(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
The ocular circulation is supplied by two sets of arteries. The central retinal artery is the main source of supply to the inner retina. The posterior ciliary artery (PCA) is the main source of blood supply to the optic nerve head (ONH), the choroid up to the equator, the retinal pigment epithelium (RPE), the outer 130 μ of the retina (and, when a cilioretinal artery is present, the entire thickness of the retina in that region), and the medial and lateral segments of the ciliary body and iris (Fig. 4.1). That makes the PCA circulation the most important component of the ocular and ONH circulation.
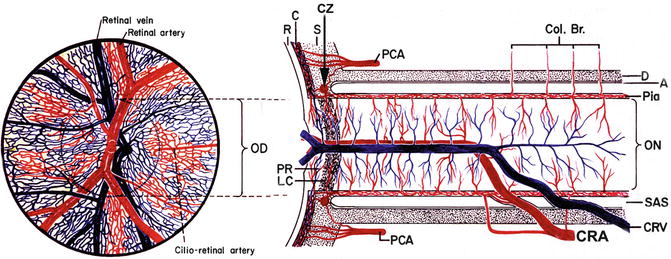

Fig. 4.1
Schematic representation of blood supply of the optic nerve (modified from Hayreh [1]). Abbreviations used: A arachnoid, C choroid, CRA central retinal artery, Col. Br. collateral branches, CRV central retinal vein, CZ circle of Haller and Zinn, D dura, LC lamina cribrosa, OD optic disc, ON optic nerve, PCA posterior ciliary artery, PR prelaminar region, R retina, S sclera, SAS subarachnoid space
Ever since Frederick Ruysch [2] first described the vascular pattern of the PCAs and their branches in about 1700 AD, the information has been drawn entirely from postmortem cast studies. Since the PCAs mainly supply the choroid, extensive anatomical studies of the choroidal vascular bed have been conducted, mostly by studying casts prepared by the postmortem injection of a variety of materials [3–9] and also by studying casts by scanning electron microscopy [10–13]. These studies formed the basis of the classical anatomical description of the choroidal vasculature. According to most of these descriptions, (1) PCAs have no segmental distribution, (2) they anastomose freely with one another as well as with the anterior ciliary arteries, (3) there are interarterial and arteriovenous anastomoses in the choroid, and (4) the choriocapillaris forms a freely communicating and an uninterrupted vascular bed in the entire choroid [9, 12]. From these anatomical descriptions, it was generally concluded that occlusion of the PCA or one of its branches should not produce an ischemic lesion. However, clinically, inflammatory, ischemic, metastatic, and degenerative choroidal lesions are usually localized. This puzzling discrepancy was well summarized by Duke-Elder and Wybar [14]: “The tendency for inflammatory and degenerative diseases of the choroid to show a considerable degree of selective localization, despite the fact that anatomically the vessels would appear to form a continuous network, has given rise to speculations regarding the anatomical isolation of specific choroidal areas.” A number of clinical and experimental observations reported in the literature [15] suggest that the PCAs have a segmental supply to the choroid. Wybar [8, 9], Ring and Fujino [4], and Uyama et al. [13] even from their cast studies concluded that these arteries were segmentally arranged and that each branch supplied a localized zone of the choroid.
Olver [16], in her scanning electron microscopic examination of methyl methacrylate microvascular casts, described the distal and paraoptic short posterior ciliary arteries as supplying wedge-shaped areas of the choroid. According to her, it is well recognized that the choriocapillaris shows regional variations and the lobules are densely packed at the posterior pole with a high capillary to intercapillary ratio. Ducournau [17, 18] in a study of 24 human eyes found that (1) the PCAs and their branches were arranged into two independent groups: paraoptic and distal PCA, (2) there was no specific macular short PCA, and (3) he observed no anastomoses between the short PCAs. In view of these discrepancies between the postmortem cast studies and the lesions seen clinically, I started to research the subject in vivo experimentally in rhesus monkeys, as well as clinically, in 1967. The advent of fluorescein angiography then enabled us to study not only the blood flow but also the physiological anatomy in vivo. I used fluorescein fundus angiography as the main research technique. These in vivo studies demonstrated, as shown below, that the PCAs have a segmental distribution and consequently are an endarterial vascular system. This has helped to explain the nature of the underlying disease process in many previously obscure fundus and ONH lesions. The description of the in vivo PCA circulation and watershed zones given here is essentially based on my studies [15, 19–46].
Anatomy of the Posterior Ciliary Arteries (PCAs)
There is much confusion about the nomenclature, number, origin, and distribution of the PCAs in man. This is evident from the information in Table 4.1 [47–64], before my studies published in 1962. Many authors designated them the “long” posterior ciliary arteries right from their point of origin. I investigated this in 59 human specimens and have described the subject in detail elsewhere [65]. The following description is based on my studies.
Table 4.1
Number of PCAs seen in a specimen (Various authors)
Author | Date | Number of PCAs |
|---|---|---|
von Haller [47] | 1781 | 2 in one, 3 in three, 4 in six, and 6 in three cases |
Murray [48] | 1794 | 3 |
Hildebrandt and Weber [49] | 1831 | 3 |
von Sömmering et al. [50] | 1841 | 3 trunks; often 2 (posterior outer and anterior inner) |
Arnold [51] | 1847 | 2 (posterior and anterior); also several more |
Meyer [52] | 1887 | 2 (inner and outer) in ten, 3 in eight, 4 in two |
Quain [53] | 1892 | 2 (inner and outer); often 1 or 2 additional |
Whitnall [54] | 1932 | Variable, 2 trunks or 6–8 vessels |
Duke-Elder [55] | 1932 | 2 trunks or 6–8 vessels |
Sudakevitch [56] | 1947 | 2 (medial and lateral), rarely 3–5 |
Wood Jones [57] | 1949 | 2 (lateral and medial) |
Brash [58] | 1951 | 6–8 |
Schaeffer and Morris [59] | 1953 | Short 5 or 6 |
Wolff [60] | 1954 | 2 |
Hollinshead [61] | 1954 | Long set 2, sometimes multiple |
Short set multiple | ||
Bedrossian [62] | 1958 | 1 or 2 |
Johnston and Whillis [63] | 1958 | Long 2, short 7 |
Gillilan [64] | 1961 | Variable |
Number of Main PCAs
My study showed that the ophthalmic artery in man gives out one (in 3 %), two (in 48 %), three (in 39 %), four (in 8 %), or five (in 2 %) PCAs. When there are more than three PCAs, the additional arteries are usually small in size.
Nomenclature
I have designated the PCAs according to their relation to the optic nerve near their site of entry into the eyeball (Fig. 4.2). They are called:
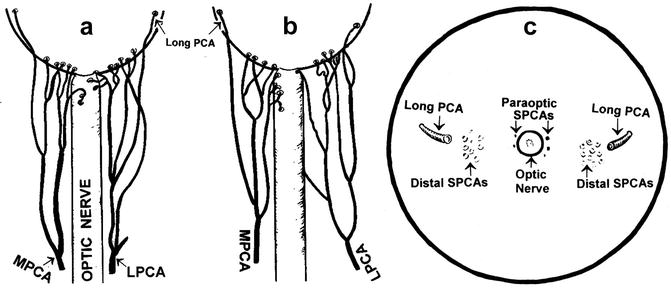

Fig. 4.2
Diagrammatic representation of actual branching pattern of medial (MPCA) and lateral (LPCA) PCAs in 2 eyes (a, b) and in (c) the site of entry of the various long and short PCAs as seen on the back of the eyeball (Reproduced from Hayreh [46])
(a)
Medial PCA: This lies medial to the optic nerve and may be one (in about 70 %) or two (in about 30 %) in number.
(b)
Lateral PCA: This lies lateral to the optic nerve, and there may be one (in about 75 %), two (in about 20 %), or none (in about 3 %).
(c)
Superior PCA: This is seen in only 9 % and may be one (in 7 %) or two (in 2 %); these are usually small in size.
Site of Origin
According to some authors, the PCAs arise from the first part of the ophthalmic artery [53, 56, 57, 60], or the second part superior to the nerve [54, 59, 62], or medial to the nerve [55, 59]; others said that they arose on either side of the optic nerve. von Sömmerring [50] mentioned that the lateral PCA arose earlier than the lacrimal, while the medial PCA arose above the optic nerve. Meyer [52] described the medial PCA and the central retinal artery arising together first, followed by the lateral PCA. In 11 of his 17 specimens, in which the ophthalmic artery crossed over the optic nerve, the medial PCA with the central retinal artery arose before the lateral PCA; in the remaining 6 the lateral PCA (alone or with other branches such as the central retinal artery or muscular arteries) arose before the medial PCA. In the 3 specimens in whom the ophthalmic artery crossed under the optic nerve, the lateral PCA always arose first, followed by the central retinal artery and then the medial PCA in common with the muscular arteries.
In my series [65], however, the medial and lateral PCAs originated from the first part in 25 specimens, and from the angle in 39, and 48 medial and 37 lateral PCAs arose distal to the angle (Fig. 4.3). Where the origin was distal to the angle, the PCAs arose not only from the second part but even from the bend and the initial portion of the third part, especially the medial PCA. In nine specimens, all the PCAs arose distal to the angle, and in all these the ophthalmic artery crossed over the optic nerve. In the remaining specimens, one or both of the medial and lateral PCAs arose before the angle. There was no relationship of any kind between the number of PCAs, which might be present in a specimen, and their site of origin. Those in which one of the PCAs was situated distal to the angle were more numerous than those in which they arose before the angle and there were no special accessory branches and anastomoses in such cases. All these points contradict the views expressed by Sudakevitch [5]. PCAs may arise in common with the central retinal artery (see Chap. 2) – this has clinical importance (see Chap. 13). The PCAs and their branches are usually markedly tortuous.
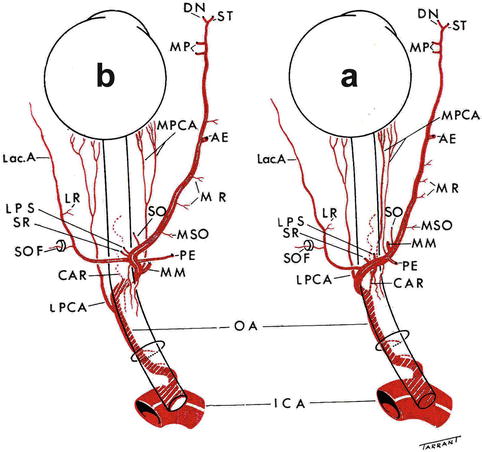

Fig. 4.3
The pattern of the branches of the ophthalmic artery when it crosses (a) over and (b) under the optic nerve (Reproduced from Hayreh SS [66]). Abbreviations used: AE anterior ethmoid artery, CAR central artery of retina, DN dorsal nasal artery, ICA internal carotid artery, Lac. A lacrimal artery, LPS muscular artery to levator palpebral superior, LPCA lateral posterior ciliary artery, LPS muscular artery to levator palpebrae superioris, LR muscular artery to lateral rectus, MM medial muscular artery, MP medial palpebral artery, MPCA medial posterior ciliary artery, MR muscular artery to medial rectus, MSO muscular artery to superior oblique, OA ophthalmic artery, PE posterior ethmoid artery, SO supraorbital artery, SOF superior orbital fissure, SR muscular artery to superior rectus, ST supratrochlear artery
Branches of the PCAs
The various PCAs run forward, divide into a large number of small branches and pierce the sclera near the optic nerve (Fig. 4.2). At that point, they are of two types:
Short PCAs (SPCAs)
They may be 10–20 in number, depending upon the intraorbital subdivisions of the PCAs before it reaches the sclera [65, 67, 68]. The SPCAs are also of two types (Fig. 4.2) [15, 17, 67, 68]:
(a)
(b)
Distal SPCAs: These constitute the majority of the SPCAs, and enter the sclera a short distance away from the optic nerve on the medial and lateral sides and run radially toward the equator. The temporal distal SPCAs pierce the sclera to enter the eyeball in the macular region (see below in section “Submacular choroid”).
Long PCAs
These are two in number – one medial and one lateral (Fig. 4.2). The long and short PCAs have frequently been confused with the main PCAs – they are in fact the branches of the main PCAs and do not arise directly from the ophthalmic artery. It is essential to have a clear understanding of the various types of PCAs to avoid confusion. It cannot be stressed too strongly that unless this terminology is clearly understood and adhered to, confusion will follow. The long PCAs enter the eyeball in the horizontal plane on the medial and lateral sides, some distance away from the distal SPCAs, and run radially in the horizontal meridian forward to the iris [29, 65]. Siam et al. [69], in a study of 12 cadaver eyes, found that the lateral long PCA enters the sclera 3 mm temporal to the optic nerve sheath and runs intrasclerally, being visible for 4 mm. Its lateral end is just overlapped by the nasal end of the fleshy inferior oblique insertion.
Distribution of the PCAs
1.
The short PCAs supply the following (Fig. 4.1):
(a)
Choroid: as far as the equator.
(b)
Retina: The choroid supplies the overlying outer 130 μ of the retina, including the RPE. If a cilioretinal artery is present, then the entire thickness of the retina is supplied in the distribution of the cilioretinal artery.
(c)
Branches from these form the circle of Haller and Zinn in the peripapillary sclera and those supply the ONH.
2.
The long PCAs supply medial and lateral segments of the ciliary body and iris, and a small sector of the choroid posterior to the equator on the nasal and temporal sides.
In Vivo Circulation and Distribution Pattern of the PCAs and Their Branches
As mentioned above, since the first description by Frederick Ruysch [2] in about 1700 AD, knowledge of the vascular pattern of the PCAs and their branches was drawn entirely from postmortem cast studies. However, fluorescein fundus angiographic studies in man and rhesus monkey, both in health and disease, have clearly shown that the choroidal blood flow, contrary to the information from the casts, is segmental in nature, as is evident from the following:
Areas Supplied by the Main PCAs
Experimentally in rhesus monkeys, I cut one or another or all the PCAs in 85 eyes and evaluated the area of their distribution in the choroid and ONH and anastomoses, by fluorescein fundus angiography (Figs. 4.4 and 4.5) [15, 23, 24, 37]. PCA occlusion caused by thrombosis is well documented in patients with giant cell arteritis, by various histopathological studies [33, 35, 70–75]. That provided a model of PCA occlusion in man, to evaluate the in vivo distribution and anastomoses of the PCA in the choroid and ONH by fluorescein fundus angiography [30, 31, 42, 76–78]. These experimental and clinical studies showed that each PCA has a segmental distribution in the choroid and ONH, and they do not anastomose either with the adjacent PCAs or with the anterior ciliary arteries [15, 23, 24, 30, 31, 37, 42, 67, 68, 76–79]. Similarly when I produced anterior ciliary artery occlusion by recession and resection of the various recti in rhesus monkeys as well as in patients with strabismus, the PCAs did not fill the area of the anterior uvea supplied by the occluded anterior ciliary artery [34, 45].
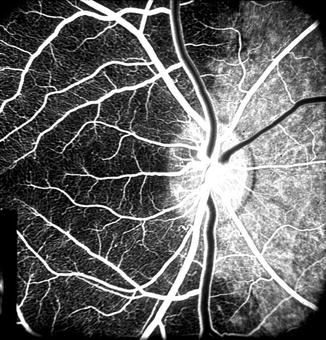
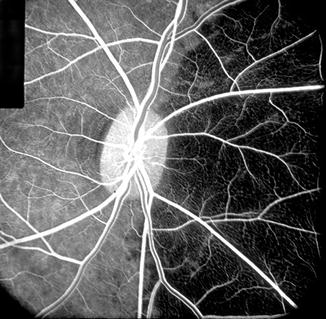

Fig. 4.4
Fluorescein fundus angiogram during retinal arteriovenous phase, of the right eye of rhesus monkey after cutting the lateral PCA. It shows no filling of the temporal choroid but normal filing of the medial choroid (Reproduced from Hayreh and Bains [15])

Fig. 4.5
Fluorescein fundus angiogram during retinal arteriovenous phase, of the right eye of rhesus monkey after cutting the medial PCA. It shows no filling of the nasal choroid but normal filing of the temporal choroid (Reproduced from Hayreh and Bains [15])
The PCAs not only supply the choroid but also play an important role in the blood supply of the ONH [19, 32, 35, 42, 67, 68, 77, 79]. These studies showed that the lateral and medial PCAs supply the corresponding parts of the choroid [15, 20, 22, 30–33, 35, 37, 39, 42, 46]. There is marked interindividual variation in the area supplied by the two PCAs in man [30–32, 35, 39, 42, 46]. The medial PCA may supply the entire nasal choroid up to the level of the fovea, including the ONH (Figs. 4.6 and 4.7), or its supply may stop short nasal to the nasal peripapillary choroid (Fig. 4.8), so that it may take no part in the blood supply of the ONH, or there may be any variation between these two extremes (Fig. 4.9). The lateral PCA supplies the area of the choroid not supplied by the medial PCA or vice versa (Figs. 4.4, 4.5, 4.6, 4.7, 4.8, and 4.9). When there are more than one medial or lateral PCAs, the area supplied by each of them may be one quadrant or only a sector (Figs. 4.10a, 4.11, 4.12, and 4.13). When the superior PCA is present, it would accordingly supply a superior sector (Fig. 4.14). Given the marked interindividual variation in number reported in the literature (Table 4.1) and in my study (see above), and distribution by the various PCAs, there can be an extremely variable pattern of distribution by them in the choroid and ONH. My clinical and experimental studies have shown that the various PCAs do not anastomose with one another, and each behaves like an end artery [15, 20, 22, 30–33, 35, 37, 39, 42, 46].
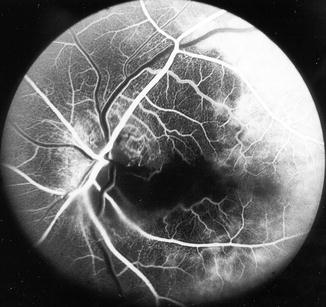
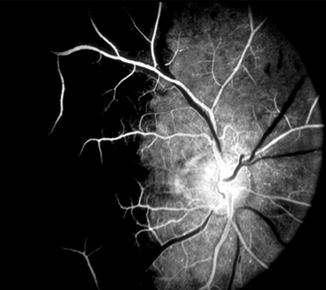
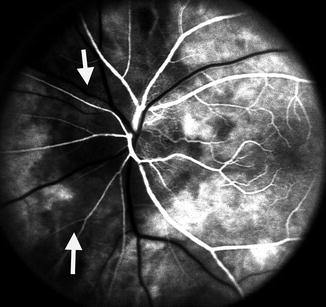
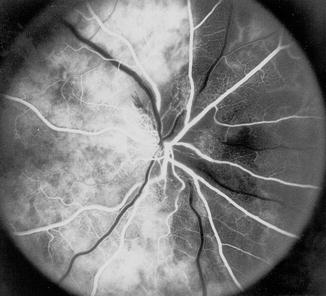
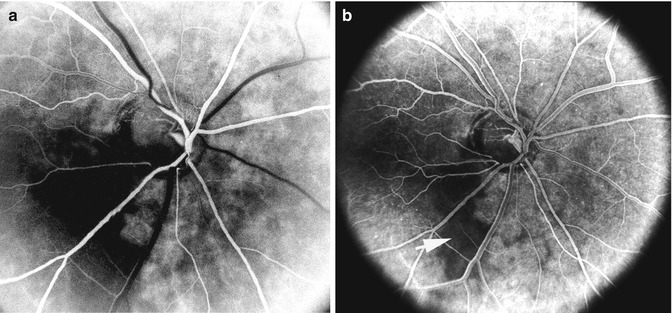
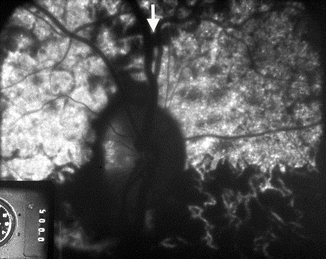
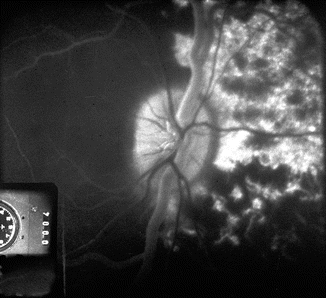
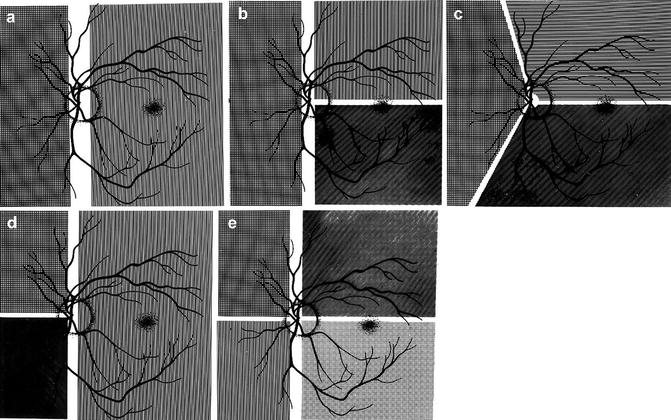
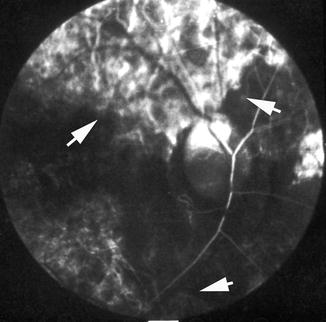

Fig. 4.6
Fluorescein fundus angiogram, of the left eye of a 63-year-old woman with arteritic anterior ischemic optic neuropathy, shows normal filling of the area supplied by the lateral PCA but no filling of the area supplied by the medial PCA (including the entire optic disc, with no perception of light) (Reproduced from Hayreh [80])

Fig. 4.7
Fluorescein fundus angiogram of the right eye showing medial PCA filling but no that of the lateral PCA – pattern reverse of that seen in Fig. 4.6

Fig. 4.8
Fluorescein fundus angiogram, of the left eye of a 45-year-old woman with non-arteritic anterior ischemic optic neuropathy, shows non-filling of the watershed zone (indicated by arrows) between the lateral and medial PCAs (Reproduced from Hayreh [42])

Fig. 4.9
Fluorescein fundus angiogram, of the right eye of a 75-year-old man with AION and negative temporal artery biopsy for giant cell arteritis, shows normal filling of the area supplied by the lateral PCA (including the temporal half of optic disc) but no filling of the area supplied by the medial PCA (including the nasal half of optic disc) (Reproduced from Hayreh [42])

Fig. 4.10
Fluorescein fundus angiograms, of the right eye of a 69-year-old woman with low-tension glaucoma and cupping of the lower half of the optic disc with corresponding visual field defect. (a) Shows a filling defect in the inferior temporal quadrant of the choroid and lower half of the optic disc. (b) 5 s after a shows non-filling of the lower half of the watershed zone (indicated by arrow) and the involved lower half of the optic disc (Reproduced from Hayreh [42])

Fig. 4.11
Fluorescein fundus angiograms of a normal right eye of a healthy cynomolgus monkey (after experimental occlusion of the central retinal artery to unmask vessels from the PCA circulation in the prelaminar region of the optic disc), with four PCAs (similar to Fig. 4.13e), show after the first injection of the dye filling of the upper half of the choroid and optic disc with a well-demarcated horizontal border. In fact this region is supplied by two superior PCAs – one medial and the other lateral, and the arrow indicates the watershed zone between the two superior PCAs (Reproduced from Hayreh [20])

Fig. 4.12
Fluorescein fundus angiograms of the same eye as in Fig. 4.11, after the second injection of the dye. It shows filling of the superior nasal quadrant, before the filling of the superior temporal quadrant of the choroid seen in Fig 4.11. The marked fluorescence of the optic disc was due to the previous injection of fluorescein (Reproduced from Hayreh [20])

Fig. 4.13
Diagrammatic representations of some examples of the locations of the borders between the territories of the various PCAs: (a) with two PCAs, one medial and the other lateral; (b–d) with three PCAs in different combinations, (b, c) have one medial and two lateral PCAs and (d) one lateral and two medial; and (e) with four PCAs, two medial and two lateral (Reproduced from Hayreh [46])

Fig. 4.14
Fluorescein fundus angiogram, of a normal human right eye, shows a Y-shaped watershed zone (indicated by arrows) between the superior, lateral, and medial PCAs (Reproduced from Hayreh [30])
Areas Supplied by the SPCAs and Their Branches
Experimental occlusion of one or another SPCA in 87 eyes of rhesus monkeys [28] resulted in a segmental choroidal filling defect in the area of distribution of the occluded artery (Figs. 4.15, 4.16, and 4.17). There are no anastomoses between the adjacent short PCAs [28, 32]. As described above, the SPCAs are of two types (Fig. 4.2) [17, 67]:

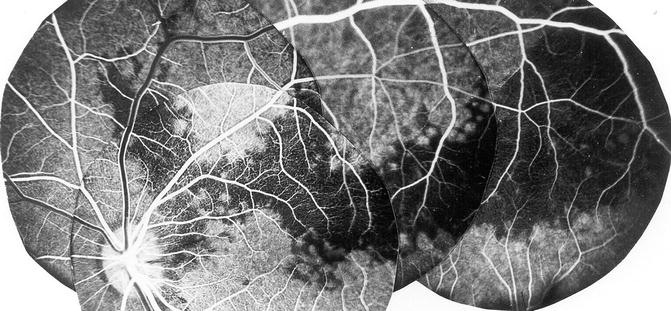
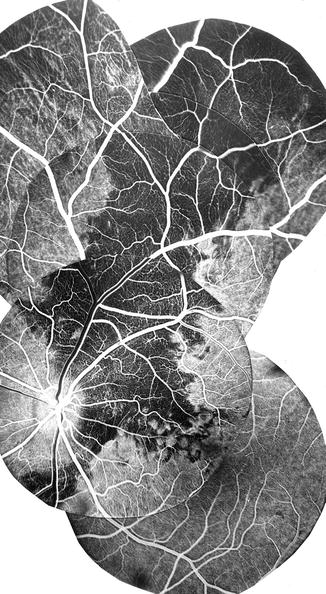

Fig. 4.15
Fluorescein fundus angiogram after occlusion of one of the mid-temporal SPCAs showing filling defects in the choroid in the region of the occluded SPCA (Reproduced from Hayreh [28])

Fig. 4.16
Fluorescein fundus angiogram after occlusion of one of the mid-temporal SPCAs showing filling defects in the choroid in the region of the occluded SPCA (Reproduced from Hayreh [28])

Fig. 4.17
Fluorescein fundus angiogram after occlusion of a superior temporal SPCA, showing filling defects in the choroid in the region of the occluded SPCA (Reproduced from Hayreh [28])
(a)
Paraoptic SPCAs: Branches from these go to the corresponding parts of the ONH, peripapillary choroid, the Circle of Haller and Zinn (when present), and recurrent branches to the retrolaminar ONH pial vascular plexus. Thus, available evidence indicates that all these branches of the paraoptic SPCAs constitute the main source of blood supply to the ONH. The circle of Haller and Zinn is discussed below and at length elsewhere [68].
(b)
Distal SPCAs: Each of these arteries supplies a sector of the choroid, usually extending from the posterior pole to the equator (Figs. 4.18 and 4.19) [28]. Each sector varies markedly in shape, size, and location and has irregular margins [13, 18, 28, 33]. The various sectors fit together like pieces of a jigsaw puzzle. Further subdivisions of the SPCAs supply correspondingly smaller segments of irregular shape and size, so that the blood supply by the various choroidal arteries has a geographic pattern – the smaller the artery, the smaller the size of the geographic area. Finally, each terminal choroidal arteriole supplies a lobule of the choriocapillaris (Fig. 4.20) [27, 28, 33, 36].
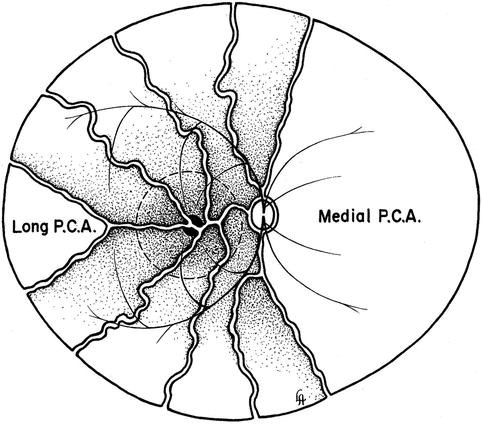
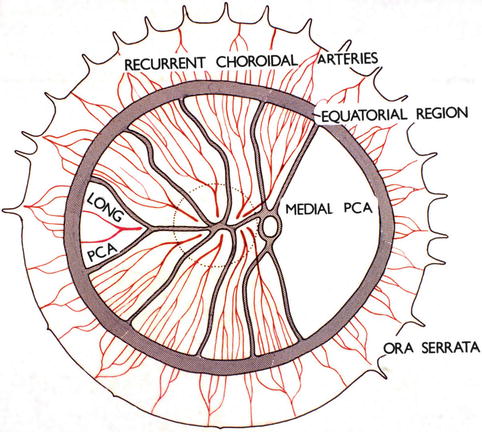
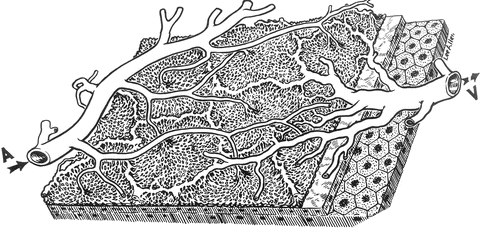

Fig. 4.18
Diagrammatic representation of distribution by various temporal short PCAs and their watershed zone in posterior part of fundus. Dotted circle in region of distribution of temporal short PCAs represents macular region. Areas of supply by medial PCA and temporal long PCA are also shown (Reproduced from Hayreh [36])

Fig. 4.19
Diagrammatic representation of the distribution by the various ciliary arteries in the choroid and their watershed zones. The choroid posterior to the equator is supplied by the medial and lateral PCAs. In the area supplied by the lateral PCA are shown the segments supplied by the various short PCAs and the one by the long PCA, with the watershed zones between them (dotted circle in this area indicates the macular region). Recurrent choroidal arteries from the anterior ciliary arteries and supposedly the greater arterial circle of the iris supply in front of the equator. The watershed zone between the anterior and posterior choroidal arteries lies in the equatorial region (Reproduced from Hayreh [36])

Fig. 4.20
A three-dimensional schematic representation of the choriocapillaris pattern. A choroidal arteriole, V choroidal veins (Reproduced from Hayreh [27])
In addition to that, the short PCAs give off branches during their intraorbital course to the pial plexus on the optic nerve, at their site of penetration into the scleral and episcleral arterial plexus and during their course in the sclera to the arterial circle of Haller and Zinn (Fig. 4.21).
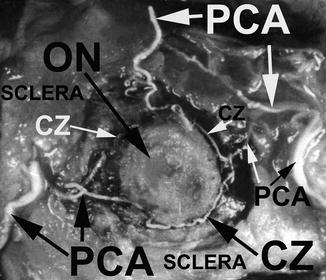

Fig. 4.21
A neoprene latex cast of the circle of Haller and Zinn (CZ), exposed in the sclera around the ONH, with its feeding arteries, i.e., medial and lateral PCAs. Optic nerve (ON) had been cut close to the eyeball (Reproduced from Hayreh [81])
Areas Supplied by the Long PCAs [29]
There was a stereotyped description of the long posterior ciliary arteries (LPCAs) in the textbooks, virtually unchanged since the early accounts [82, 83]. According to these earlier accounts, there were two such arteries, one medial and the other lateral (Fig. 4.2), arising from the ophthalmic artery either independently or by a common trunk, and sometimes one or the other arose from the ophthalmic artery in common with the central retinal artery or SPCAs [82, 84, 85]. From this description, it seems they were confused with the Main PCAs. The arteries pierced the sclera nasal and temporal to the optic nerve (the former 3.6 and the latter 3.9 mm from the optic nerve [85, 86]) and ran horizontally at first in a scleral canal (3–7 mm long [82] and 3–5 mm long [86]), and then in the suprachoroidal space, as far forward as the posterior part of the ciliary body, where each artery divided into two main divisions – superior and inferior. The superior and inferior divisions of the two LPCAs united to form the major arterial circle of the iris in the anterior part of the ciliary body behind the root of the iris. Of the anterior ciliary arteries (usually two from each rectus muscle except that there is only one from the lateral rectus), some joined the greater arterial circle of the iris and others the LPCAs or their branches [83]. The LPCA, according to all these descriptions, gave no branches throughout its entire course until it divided into its two main divisions in the ciliary muscle. It was held that recurrent choroidal branches, supplying the anterior part of the choroid up to the equator and anastomosing with the SPCAs at the equator, arose from the greater arterial circle of the iris, anterior ciliary arteries, and terminal part of the LPCAs [8, 9]. No anastomoses between the SPCAs and the recurrent choroidal arteries at the equator were found in rabbits [5, 82]. Thus, according to all the available descriptions, the LPCAs supplied the choroid only in front of the equator via the recurrent choroidal arteries, with no supply to the choroid behind the equator.
There are many studies on the effects of experimental occlusion of the LPCAs on the intraocular pressure and the anterior segment of the eye [87–93]. Most of these studies have been carried out in rabbits, and the production of ocular hypotony, phthisis bulbi, iris neovascularization, cataract, and anterior segment necrosis has been recorded. The occurrence of anterior segment ischemia following diathermy, scleral buckling, and some other procedures for retinal detachment has been attributed to interference with the LPCAs. Unsuccessful attempts were made to find out if occlusion of the LPCA alone or with anterior ciliary arteries would help to control the raised intraocular pressure (lOP) in glaucoma patients [90, 94–101]. Conflicting claims of temporary falls in lOP, of variable amounts and for varying periods, have been made.
My study, in 22 rhesus monkeys after occlusion of the temporal LPCA and evaluation by fluorescein fundus angiography, revealed that the LPCA supplies the choroid forward from the point where it enters the eyeball, and occlusion of the LPCA produces no significant effect on the IOP or the eye.
Supply of the Choroid by the Temporal Long Posterior Ciliary Artery
Contrary to the earlier classical concept, my in vivo study in rhesus monkeys revealed that the artery invariably supplies an area of the choroid posterior to the equator, starting almost immediately from the point where it joins the choroid and extending forward (Figs. 4.18, 4.19, 4.22, and 4.23). The temporal LPCA joins the choroid 5 mm temporal to the optic disc, and the area of the choroid supplied by the artery starts 4.6–9 mm (average 6.2 ± 1.0 mm) from the disc (Figs. 4.18, 4.19, 4.22, and 4.23). In rhesus monkeys, the distance of the equator and ora serrata on the temporal side from the optic disc is 13 and 16.5 mm, respectively. This shows that the supply by the LPCA to the choroid extends well posterior to the equator.
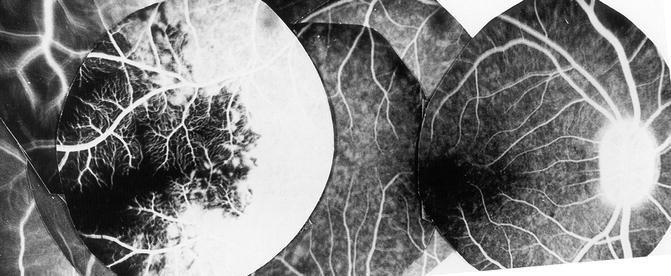
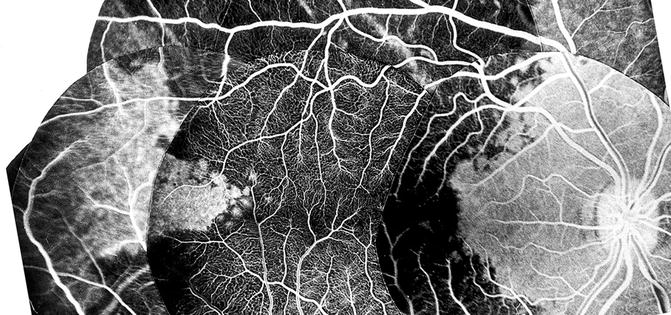

Fig. 4.22
Composite fluorescein fundus angiograms of rhesus monkey eye, after cutting the temporal long PCA, showing a sectoral non-fluorescence of the choroid in the extreme temporal periphery, temporal to the macular region (Reproduced from Hayreh [29])

Fig. 4.23
Composite fluorescein fundus angiograms of rhesus monkey eye, after cutting the temporal short PCAs but leaving intact long PCA, showing the filling of a sector of the posterior choroid temporal to the macular region in the area supplied by the intact temporal LPCA (Reproduced from Hayreh [29])
The distribution is sectoral, with the apex of the sector pointing backward (Figs. 4.18, 4.19, 4.22, and 4.23). The distribution by the artery is segmental, with little direct anastomosis with the adjacent choroidal arteries. On follow-up of the eyes with occluded temporal LPCA, however, no fundus lesion was seen. These two statements may seem to contradict one another. The explanation for the nonoccurrence of fundus lesions on the experimental occlusion of small branches of the PCAs is discussed elsewhere [33, 102].
In patients, chorioretinal lesions are known to occur due to occlusion of the LPCA. I found a whitish lesion in the distribution of the LPCA 3 days after the onset of severe posterior scleritis (Fig. 4.24). This resolved through pigmentary degeneration and left a sector-shaped pigmentary degenerative lesion. This lesion resembled in appearance those which I produced on experimental occlusion of the PCAs [23, 103]. Amalric [104] reported similar triangular chorioretinal lesions situated in the area corresponding to the distribution of the LPCAs (Fig. 4.25). In posterior scleritis, the LPCAs would be much more vulnerable to occlusion (because of their long, oblique course through the scleral canal) than the SPCAs, which have a straight, extremely short course through the sclera. The scleral canal for the LPCA and the accompanying nerves is 3–7 mm long in man [82], divided into two compartments (for artery and nerves) by a fibrous septum, almost parallel to the outer surface of the sclera in the posterior part and more steep and narrow in the anterior part. Thus the canal forms a flat bow with its concavity inward [82, 83]. Scleral edema and swelling in this region would occlude the canal and consequently the LPCA. The difference in the response of the choroid to choroidal ischemia in experimental animals and patients is understandable; it is due to multiple factors, e.g., the normal healthy vascular bed in young healthy monkeys, as compared to the diseased vascular bed in patients, and the frequent intrachoroidal involvement of small arteries in patients, as compared to the extraocular occlusion produced in experimental animals, the former being much more likely to produce ischemic lesions than the latter.
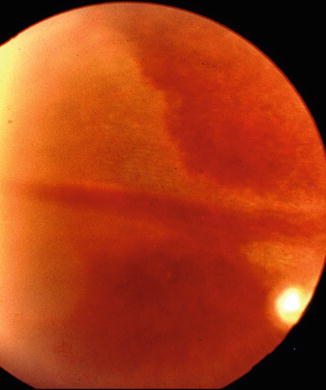


Fig. 4.24
A whitish choroidal ischemic lesion in the distribution of the long PCA 3 days after the onset of severe posterior scleritis (Reproduced by kind courtesy of Dr. Michael H. Goldbaum)

Fig. 4.25
A triangular chorioretinal lesions situated in the area corresponding to the distribution of the resolved ischemic lesion of long PCA (Reproduced by kind courtesy of late Dr. Pierre Amalric)
Ruskell [5] mentioned that the LPCAs of rabbits, while passing through the sclera, give off several branches, which contribute to the choroidal vascularization. This seems to support the findings of my study.
The temporal LPCA is intimately related to the insertion of the inferior oblique muscle; the artery pierces the sclera at the nasal margin of the attachment of the tendon, and the scleral canal of the artery lies along the line of attachment of the tendon. This fact may be important to bear in mind in surgical intervention on the inferior oblique muscle. It is not stressed adequately in the literature.
Each LPCA extends radially in the horizontal meridian – one on the medial and the other on the lateral side (Fig. 4.2). On the temporal side, the long PCA supplies a sector of the choroid temporal to the macular region, with its apex posteriorly (Figs. 4.18, 4.19, 4.22, and 4.23). A detailed account of the blood supply by the LPCAs is given elsewhere [29]. Like the short PCAs, their smaller subdivisions supply smaller geographic segments. In addition to supplying a sector of the peripheral choroid, each artery also supplies a small sector of the ciliary body and iris on the medial and lateral sides.
Peripapillary Choroid
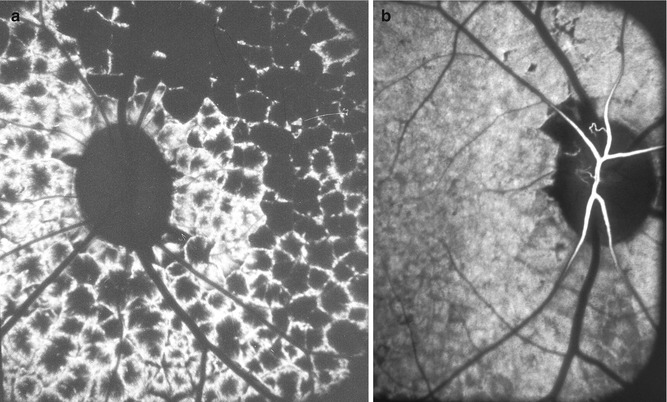
This part of the choroid is supplied by branches from the short PCAs. As in the rest of the choroid, postmortem cast studies have consistently shown free anastomoses in the peripapillary choroidal arteries so as to form a continuous vascular network [12]. Shimizu and Ujiie [12] in their choroidal casts found that branches of the short PCAs run centripetally while undergoing a series of bifurcations; an incomplete arterial ring was found along the margins of the optic nerve head, formed by the numerous arteries entering the arterial ring, and the latter was regarded by the authors as an interarterial anastomosis. They concluded that the peripapillary choroidal arterioles therefore cannot be regarded as end arteries.
The peripapillary choroid is a very important part of the choroidal vasculature, since it is the main source of blood supply to the prelaminar and retrolaminar parts of the optic nerve (Fig. 4.1) [12, 19, 20, 32, 42, 46, 105]. There are some serious misunderstandings prevailing on the subject. It is not uncommon to find authors stating that if the peripapillary choroid is so very important in the blood supply of the anterior part of the optic nerve, then why does the optic disc remain normal in eyes with peripapillary choroidal atrophy? The branches supplying the optic nerve arise essentially from the main arteries in the peripapillary choroid and not from the small branches, and the choriocapillaris stops abruptly at the disc margin (Figs. 4.26 and 4.27). In almost all peripapillary choroidal atrophy which is ophthalmoscopically visible, the loss is limited to the choriocapillaris, fine vessels, and overlying retinal pigment epithelium, with the large vessels still intact (Fig. 4.28).