The Optic Nerve in Glaucoma
Douglas R. Anderson
THE NORMAL RETINA AND OPTIC NERVE
The approximately 1 million axons that form the optic nerve arise from the retinal ganglion cells and course toward the optic disc in a well-known pattern (Fig. 48.1). From the nasal retina, the fibers take a straight course toward the disc. Axons that originate temporal to the fovea arc around the macula to enter the upper and lower poles of the optic nerve head according to whether the ganglion cell is above or below the temporal raphe, with very slight overlap. There is some individual variation, such that bundles from these arcades may enter somewhat to the nasal side of the disc, crossing through bundles from above or below, which also enter the poles of the disc (Brad Fortune, Portland Oregon, personal communication). The axons from the region around the fovea pass directly to the temporal quadrant of the disc in the papillomacular bundle.
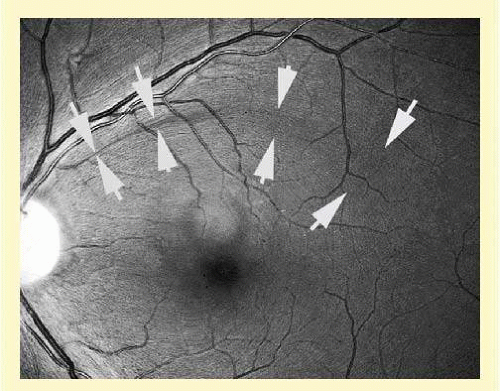
The axons maintain an orderly grouping in the retina as bundles partitioned by sheets of Müller cells.1,2,3 A sheen reflected from the many bundles taking parallel courses gives the retina a striated appearance4 that is most prominent where the nerve fiber layer is thickest— near the disc, especially in the arcuate bundles arriving at the poles of the disc from the temporal arcades.5,6 The sheen results from a backscatter from parallel cylindrical structures in the nerve fiber bundles,7,8 approximately a third to a half of which is from axonal microtubules.9,10 The microtubules also produce a birefringence,11,12 which causes retardation of polarized light transmitted through the nerve fiber layer and reflected back from a deeper layer. The retardation can be measured by laser scanning polarimetry.13,14,15 An attentive observer can see the striae of light reflected from these bundles and recognize their partial or total absence when bundles of axons disappear in glaucomatous or other forms of optic atrophy (Fig. 48.2).4,16,17,18,19 The manner of layering of axons within intraretinal bundles may not be exactly the same in all primate species.2,3,6,20 A basic orderliness is present in the retina and is maintained into the orbital portion of the optic nerve, where a rearrangement of axons separates those that will cross over at the chiasm from those that do not, maintaining, however, a systematic arrangement through the chiasm and beyond to the lateral geniculate body.21,22,23,24 Upon arriving at the lateral geniculate body, the axons are still organized and synapse in an orderly array. Discrete scotomas and other defects due to damage to nerve fiber bundles are produced by very localized insults in the retinal nerve fiber layer or in the optic nerve head. Pressure on the posterior optic nerve by a mass lesion typically produces a central scotoma or diffuse visual dysfunction, but occasional instances of more localized damage in this region produce well-localized nerve fiber bundle defects, attesting to the existence of orderly bundles in this region also.21,22,23,24
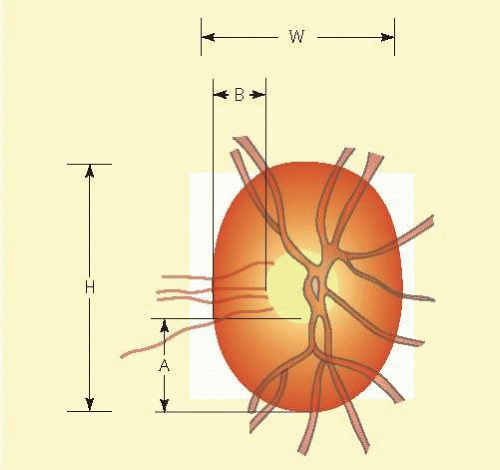
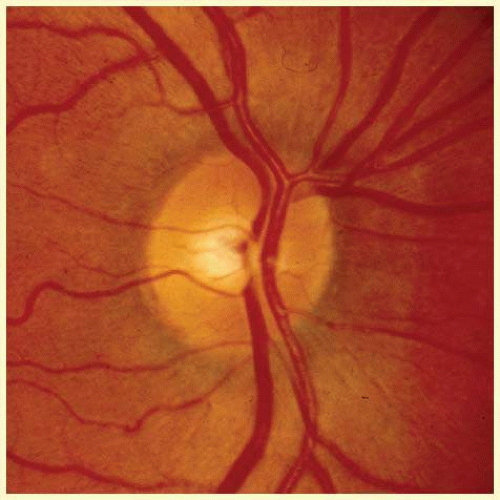
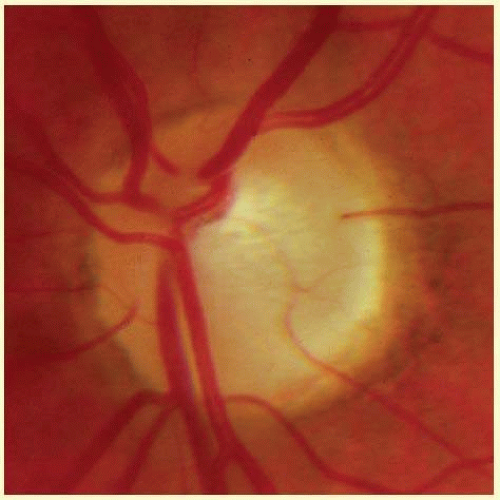
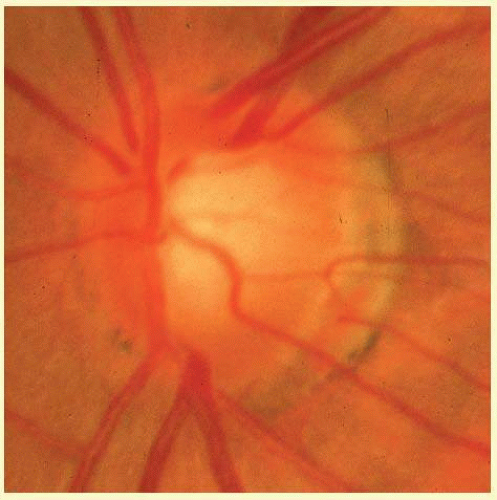
In the retinal nerve fiber layer, axons converge toward the optic disc and turn to enter the optic nerve through openings in the outer retina, Bruch membrane, the choroid, and the sclera, which together form the walls of a channel, which may be called the neural canal. Viewed on end, as by ophthalmoscopy, the neural canal that enclosed the optic nerve head tissue appears as a disc (the optic disc) that is typically somewhat taller than it is wide (Figs. 48.3 and 48.4). A physiologic excavation results whenever this neural canal is larger than required for the 1 million axons along with the supporting astroglial cells and blood vessels. The size of the excavation depends on how much larger the size of the canal is compared with the area needed to accommodate all the axons (Figs. 48.5, 48.6 and 48.7).25,26 Because the number of nerve fibers entering the upper and lower poles of the disc is greater than that in the temporal and nasal sectors, the boundary of the physiologic excavation is more or less circular (Figs. 48.3, 48.4 and 48.5).27
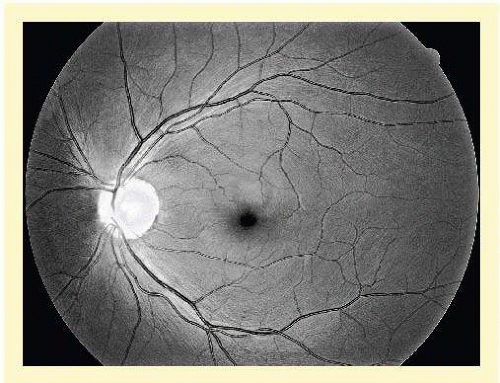 FIG. 48.1 Fundus photograph shows the normal pattern of the retinal nerve fiber layer. (Courtesy of Anja Tuulonen, M.D., Tampere, Finland, and Jukka Veijola, Oulu, Finland). |
As the optic nerve emerges through the lamina cribrosa, the nerve becomes of greater diameter because of fibrous septa that support and guide the nerve fiber bundles through the orbit, plus the added bulk due to myelination of the axons.28 For this reason, the wall of the scleral canal and the vaginal sheaths angle outward in a prototype configuration with the optic nerve exiting perpendicular to the ocular surface. In such instances, the nerve exits perpendicular to the scleral surface in all meridians, and the cup may have a straight cylindrical wall or have a pure conical shape. Figures 48.5, 48.6 and 48.7 were selected to illustrate optic nerve heads of this type.
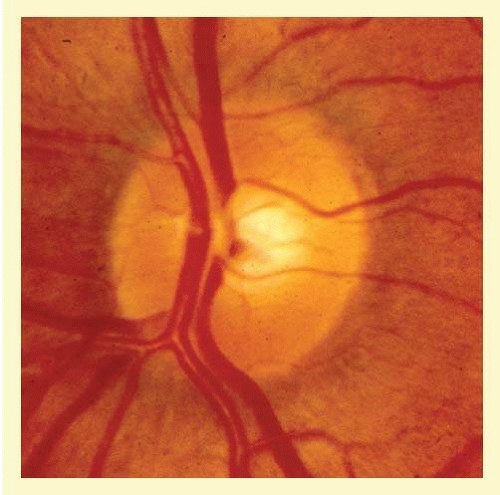 FIG. 48.4 Color fundus photograph that shows the features illustrated schematically in Figure 48.3. |
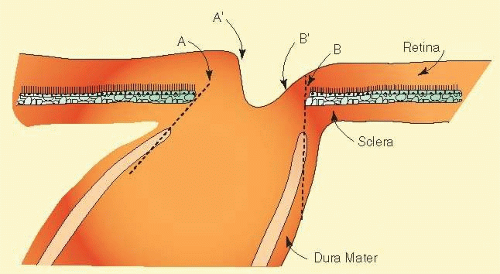
However, more often than not, the nerve exits the eye obliquely heading superiorly and nasally. The shape of the physiologic cup is affected by any obliquity of the exit through the neural canal. The wall of the cup is steep in sectors where the wall of the canal is angled outward.28 In sectors where the canal has a wall perpendicular to the ocular coats, or perhaps even angled inward, the cup has a sloping wall (Fig. 48.8). Very often discs exit through a modestly sloped canal so that the superior and nasal sides of the cup are steep, corresponding to an outward slope of the canal wall in those sectors, whereas the temporal and inferior wall of the cup is sloped, corresponding to a perpendicular orientation of the wall of the canal, or less frequently even an inward slope (Figs. 48.8 and 48.9). Infrequently the exit is so oblique that the wall of the cup undermines the neuroretinal rim. There is considerable individual variation in the size and shape of the chorioscleral canal as it exits the eye, and therefore of the optic cup.
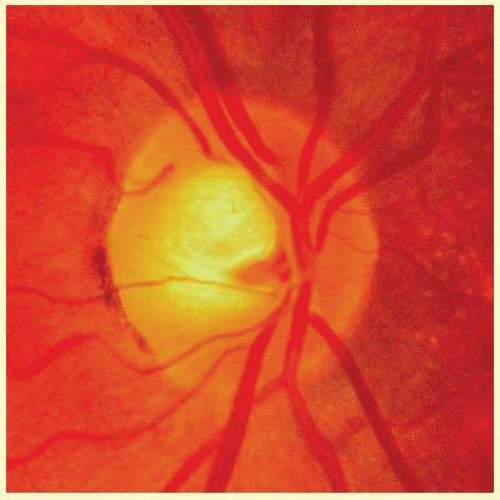 FIG. 48.6 Color fundus photograph of a disc with a somewhat larger neural canal. The cup is proportionately larger than in Figure 48.5. Note that the comparative abundance of the neuroretinal tissue remains greater at the upper and lower poles than elsewhere around the circumference. |
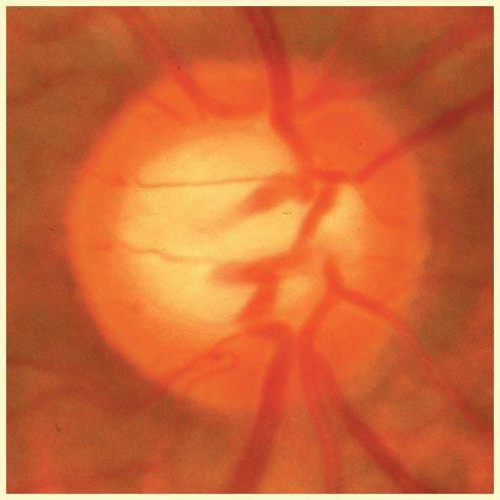 FIG. 48.7 Configuration of an optic disc with a large neural canal through the outer ocular coats. The disc is only slightly taller than it is wide. The radial width of the neuroretinal rim is greater at the poles of the disc than it is nasally and temporally; thus, again, the physiologic cup is round. Note that the vessels are the same size as those in Figure 48.5, and that a large physiologic cup is the expected finding when the disc diameter is large relative to the size of the vessels. (See Figure 48.30 for another example of a large disc with a large physiologic excavation.) |
As noted, the neural canal through which the optic nerve exits from the eye traverses several layers of peripapillary tissue: the outer retina, the pigment epithelium, Bruch membrane, the choriocapillaris, the outer choroid, and the finally the sclera. Variations in the disc appearance are based on variations in the histologic components of the optic nerve head and these peripapillary tissue layers. Anteriorly the axon bundles are supported by a scaffold of fibrous astrocytes. Posteriorly the opening in the sclera is bridged by supportive layers of perforated connective tissue, the lamina cribrosa. It is not composed of perforated sheets of scleral tissue but is a unique connective tissue of different developmental origin and of different microscopic structure. It is inserted onto the scleral connective tissue, while the scleral coat itself turns posteriorly to form the vaginal sheaths of the optic nerve behind the eye. The scleral collagen fibers, randomly arranged elsewhere, become arranged in a circular manner around the scleral opening. In some eyes (probably most), in all or some meridians, a flange of sclera extends as far forward as Bruch membrane, forming something of a connective tissue wall for the neural canal, which is lined by a glial coat.29,30 The connective tissue sleeve is known as the “border tissue” as first designated by Elshnig,29 although confusingly Jacoby31 used the term “border tissue” to refer to the glia that line the inner surface of what Elshnig termed the “border tissue.” The connective tissue cuff, Elshnig’s border tissue, can be seen ophthalmoscopically in most eyes as a “scleral rim” around the disc (Figs. 48.10 and 48.11). Usually (nearly always), Bruch membrane, devoid of retinal pigment epithelium (RPE), extends over the peak of the cuff and enhances its brightness when viewed by ophthalmoscopy. The opening in Bruch’s membrane is most often the narrowest channel through which the nerve fibers exit. This white rim is not visible or conspicuous if hidden beneath a thick, capillary-rich layer of nerve fibers or beneath an extension of the RPE (Fig. 48.11).
With or without a conspicuous scleral flange or termination of Bruch membrane, the several chorioretinal layers may not be perfectly aligned with one another. Perhaps most typically, as the optic nerve exits obliquely in an upper nasal direction, the lower temporal sector of the disc viewed ophthalmoscopically is seen to have a crescent of misaligned edges of the several chorioretinal layers, such that the outer choroid (which is thinner near the optic nerve than elsewhere) and the sclera may be left uncovered by the choriocapillaris and pigment epithelium (Figs. 48.12 and 48.13). In highly myopic eyes, the crescent is often more directly temporal, rather than inferotemporal, and in yet other eyes, any part or even the entire circumference may have retraction of the various chorioretinal layers. The sector of the disc with the most dominant crescent, resulting from oblique exit canal of the nerve, may mark the sector of the disc that is most susceptible to injury from elevated intraocular pressure32,33; for example, the lower temporal sector of the disc is most frequently the first sector affected with nerve loss and cupping.
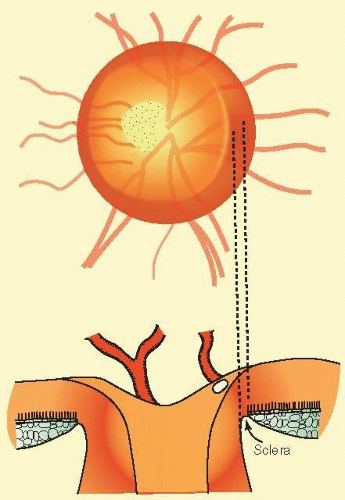 FIG. 48.10 A common anatomic configuration in which a flange of sclera marks the boundary of the disc, separating the choroid from the disc tissue. |
Based on clinical appearance, the peripapillary crescent has been descriptively divided into zones alpha and beta. Zone beta, nearest the optic disc, is a region of exposed sclera or choroid not covered by RPE. Zone alpha, at the boundary between the peripapillary crescent and halo, is a transition zone of irregular pigment epithelium.33,34
The peripapillary crescent is often an anatomic result of developmental configuration associated with an oblique neural canal exiting the eye. Sometimes the exposure of the choroid results from atrophy of the RPE, in addition to any preexisting absence of RPE. This occurs naturally with age in a small percentage of people, and somewhat more often in those with glaucoma.35 However, many eyes, even with severe glaucoma, do not develop such atrophy of the RPE.36
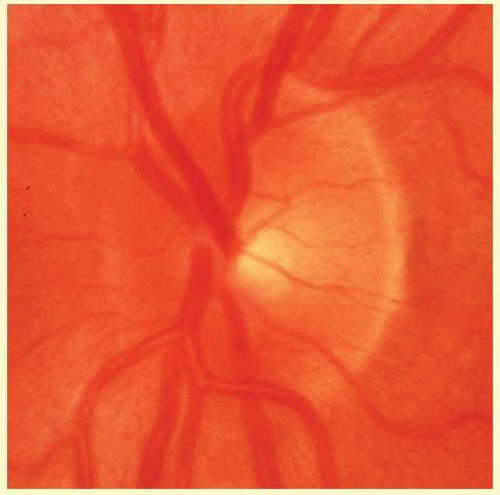 FIG. 48.11 Color photograph of an optic disc with the configuration shown schematically in Figure 48.10. The scleral flange is prominent temporally and can be perceived nasally but is difficult to see where the major vessels and thick layer of axons pass over it just nasal to the upper and lower poles. |
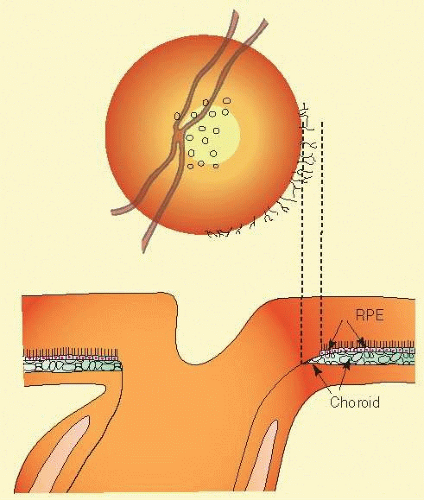 FIG. 48.12 Peripapillary crescent due to misalignment of the several layers of the retina, choroid, and sclera. The choroid is not covered completely by the choriocapillaris and RPE. |
PRESSURE-INDUCED DAMAGE
In the distant past, some believed that the damage to the optic disc in glaucoma occurred as the result of some sort of local sclerosis. Elevated intraocular pressure was thought by some not to be the cause but simply an accompanying manifestation of a sclerotic process that affected both the optic disc and the trabecular meshwork. Seeming evidence for this idea was a dissociation between the degree of cupping and the degree of pressure elevation,37,38,39 which is now attributed to individual variation in susceptibility of the optic nerve to damage from intraocular pressure.40 To understand glaucoma, it is very important to recognize that the level of intraocular pressure is an important factor in the development of glaucomatous optic nerve damage, but that factors about which we know little determine what pressure level will prove harmful and how much damage it will do. Thus, glaucoma may occur with an intraocular pressure in the normal range, and it may be absent with considerable elevation of intraocular pressure.
The causative role of intraocular pressure is evident from cases of secondary glaucoma and from the fact that nerve damage is produced by elevated intraocular pressure in glaucoma produced experimentally in animals. Moreover, when the intraocular pressure is consistently asymmetric between the two eyes, the eye with higher pressure nearly always has more damage to the optic nerve41,42 Experienced clinicians agree that progressive damage to the disc is halted when satisfactory lowering of intraocular pressure is achieved,43 and randomized clinical trials have now confirmed that occurrence of damage is reduced with lowered intraocular pressure.44,45,46,47 Factors that determine the susceptibility of the optic nerve to pressure-induced injury must vary in amount from one person to another to explain the individual variation the degree of damage from a given pressure, or stated differently, what level of pressure that proves harmful.
Although the occurrence of damage does correlate with the level of intraocular pressure (and with its correlate, the resistance to aqueous humor outflow), the relationship to age is equally strong.37,38 Family history and other factors affect the occurrence and severity of injury. Although the pathogenic process is not completely understood, it is clear that a number of other properties are as important as the intraocular pressure, or even more important, in determining the degree and rapidity of nerve damage and visual loss in glaucoma. The other abnormal traits that interact with the intraocular pressure to cause harm are presumably more frequently present or quantitatively more abundant when damage occurs at modestly elevated intraocular pressure, or even with pressure within the “normal” range as found in eyes without disease. In such cases, the nonpressure factors may be more easily detected or even be conspicuous. In contrast, the nonpressure abnormal traits that contribute to the pathogenic process may be absent or in lesser quantity in cases in which the nerve is not damaged despite abnormally elevated intraocular pressure (i.e., cases of “ocular hypertension”).
The pathophysiologic mechanism by which intraocular pressure produces optic nerve damage, the exact nature of all the susceptibility factors, and how they act together remain unknown.48 One hypothesis is that there is an abnormal or inadequate ability to regulate the blood flow when circulation is challenged by the intraocular pressure. Another idea is that pressure-induced deformation of the lamina cribrosa initiates injury to the lamina cribrosa and the axons passing through it. At present, we know some features that place an eye at risk when the pressure is elevated,37,38,49,50,51,52 but we can be certain that a nerve is vulnerable only when we actually observe that the damage has begun.40 Presently, the intraocular pressure is the only risk factor in the pathogenic mechanism that we know how to measure and to treat.
The susceptibility factors seem in large part to be in the constitution of the person, either because there are systemic influences (e.g., vascular status) or because both eyes of a person possess similar anatomic and physiologic traits on a genetic or developmental basis. Thus, if one eye has suffered damage from elevated intraocular pressure, the other eye is likely to follow if it, too, has or develops a pressure elevation.49 The two eyes may not always be damaged to the same degree. When one eye carries a somewhat higher pressure, even in the statistically normal range, it is usually the more damaged eye.41,42 In like manner, an anatomic difference between the two eyes (e.g., marked anisometropia), or some unidentified factor(s), can make one eye more susceptible than the other, even with symmetric intraocular pressures.
It appears that the susceptibility factors are absent, or present only to a very low degree, in most people, especially the young. Therefore, many people can carry an intraocular pressure mildly or moderately above the normal range for many years without damage, but there is a limit, and few eyes will tolerate a pressure of 50 or 60 mm Hg for very long. Approximately half of eyes in an epidemiologic populations study that have an intraocular pressure of 35 mm Hg will show harm.39 A minority of eyes without glaucoma with an intraocular pressure in the 20s will develop glaucomatous damage within a decade,47 but most eyes tolerate pressures outside the normal range for a long time. On the other hand, some people are particularly susceptible to damage, and they suffer damage from a pressure in the mid to high teens (called, “normal-tension glaucoma”, or in order to emphasize that it is not a distinctly separate disease, referred to by the awkward longer phrase “primary open-angle glaucoma with intraocular pressure in the statistically normal range”). Damage from pressure in the low teens occurs even more rarely. Yet, the facts are that at least a third of all glaucoma occurs with an intraocular pressure in the statistically normal range (between 10 and 21 mm Hg, most often at the upper end of this range), that the average pressure at which glaucoma is found is only 23 to 24 mm Hg, and that a small proportion of individuals with glaucoma present with more distinctly abnormal pressure, say above 30 mm Hg. These facts point to the considerable role that traits other than intraocular pressure play in producing glaucoma. The fact is that a pressure that is high enough that it is found infrequently among normal eyes (i.e., statistically outside the normal range) does not make that pressure unsafe. In contrast, a few eyes are not safe with a pressure that is found in many normal eyes (statistically “within normal limits”).
In typical cases of chronic glaucoma, the damage occurs over quite a number of years, although the rate of visual loss varies considerably among individuals.53 After the most susceptible axons are already damaged, damage to other, previously unaffected axons (progression of glaucoma) may be speculated to occur as a result of one or more of the following:
1. Age causes a reduction in resistance to damage so that the next most susceptible axons become damaged at the pressure that is being carried.
2. Weakening of the disc by partial cupping increases the susceptibility of the remaining axons.
3. Intraocular pressure progressively rises or has higher peaks of pressure as glaucoma continues.
4. Damage depends on not only the pressure level, but also the time of exposure to the pressure (thus, the damage occurs as a cumulative effect of the force exacted by the intraocular pressure).
However, in the infrequent susceptible individual, glaucomatous cupping of the disc can proceed rapidly, within a month or two, even with moderate pressure elevation (25 to 30 mm Hg). This situation is most typically encountered in secondary glaucomas of abrupt onset. Many eyes do not suffer harm from a sudden pressure rise to 25 or even 35 mm Hg from trauma or uveitis, but a few eyes may rapidly develop severe or nearly total cupping in a few weeks after a rapid, but modest elevation of pressure (e.g., 25 to 30 mm Hg). Therefore, cases with a recent rise in pressure must be monitored closely until it is clear whether the particular eye is being damaged or not.
Sometimes in chronic glaucoma, the cupping and field loss are recognized to progress in a series of small episodes, perhaps as a result of episodic upward swings of intraocular pressure or episodic swings of constitutional factors that decrease the tolerance level. For example, a dramatic episode of transient vulnerability is sometimes blamed for nerve damage and field loss in patients who have had a hemodynamic crisis.54 The optic nerve damage may not be noticed for some time, and after being detected, patients with such a history are less likely than others to progress,54 perhaps provided they have returned to good health and if episodes of severe ill health are not recurrent.
Progression may be accompanied by the occurrence of splinter hemorrhages that appear transiently within the bundles of axons in or at the boundary of the optic nerve head, either before or after a new visual field defect is noted,55,56,57,58,59,60,61 but they are also observed without progression. The implications for the pathogenic process of glaucomatous damage are not clear. They are seen most often in cases of normal-tension glaucoma, perhaps because they most likely have an abundance of susceptibility factors with pressure that has not been lowered very much. A clear relationship of hemorrhages and progression to physiologic stress affecting the vascular system is not often observed, but anecdotally has been seen in unique situations, for example, in a person with vascular dysregulation who recently exerted himself skiing in the cold at high altitudes (personal observation).
The most characteristic and most easily recognized feature of glaucomatous damage to the optic nerve is that one region of the disc is affected earlier and more severely than another. When axon loss occurs more predominantly in a certain sector of the disc, cupping characteristically extends toward the disc rim in that sector, and the neuroretinal rim becomes locally narrow. If the preferential loss is in the most typical location (in the polar sectors), the cup expands vertically27 and may form a notch at the disc rim, most often at the poles of the optic disc.
In contrast, when axon loss is evenly diffuse, the cup expands concentrically,62,63 and there is an accompanying generalized depression of the field. Probably many or even most cases begin with some degree of diffuse loss of tissue and stretching of the support tissue behind and around the optic nerve head, but in most cases it is not long before the damage is more marked in some regions. Thus, as the cup extends and deepens vertically, there is also temporal and nasal unfolding of the cup. The loss of axons, with thinning of the neuroretinal rim and local expansion of the cup, is typically more prominent in sectors of the disc with exposed choroid or sclera (crescent, beta zone).32,33
There are exceptions. In infants, the ocular coat stretches, including the exit neural canal. The disc and its cup may become larger with or without loss of axons, either diffusely or in local areas. Likewise, in younger people with secondary glaucoma, often with particularly high pressure compared with the usual pressure in the low 20s typical of primary glaucoma in older people, there may be elastic stretching of the lamina cribrosa and surrounding sclera to produce an enlarged disc with a deep round cup that is more conspicuous than any localized loss of axons. Such cases may have a reversal of the anatomic excavation when the pressure is lowered therapeutically, most strikingly after surgical lowering of the intraocular pressure. The loss of axons and of function in the visual field may be less than expected from the prominently enlarged and deepened cup. In many, however, the optic nerve is left with axonal damage in a characteristic localized pattern superimposed on the elastic stretching that can be caused by a very high pressure in a young eye.
VISUAL EFFECTS
It is important that visual function be evaluated along with the funduscopic findings, and some general principles are mentioned here briefly, because diagnosis of glaucomatous optic nerve damage may remain uncertain unless confirmed by corresponding effects on vision. Loss of axons naturally affects visual function in the regions of the retina from which the axons arose. Color sense,64,65,66,67 contrast sensitivity,67,68,69,70 and acuity71,72 are among the functions lost, in addition to the differential intensity visual threshold typically measured in classic visual-field testing.73,74,75,76,77,78,79 A certain proportion of nerve fibers must be lost in the affected retinal region before the diminished visual function is recognizably reduced from the normal range, is reduced from the previously documented visual status, or is out of keeping with the threshold in the surrounding unaffected (or less affected) region or the opposite vertical hemifield, or is asymmetric with the opposite eye.80,81,82,83 It is fortunate that in nearly all cases, axon damage is more pronounced in certain bundles than in others almost from the beginning, even if a mild diffuse loss may be an early event in many. A general depression from diffuse loss of half the axons may not be recognized, but a localized scotoma may also be recognized after a major loss of axons in a confined region accounting for loss of fewer than 5% of the total number of axons, whereas no axons are lost in 95% of the disc area.
When the axon loss is more diffuse, the resulting mild, widespread depression may remain within the range of normal visual sensation. It is difficult to recognize the cause as glaucoma, because such general depression is more often caused by mild cataract often seen in the same age group. A glaucomatous cause for diffuse axon loss and visual change can be recognized if comparison is made either with a previously documented baseline in that person or with the opposite, less affected eye, if corresponding clinical findings are found (such as abnormal intraocular pressure, glaucomatous appearance of the optic nerve), and if other potential causes (cataract, anisometropia or amblyopia, etc.) are absent.67,69,74,75,77,81,82,84,85,86 It is difficult to assess the effect of any media opacity (most often cataract) that is nearly always present to some degree and of its scattering effect, which produces veiling glare and consequently reduces contrast for field testing. At present this is a qualitative estimate, but it is technically feasible to quantify if instruments that measure glare or scatter are developed and come into common usage.
Fortunately, bilaterally equal diffuse loss of nerve fibers with bilaterally equal broadening of the cup and bilaterally equal loss of visual field is infrequent in glaucoma, or is found only in the earliest stages. Therefore, glaucomatous nerve damage and visual-field loss can usually be recognized by localized damage at an acceptably early stage by a combination of fundus examination and automated perimetry.83 Diagnostic findings include characteristic and corresponding localized cupping and field loss (in the early stages removing effect of cataract or early glaucomatous generalized depression), asymmetry of visual field in the upper and lower positions,82 by asymmetry of cupping or field loss between the two eyes81 that is in keeping with asymmetry of intraocular pressure or other physiologic asymmetry, or by a change from a previous baseline disc photograph or baseline threshold perimetry unexplained by cataract development.
Both examinations of the optic disc are used not only for diagnosis, but also for monitoring the disease for progressive damage. Two general principles are used, documenting a change compared with the baseline condition and monitoring some quantified feature to define a rate of change. Some of these are described in sources cited here,83,87,88 but there are several others developed in the last decade and in evolution. Of particular interest are new methods to evaluate the rate of progression both for visual fields and various measurements of the optic nerve and retina; they are not be covered in this chapter.
FUNDUSCOPIC FINDINGS
When evaluating the optic disc clinically, for diagnosis or for monitoring progression, it is helpful to have a mental image of the underlying tissue changes. These are 2-fold: altered configuration of the lamina cribrosa and other connective tissues and loss of neuronal tissue.
Connective tissue changes consist of elastic stretching caused by the force of the intraocular pressure, and the elastic stretching may be reversible, to a greater degree in younger eyes than older ones. When the intraocular pressure stress is reduced, release of the elastic backward bowing of the lamina cribrosa and outward stretching of the scleral border of the optic nerve head apparently accounts for the “reversibility of cupping” sometimes seen when the intraocular pressure is lowered in younger patients with glaucoma, particularly infants (see below). However, with prolonged stretching of the disc, the connective tissue becomes disrupted with collapse of the layered sheets of the lamina cribrosa, and ultimately a tissue remodeling that is not reversible. The connective tissue disruption with backward collapse and widening of the scleral opening thus produce an irreversible permanent excavation, which is only in small part due to the loss of axons and perhaps of some astroglia. The permanent change in the lamina cribrosa and other connective tissue accounts for the fact that glaucomatous atrophy of the optic disc differs in appearance from the other optic atrophies, which have only loss of axons, but no stretching of the connective tissues. When there is considerable backward bowing of the lamina cribrosa without much axon loss, the disc may be severely cupped with surprisingly little visual effect; this is sometimes seen in young people, and particularly in infants. Vision loss after pressure-induced distension of the globe and of support tissue in the disc (with cupping) in infants may be mainly the result of ametropic or anisometropic amblyopia, although axon loss obviously can contribute.
Adding to the excavation due to backward bowing and collapse of the lamina cribrosa is the loss of axons, as well as reduction or displacement of the astroglia and microvasculature within the optic nerve head. This “neuroretinal tissue” constitutes the rim around the cup and with disease diminishes in radial width as any physiologic excavation that may have been present expands. Thus, there are two anatomic features: the boundary of the cup expands outward and the disc surface recedes backward. In patients in whom slight backward movement of the disc surface is the earliest sign recognized, the disc has sometimes been described as “saucerized.”89 The lateral expansion of the cup is easier to appreciate than the backward recession of the surface, and often glaucomatous cupping has gauged as an increase in the cup diameter compared with the disc diameter (cup/disc ratio). More recently, various imaging instruments can make more sophisticated measurements, such as neuroretinal rim area, cross-sectional thickness of the retinal nerve fiber layer, and even measurement of the ganglion cell layer thickness in the macula—which are covered in a separate chapter. Our attention here is on the appearance of the optic disc on clinical examination, mainly with regard to diagnostic recognition of glaucoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree