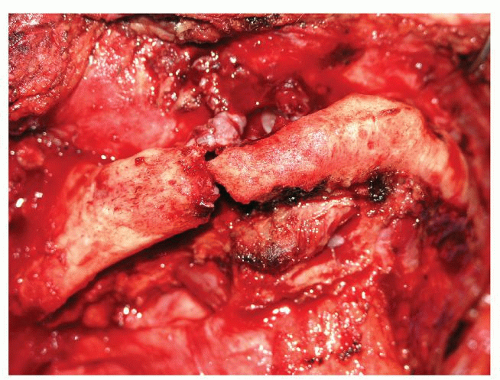
The effects on soft tissue of chemotherapy and chemoradiation will be greater than that of just radiation alone. The soft tissue fibrosis and long-term scarring will be more extensive. Combined with the patient’s inability to heal well, the patients who undergo salvage surgery following chemoradiation are more prone to develop local morbidities and complications from poor tissue and wound healing. Not only must the primary closure site of the ablated defect be considered, but the prophylactic reconstruction of the soft tissues that have been affected by the previous treatments must also be considered if one is to rehabilitate these patients and allow them to heal the arterial response to radiation that occurs most severely within the capillary endothelium and is the mechanism of late tissue manifestations of radiation therapy (XRT). The results of histologic changes to the capillaries lead to tissue anoxia and dilation of remaining capillaries (26,27). Larger arteries have different responses to XRT based on their sizes; small (less than 100 µm) to medium (100 to 500 µm) vessels suffer adventitial fibrosis, medial hyalinization, and intimal foam cell accumulation, whereas large arteries (greater than 500 µm) tend to be more resistant to these changes (28,29,30). Large arteries can suffer complications associated with neointimal proliferation, atheromatosis, thrombosis, and rupture, whereas the effect of chronic radiation on medium to large vessels is caused by injury to the vasa vasorum owing to a greater incidence of atherosclerosis within these vessels—a major concern in the use of microvascular free flaps (Fig. 112.1). This phenomenon is well documented in studies of irradiated carotid arteries, to which most head and neck reconstructive free flaps are anastamosed (31). Finally, the vascular physiology itself is altered by radiation therapy such that less blood flow occurs due to increased smooth muscle vasoconstriction and increased pressure in the capillary system secondary to tissue fibrosis of surrounding tissues (32).
Additional tissue-specific treatment effects of particular interest to oncologic surgeons include late toxicity to the skin, mucosa, salivary tissue, mandible, and laryngotracheal cartilages. It is well established that when exposed to radiation therapy, mucocutaneous tissue changes that include increases in vascular permeability, fibrin deposition, collagen formation, and ultimately, tissue fibrosis occur (33). Salivary tissue is exquisitely sensitive to XRT causing marked degeneration and loss of saliva. In a series of studies on XRT-related damage to the mandible, Marx (34) demonstrated that the triad of hypoxia, hypocellularity, and hypovascularity in posttreatment bone leads to tissue breakdown and chronic nonhealing wounds associated with osteoradionecrosis (ORN) of the jaw. It is thought that this process occurs in up to 56% of patients that undergo XRT to the head and neck, but it is unclear how many will manifest clinical symptoms; however, as many as half of those that become clinically apparent will require surgical interventions (35) (Fig. 112.2). A similar process is plausible in the cartilaginous structures of the
upper aerodigestive tract leading to chondroradionecrosis (CRN) with less frequency than mandible necrosis—5% to 12% of radiated laryngeal cancers (36). These latter two tissue effects can mimic cancer recurrence and thus often require a thorough workup for recurrence. In addition, the effects on bone and cartilage may be severe enough to warrant ablative surgery of the structure in question with reconstruction, even in the absence of recurrent disease, to improve pain and function.
upper aerodigestive tract leading to chondroradionecrosis (CRN) with less frequency than mandible necrosis—5% to 12% of radiated laryngeal cancers (36). These latter two tissue effects can mimic cancer recurrence and thus often require a thorough workup for recurrence. In addition, the effects on bone and cartilage may be severe enough to warrant ablative surgery of the structure in question with reconstruction, even in the absence of recurrent disease, to improve pain and function.
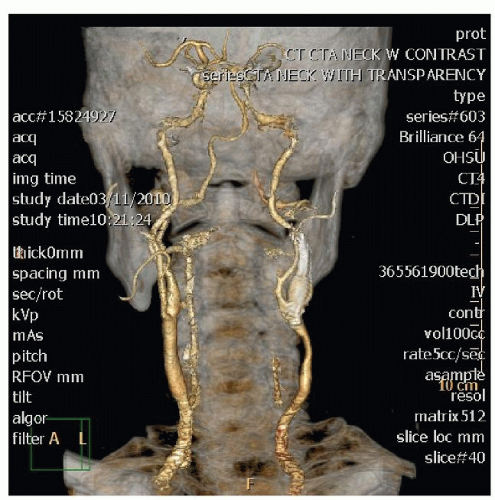 Figure 112.1 This CT angio of the carotids demonstrates severe disease of the left carotid system. There is a stent present, but the common carotid below the stent has severe atherosclerotic changes. |
RECURRENT HNSCC: EFFICACY OF TREATMENT
Surgery remains the standard of care and occasionally is the only available treatment for recurrent HNSCC that is deemed resectable by multidisciplinary care teams (37). In a large meta-analysis, Goodwin established the survival, complications, operative mortality, and treatment effect of salvage surgery for patients with recurrent HNSCC (13). In addition, his concomitant observational study collected QOL data, performance status, and financial considerations to assess the overall effectiveness of salvage on these parameters.
It was established within Goodwin’s meta-analysis that the 5-year overall average survival after salvage surgery is 39%, but a large separation was noted between early (Stage I or II) larynx recurrence (83%) versus late (Stage III or IV) larynx (37%), oral cavity (43%), and pharynx (26%) recurrences. Disease-free survival was similarly stratified denoting a clear difference in outcome for patients with early laryngeal recurrence. Surgical complications and mortality were also assessed in this analysis with a 5.2% risk of death from surgical salvage or its complications. Although the definition of major and minor complication is variable within the literature, a consistent risk in the range of 30% to 40% for salvage surgery of the primary is cited in the literature (10,11), with better outcomes in patients requiring only planned neck dissections or dissections for persistent disease (38).
In the prospective arm of Goodwin’s study, survival for 109 patients was 21.5 months. Recurrent stage was again a significant survival predictor, with decreasing survival as stage advanced, but original cancer stage was not a predictive factor. Interestingly, patients that had received prior chemotherapy had worse survival than those that did not, but no survival effect was noted for prior radiation or surgical treatments. QOL was assessed by the Functional Living Index-Cancer Questionnaire before and after surgery and demonstrated a decline with increasing stage of recurrent disease; the percentage of patients showing improvement was 64%, 65%, 41%, and 39% for Stage I, II, III, and IV, respectively. No significant difference in QOL existed for site of recurrent disease. Performance status was also assessed via the Performance Status Scale for Head and Neck Cancer Patients. Normalcy of diet varied considerably by site of recurrence and declined with advancing stage of recurrence and showed that only 47% of patients had a successful outcome after surgery. Forty-one percent (41%) had a successful outcome with respect to understandability of speech following surgery. Public eating behavior was improved in 50% of patients following surgery and again varied with recurrent stage. Operative mortality was slightly less (1.8%) in this series than in the retrospective review as were complications (20.2%). Average length of stay in the hospital following salvage was 11.5 days with an average cost of $5,700 per day in the year 2000.
This study was paramount to the understanding of outcomes for patients undergoing surgical salvage of recurrent HNSCC; however, many of these patients had undergone radiotherapy as the sole modality of treatment, and there was no analysis of free-flap surgery’s contributions to complications, QOL, or performance status. Recent studies have suggested that QOL may be maintained at a stable level (despite some performance impairments) with the use of free-flap reconstruction after head and neck ablative surgery (14). Bozec et al. employed the European Organization for Research and Treatment of Cancer’s Core Quality of Life Questionnaire (EORTC-QLQ-C30) before surgery and at 6 and 12 months in 13 patients with recurrent tumors to determine that no drop in global QOL occurred.
In addition, Kim et al. (22) studied the outcomes of patients undergoing salvage surgery with free-flap reconstruction for recurrent HNSCC. One hundred and six patients treated at the University of California at Los Angeles were reviewed. Sixty-nine patients were radiation alone failures, 14 were chemoradiation failures, and the remaining patients failed after either surgery alone or combined therapy. Major perioperative complications occurred in 34% of patients and were comprised mostly of skin necrosis (7.5%) or fistula (3.8%). Many of these patients re-recurred (74%) at an average time interval of nine months, an occurrence that was predicted by recurrent T class and persistent smoking. These results are disappointing with respect to tumor control, but remain consistent with Goodwin’s review and reflect similar rates of survival and complications to reirradiation protocols (37,39,40,41,42). No functional or QOL data are available from this study; however, based on the previously mentioned QOL study by Bozec, one can extrapolate that patients may die before reaching maximal QOL; average time to death was 9 months in the 74% of patients that re-recurred, but QOL is not maximized until between 6 and 12 months following surgery. This creates a significant treatment dilemma that requires sound clinical judgment and open communication with patients to make appropriate decisions on an individual basis.
Although some evidence exists suggesting that primary closure is superior to flap reconstruction for swallowing outcomes following ablative head and neck surgery, previous treatment limits the availability of healthy tissue for primary closure (43). As evidenced by Khariwala et al. (25), four out of five patients undergoing ablation followed by free-flap reconstruction will resume oral intake, which compares favorably to historical data using pedicled or
local tissue reconstructions. In this study, nearly half of patients had been previously irradiated, and nearly 20% had previous surgery, further adding to the impressiveness of their swallowing results. They also determined that preoperative irradiation, total glossectomy, hypopharyngeal defect, base of tongue surgery, and oropharyngeal surgery were risk factors for poor swallowing (g-tube dependence) following reconstruction. The authors recognized that in addition to subsite importance, the size of defect and reconstruction was important to include in the swallowing prognosis when counseling a patient.
local tissue reconstructions. In this study, nearly half of patients had been previously irradiated, and nearly 20% had previous surgery, further adding to the impressiveness of their swallowing results. They also determined that preoperative irradiation, total glossectomy, hypopharyngeal defect, base of tongue surgery, and oropharyngeal surgery were risk factors for poor swallowing (g-tube dependence) following reconstruction. The authors recognized that in addition to subsite importance, the size of defect and reconstruction was important to include in the swallowing prognosis when counseling a patient.
Taken as a whole, these studies suggest that salvage surgery offers a viable, albeit low likelihood, chance for longterm survival with acceptable QOL and swallowing rates. Therefore, in properly selected patients that are motivated to attempt salvage, ablation with free tissue reconstruction should be considered after in-depth counseling.
FREE TISSUE TRANSFER TO A PREVIOUSLY TREATED FIELD
Free tissue transfer is now accepted as the standard of care in the reconstruction of many head and neck defects, with high probability success rates (95%) (44). However, transferring free tissue to a previously treated head and neck site poses several challenges. Whether the initial treatment was surgery, radiation, chemoradiation, or a combination thereof, clear tissue effects may limit local reconstructive options as well as the ability to transfer free tissue (45).
Multiple studies document that decreased vascularity of skin results in relatively high rates of local skin flap necrosis for salvage surgery following XRT or CRT (Fig. 112.3) (10,11,22). The reconstructive surgeon must be prepared to deal with local wound complications resulting from skin changes and should counsel patients preoperatively of the potential for a protracted wound healing course. Some authors advocate the use of concomitant regional flaps for superficial coverage of important structures due to the expectation of some degree of skin loss in the heavily treated neck, particularly when single free-flap procedures will not be adequate to provide the volume of tissue necessary to reconstitute the internal defect and bolster marginal skin (19). Surgical planning is paramount to limiting skin flap loss, as prior incisions must be coordinated with planned salvage incisions in a way that limits devascularization of tenuous skin. Proper planning can limit minor skin flap loss as well as potentially lethal exposures of deeper neck structures such as the carotid arteries.
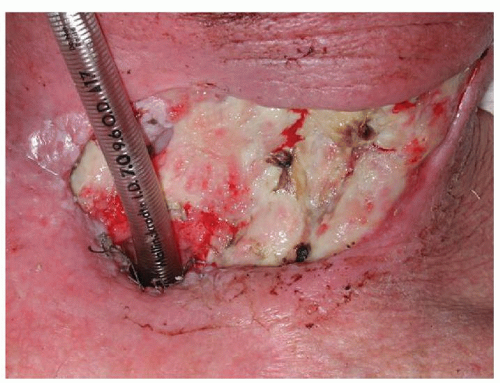 Figure 112.3 Representative example of skin flap necrosis—a common wound complication encountered in salvage surgery. |
Nonetheless, free tissue transfer has proven feasible in the previously treated patient with similar rates of flap failure (15,16,17,19,22,23,24,46). In a study by Choi et al. (16) which compared patient cohorts that had no XRT to those undergoing preoperative or postoperative XRT, no difference in rate or severity of local postoperative complications was noted, but high complication rates were cited in all groups (46% to 65%). Additionally, Cohn et al. (17) examined the use of free tissue reconstruction after twice previous chemoradiation and found that wound complication was common (66%), but flap loss was not (6%). However, of the 24 patients undergoing free-flap reconstruction, four required additional free flaps for wound healing issues, and eight required additional regional flaps for the same.
In contrast, other authors have noted increased free-flap loss in addition to expected delays in wound healing and increased infections in second flap procedures (21). The largest such cohort examined results with 50 second flaps and 12 third flaps and reported 88% and 75% success, respectively, compared with 94% in primary flap surgery (45). Whether the lower success rates are related to vessel selection, flap choice, patient physiology, or other reasons remains speculative.
The emerging sentiment from these studies seems to be that even though free-flap success rates are high, the surgeon should expect some degree of minor wound complication with the potential for more severe healing problems that require second flaps. The small cohort of patients reported by Dubsky et al. (19) lends promise to the concept of double flap reconstruction; they utilized jejunumfree tissue with pectoralis major rotational flaps in the reconstruction of recurrent hypopharyngeal tumors and suffered no flap losses or wound complications requiring further surgical therapy.
The selection of recipient vessels remains a challenge in the previously treated patient; some authors report a 50% rate of contralateral vessel use when previous neck surgery had been performed, necessitating long vascular pedicles in reconstruction (47). In addition, the surgeon must be prepared to utilize out of field recipient vessels if the health of in-field vessels is suboptimal. Particularly in the previously irradiated and surgically treated neck, the internal mammary artery and transverse cervical arteries and veins should be held in reserve, and saphenous vein grafts may be necessary (17) (Fig. 112.4).
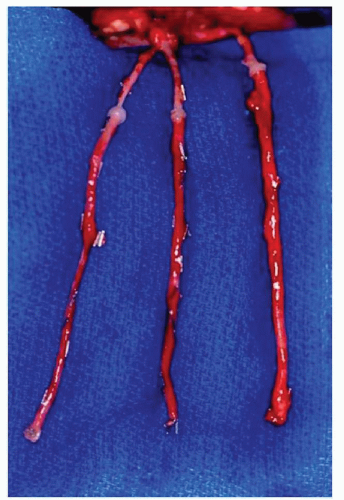 Figure 112.4 A RFFF is demonstrated here. The vascular pedicle has been lengthened by the addition of saphenous vein grafts to the radial artery and the two venae comitantes. |
Few papers exist specifically addressing the use of free flaps in the salvage setting. Below is a summary of pertinent literature on the use of free flaps in previously treated patients. Some reference is made to larger series that include treated and treatment naïve patients in the interest of completeness.
Nasopharynx/Skull Base/Paranasal Sinuses
Intergroup Study 0099, a phase III randomized controlled study comparing chemoradiotherapy to XRT alone for nasopharyngeal carcinoma (NPC), established chemoradiation as the primary treatment of NPC (48). Recurrent nasopharyngeal HNSCC occurs in approximately 30% of patients; treatment options include reirradiation with either external beam or brachytherapy or nasopharyngectomy (49). Survival following reirradiation ranges between 16% and 45% with 5-year local control of 50% to 60%. Reirradiation of locally recurrent NPC has been well described in the literature. The inaccessibility and the reluctance of many centers to undertake nasopharyngectomy have led to a large experience in the management of local failure. The patients with local recurrence do well with nasopharyngeal retreatment. Mortality rates are less than 5%. Patients who have limited volume or current disease may be treated with break therapy and also have excellent local control rates.
While local control rates with reirradiation can be achieved, the patient population often is preselected. Patients will have significant morbidity secondary to the tumor as well as from the treatment. The definitive answer to the role of reirradiation awaits being answered by a multiinstitutional controlled study. Nasopharyngectomy results in survival between 4% and 67% with local control from 43% to 67% (50




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree